Advanced Synthesis of Levomilnacipran Intermediates: Scalable Routes for Global Pharma Supply
Introduction to Next-Generation Levomilnacipran Intermediate Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antidepressant agents like Levomilnacipran, particularly focusing on the efficient production of its key chiral intermediate, (1S,2R)-1-phenyl-2-(phthalimide)methyl-N,N-diethyl-cyclopropanecarboxamide. Patent CN103694162A introduces a transformative methodology that addresses long-standing challenges in impurity control and process scalability. This innovation pivots away from harsh acidic conditions traditionally associated with thionyl chloride activations, offering a milder, high-yielding alternative suitable for modern GMP manufacturing environments. By utilizing specific activating reagents such as chloromethylene dimethyl ammonium chloride or methanesulfonyl chloride, the process ensures the integrity of the diethylamide moiety is preserved throughout the synthesis. This technical advancement is not merely a laboratory curiosity but represents a viable industrial solution for reliable pharmaceutical intermediate supplier networks aiming to reduce lead time for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
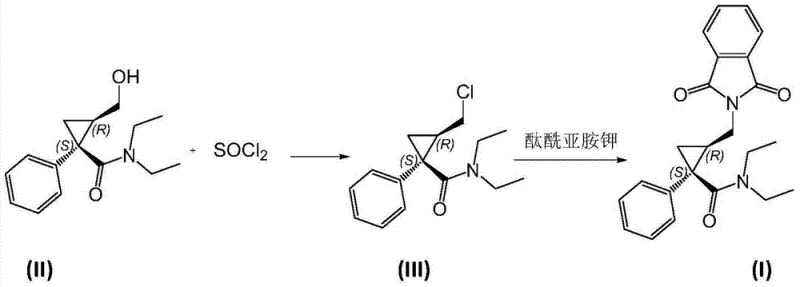
Historically, the synthesis of this critical cyclopropane derivative relied heavily on the use of thionyl chloride (SOCl2) to activate the hydroxyl group, as documented in prior art such as Patent WO2010086394. While chemically feasible, this conventional approach suffers from severe drawbacks due to the generation of strong acidic byproducts during the reaction. The highly acidic environment promotes unwanted side reactions, specifically the de-ethylation of the diethylamide group, leading to the formation of persistent impurities designated as VI, VII, and VIII. These de-ethylated byproducts possess physicochemical properties strikingly similar to the target molecule, making their removal via standard recrystallization techniques extremely difficult and costly. Furthermore, alternative literature methods, such as those reported in the Journal of Medicinal Chemistry, have demonstrated poor overall efficiency with yields dropping significantly after purification steps, rendering them economically unviable for cost reduction in pharmaceutical manufacturing on a commercial scale.
The Novel Approach
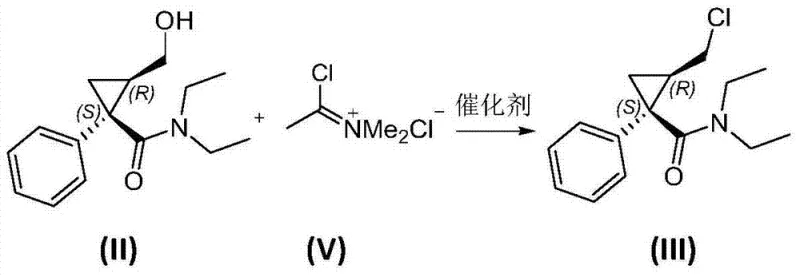
In stark contrast to the harsh conditions of the past, the novel methodology described in CN103694162A employs sophisticated activating agents that operate under significantly milder parameters. The core innovation involves converting the hydroxyl precursor (II) into an activated intermediate (IX) using reagents like chloromethylene dimethyl ammonium chloride (V) or methanesulfonyl chloride. This strategic shift eliminates the generation of free mineral acids that drive degradation, thereby virtually suppressing the formation of de-ethyl impurities. The reaction conditions are温和 (mild), typically proceeding at temperatures between -10°C and 40°C, which enhances operational safety and control. This approach not only simplifies the workup procedure but also allows the intermediate to be telescoped directly into the subsequent substitution step without rigorous purification, representing a paradigm shift for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Hydroxyl Activation and Substitution
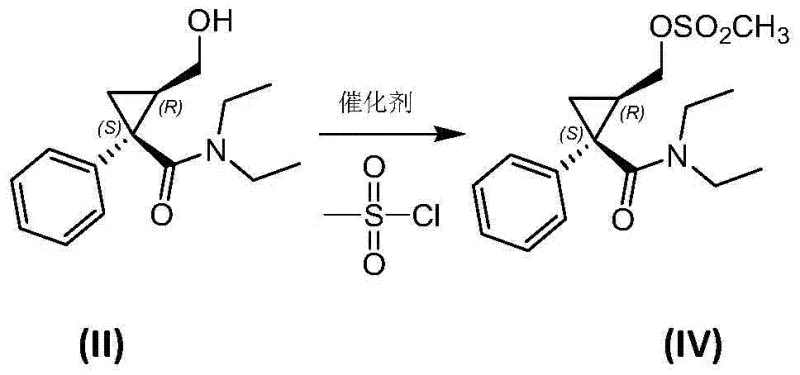
The mechanistic elegance of this process lies in the precise activation of the primary alcohol on the cyclopropane ring without compromising the sensitive amide functionality. When using chloromethylene dimethyl ammonium chloride, the reagent acts as a potent chlorinating agent that converts the hydroxyl group into a chloride leaving group (Intermediate III) with exceptional efficiency, often exceeding 95% conversion. Alternatively, the use of methanesulfonyl chloride transforms the hydroxyl into a mesylate ester (Intermediate IV), another excellent leaving group for nucleophilic attack. Both pathways avoid the protonation of the amide nitrogen that typically triggers N-dealkylation in acidic media. The subsequent step involves a classic SN2-type nucleophilic substitution where potassium phthalimide attacks the activated carbon center. This displacement occurs cleanly in high-boiling solvents like toluene or DMF, driven by the stability of the phthalimide anion and the quality of the leaving group established in the first step.
From an impurity control perspective, the absence of strong acid catalysts is the defining feature that ensures product purity. In traditional routes, trace amounts of HCl can catalyze the hydrolysis or elimination of the ethyl groups on the nitrogen, creating impurities that co-elute with the product. By maintaining a neutral to slightly basic environment (often utilizing organic bases like triethylamine if necessary, though the novel reagents often require none), the structural integrity of the N,N-diethyl group is locked in place. This mechanistic control translates directly to a cleaner crude reaction profile, reducing the burden on downstream purification units. For R&D directors, this means a more predictable impurity profile and a reduced risk of batch failure due to out-of-specification related substances, facilitating faster regulatory filings and technology transfer.
How to Synthesize (1S,2R)-1-phenyl-2-(phthalimide)methyl-N,N-diethyl-cyclopropanecarboxamide Efficiently
The synthesis protocol outlined in the patent provides a clear, two-step sequence that is amenable to standard reactor setups found in most fine chemical facilities. The process begins with the activation of the alcohol precursor in a halogenated solvent like dichloromethane, followed by a substitution reaction in an aromatic or amide solvent. The operational simplicity allows for flexible scheduling and resource allocation within a multipurpose plant.
- Activate the hydroxyl group of the cyclopropane precursor (II) using chloromethylene dimethyl ammonium chloride or methanesulfonyl chloride under mild conditions to form the leaving group intermediate.
- Perform a nucleophilic substitution reaction between the activated intermediate and potassium phthalimide in a high-boiling solvent like toluene.
- Isolate the final product (I) through standard aqueous workup and solvent removal, achieving high purity without complex recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification train. By eliminating the formation of tenacious de-ethyl impurities, the need for multiple recrystallization cycles or complex chromatographic separations is removed. This reduction in processing steps directly correlates to significant cost savings in terms of solvent consumption, energy usage for heating and cooling, and labor hours. Furthermore, the raw materials required, such as the activating reagents and potassium phthalimide, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialty syntheses reliant on exotic catalysts.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the high efficiency of the reaction and the elimination of yield-lossing purification steps. Traditional methods often suffer from substantial material loss during the removal of acidic byproducts and structurally similar impurities. By preventing these impurities from forming in the first place, the overall mass balance of the process is optimized. This leads to a higher throughput per batch and a reduction in the cost of goods sold (COGS), allowing for more competitive pricing strategies in the generic pharmaceutical market without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply timeline. The mild temperature requirements (-10°C to 40°C) mean that the process does not demand specialized cryogenic equipment or high-energy steam inputs, making it easier to replicate across different manufacturing sites globally. Additionally, the ability to potentially telescope the intermediate without isolation reduces the total cycle time from raw material intake to finished intermediate. This agility allows suppliers to respond more rapidly to fluctuations in demand for Levomilnacipran APIs, ensuring continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, moving away from bulk thionyl chloride usage reduces the generation of corrosive sulfur dioxide and hydrogen chloride gases. This lowers the load on scrubber systems and minimizes the hazardous waste footprint of the facility. The process aligns well with green chemistry principles by improving atom economy and reducing solvent intensity. For large-scale operations, this translates to easier permitting, lower waste disposal costs, and a safer working environment for plant personnel, all of which are critical factors for long-term sustainable manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating technology transfer packages or conducting vendor audits for critical raw materials.
Q: How does this new method prevent de-ethylation impurities?
A: Unlike conventional thionyl chloride methods that generate strong acidic conditions leading to N-dealkylation, this patent utilizes mild activating reagents like chloromethylene dimethyl ammonium chloride. This avoids the formation of difficult-to-remove de-ethyl impurities (VI, VII, and VIII), significantly simplifying downstream purification.
Q: What are the key advantages for large-scale manufacturing?
A: The process operates under mild temperatures ranging from -10°C to 40°C for the activation step, reducing energy costs and safety risks associated with exothermic reactions. Furthermore, the intermediate can often be used directly in the next step without purification, streamlining the production workflow and reducing solvent consumption.
Q: Is the starting material readily available for commercial production?
A: Yes, the synthesis begins with (1S,2R)-1-phenyl-2-hydroxymethyl-N,N-diethyl-cyclopropanecarboxamide, a known precursor. The activating reagents, such as methanesulfonyl chloride or the in-situ generated chloromethylene dimethyl ammonium chloride, are industrially accessible, ensuring a robust and continuous supply chain for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S,2R)-1-phenyl-2-(phthalimide)methyl-N,N-diethyl-cyclopropanecarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process validation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect even trace levels of de-ethyl impurities, guaranteeing that every batch meets the exacting standards required for API synthesis. Our commitment to quality ensures that your supply chain remains uninterrupted and compliant with global regulatory expectations.
We invite you to leverage our technical capabilities to optimize your Levomilnacipran supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this advanced chemistry can drive value for your organization. Let us partner with you to deliver high-quality intermediates that power the next generation of antidepressant therapies.
