Advanced Copper-Catalyzed Synthesis of 4-Bromo-2,3,5,6-Tetrafluorophenylacetic Acid for Commercial Scale-Up
Introduction to Patent CN101417945A Technology
The pharmaceutical and fine chemical industries are constantly seeking safer, more efficient pathways for synthesizing complex fluorinated intermediates. Patent CN101417945A introduces a groundbreaking method for preparing 4-bromo-2,3,5,6-tetrafluorophenylacetic acid, a critical building block for various bioactive molecules. Unlike conventional routes that rely on hazardous cyanation reactions, this innovation employs a copper-catalyzed nucleophilic substitution strategy using diethyl malonate. This shift not only mitigates significant safety risks associated with toxic reagents but also streamlines the purification process by generating benign byproducts such as ethanol and carbon dioxide. The technical breakthrough lies in the precise control of reaction conditions, utilizing cuprous salts and strong bases to achieve selective substitution on the pentafluorobenzene ring.
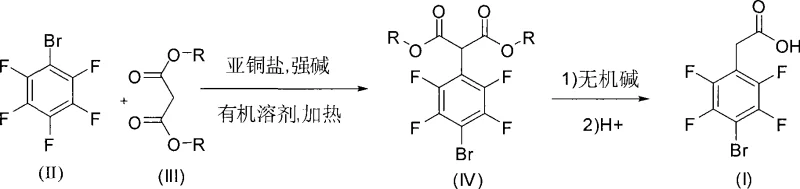
For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is vital for evaluating its commercial viability. The process begins with the reaction of 2,3,4,5,6-pentafluorobromobenzene and diethyl malonate under nitrogen protection, ensuring an inert atmosphere that prevents oxidative degradation of the sensitive intermediates. The subsequent hydrolysis and decarboxylation steps are designed to be operationally simple, avoiding the need for extreme pressures or temperatures that often complicate scale-up. By adopting this methodology, manufacturers can transition away from legacy processes that burden supply chains with high compliance costs and environmental liabilities, positioning themselves as leaders in sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylacetic acid derivatives, particularly those with heavy halogenation like the tetrafluoro-bromo motif, has relied heavily on cyanation-hydrolysis sequences or carbonylation reactions. These traditional pathways present severe drawbacks, primarily centered around the use of highly toxic cyanide salts which necessitate rigorous safety protocols and expensive waste disposal systems. Furthermore, carbonylation methods often require precious metal catalysts like rhodium, which introduce significant raw material costs and potential contamination issues that are difficult to resolve in final API products. The operational complexity of managing these hazardous materials often leads to extended lead times and increased regulatory scrutiny, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a malonate ester displacement strategy that fundamentally alters the risk profile of the synthesis. By employing diethyl malonate as the nucleophile, the process eliminates the need for cyanide entirely, replacing it with a reagent that is widely available and significantly less toxic. The reaction proceeds through a well-defined intermediate, the diethyl 2-(4-bromo-2,3,5,6-tetrafluorophenyl)malonate, which can be isolated or processed directly into the hydrolysis step. This continuity allows for a more streamlined workflow where the byproducts are easily removed, and the final decarboxylation yields the target acid with high structural integrity. This method represents a paradigm shift towards greener chemistry without sacrificing yield or purity.
Mechanistic Insights into Cu-Catalyzed Nucleophilic Substitution
The core of this synthetic innovation is the copper-catalyzed nucleophilic aromatic substitution, a reaction that leverages the electron-withdrawing nature of the multiple fluorine atoms on the benzene ring. In the presence of a strong base like potassium tert-butoxide, the diethyl malonate is deprotonated to form a reactive enolate species. The cuprous salt, preferably cuprous iodide, acts as a mediator that facilitates the attack of this enolate on the para-position of the pentafluorobromobenzene ring. This specific catalytic cycle ensures that the substitution occurs selectively at the desired position, minimizing the formation of ortho-substituted isomers which are common impurities in non-catalyzed thermal reactions. The stability of the copper complex under the reaction conditions of 70-110°C allows for complete conversion of the starting material over a period of 10 to 25 hours.
Following the substitution, the mechanism transitions to a classic ester hydrolysis and decarboxylation sequence. The addition of inorganic bases such as sodium hydroxide cleaves the ester bonds, releasing ethanol which is subsequently removed under reduced pressure to drive the equilibrium forward. The resulting dicarboxylic acid intermediate is thermally unstable upon heating in an acidic medium, leading to the loss of one carboxyl group as carbon dioxide. This decarboxylation step is crucial for establishing the final acetic acid side chain structure. The entire sequence is designed to maximize atom economy while ensuring that the fluorine atoms, which are critical for the biological activity of downstream derivatives, remain intact throughout the harsh basic and acidic treatments.
How to Synthesize 4-Bromo-2,3,5,6-Tetrafluorophenylacetic Acid Efficiently
Executing this synthesis requires careful attention to the order of addition and temperature control to maintain the integrity of the catalytic system. The process begins with the preparation of the nucleophile in an anhydrous organic solvent, followed by the slow introduction of the electrophile to manage exothermicity. Detailed standard operating procedures regarding stoichiometry, specifically the molar ratios of base to substrate, are critical for optimizing yield and minimizing side reactions. For a comprehensive breakdown of the specific experimental parameters and workup protocols, please refer to the standardized guide below.
- Perform nucleophilic substitution of 2,3,4,5,6-pentafluorobromobenzene with diethyl malonate using cuprous iodide and potassium tert-butoxide in 2-methyltetrahydrofuran at 70-110°C.
- Hydrolyze the resulting diester intermediate with aqueous sodium hydroxide under reflux conditions to remove alcohol byproducts.
- Acidify the mixture to pH 1-3 and heat to induce decarboxylation, followed by recrystallization to obtain the high-purity final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patent technology offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for fluorinated intermediates. The elimination of cyanide-based reagents removes a major category of hazardous material from the facility, drastically reducing the costs associated with safety compliance, specialized storage, and hazardous waste treatment. Furthermore, the reliance on commodity chemicals like diethyl malonate and common copper salts ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with precious metal catalysts or specialized toxic reagents. This stability translates directly into more predictable pricing and reliable delivery schedules for long-term contracts.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the replacement of expensive and hazardous reagents with cost-effective alternatives. By avoiding the use of precious metals like rhodium or palladium, which are subject to significant market price fluctuations, manufacturers can stabilize their raw material costs. Additionally, the simplified workup procedure, which avoids complex heavy metal scavenging steps often required with transition metal catalysts, reduces the consumption of auxiliary materials and processing time. These efficiencies compound to lower the overall cost of goods sold, allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved due to the widespread availability of the key starting materials. 2,3,4,5,6-Pentafluorobromobenzene and diethyl malonate are produced by multiple suppliers globally, reducing the risk of single-source dependency. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, providing flexibility in production planning. This geographic and supplier diversity ensures that production schedules remain uninterrupted even during regional disruptions, securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor equipment and solvents like 2-methyltetrahydrofuran that are compatible with existing infrastructure. The environmental profile is markedly superior to traditional methods, as the primary byproducts are ethanol and carbon dioxide, which do not require complex remediation. This alignment with green chemistry principles simplifies the permitting process for new production lines and enhances the corporate sustainability profile. For supply chain leaders, this means faster time-to-market for new capacity expansions and reduced regulatory friction in environmentally stringent jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their production pipelines. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What are the primary safety advantages of this synthesis method compared to traditional cyanation?
A: Traditional methods often rely on highly toxic cyanide reagents which pose severe health risks and require specialized waste treatment. This patented method utilizes diethyl malonate, a low-toxicity nucleophile, generating only ethanol and carbon dioxide as byproducts, significantly reducing environmental impact and operator hazard.
Q: How does the copper-catalyzed mechanism improve impurity profiles?
A: The use of cuprous salts facilitates a controlled nucleophilic aromatic substitution that selectively targets the para-fluorine position relative to the bromine. This specificity minimizes the formation of regio-isomers common in non-catalyzed harsh conditions, simplifying downstream purification and ensuring higher purity specifications for sensitive pharmaceutical applications.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes robust reaction conditions (40-120°C) and common organic solvents like 2-methyltetrahydrofuran. The workup involves standard extraction and recrystallization techniques without requiring cryogenic temperatures or exotic reagents, making it highly adaptable for multi-kilogram to ton-scale production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-2,3,5,6-Tetrafluorophenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-bromo-2,3,5,6-tetrafluorophenylacetic acid meets the exacting standards required for pharmaceutical synthesis. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry with precision and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and sustainability in the competitive global market.
