Advanced Manufacturing of 6-Chloro-4-Iodoindazole for High-Value Kinase Inhibitors
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed halogenated indazoles at the forefront of oncology research. Patent CN102372675B introduces a groundbreaking methodology for the synthesis of 6-chloro-4-iodoindazole, a pivotal building block for next-generation therapeutics. This technical disclosure outlines a highly efficient three-step sequence starting from 3-nitro-5-chloro-2-methylaniline, leveraging Sandmeyer chemistry and precise cyclization protocols. The strategic placement of halogen atoms on the indazole core provides exceptional versatility for downstream functionalization, making it an indispensable asset for medicinal chemists targeting VEGFR and PDGFR pathways. By optimizing reaction conditions to minimize by-product formation, this process achieves a purity profile approaching 99%, addressing the stringent quality requirements of modern API manufacturing. 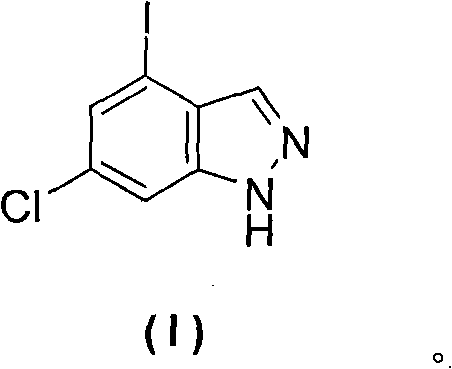
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted indazoles has been plagued by regioselectivity issues and harsh reaction conditions that compromise overall yield. Traditional routes often rely on multiple protection and de-protection steps to differentiate between reactive sites on the aromatic ring, leading to excessive waste generation and prolonged cycle times. Furthermore, conventional iodination methods frequently suffer from poor atom economy, requiring stoichiometric amounts of expensive iodine sources that drive up the cost of goods significantly. The presence of multiple halogens in earlier synthetic strategies often resulted in ambiguous coupling positions during subsequent cross-coupling reactions, necessitating difficult chromatographic separations that are impractical at a commercial scale. These inefficiencies create bottlenecks in the supply chain, delaying the availability of critical intermediates for clinical trial material production.
The Novel Approach
In stark contrast, the methodology described in CN102372675B utilizes a direct and linear synthetic pathway that eliminates unnecessary synthetic operations. By initiating the sequence with a readily available nitro-aniline derivative, the process capitalizes on the inherent directing effects of the existing substituents to ensure high regioselectivity during the Sandmeyer iodination step. The subsequent reduction and cyclization steps are optimized to proceed under mild conditions, preserving the integrity of the sensitive carbon-iodine bond which is crucial for later diversification. This approach not only simplifies the operational workflow but also significantly enhances the environmental profile of the manufacturing process by reducing solvent usage and hazardous waste. 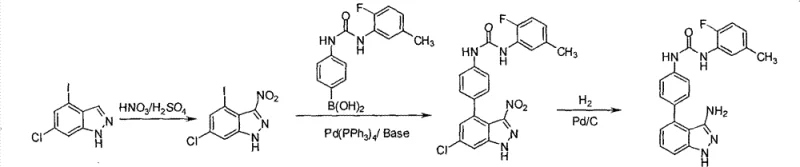
Mechanistic Insights into Sandmeyer Iodination and Cyclization
The core of this synthetic innovation lies in the precise control of the diazotization and subsequent iodination kinetics. In the first step, the formation of the diazonium salt is carefully managed at low temperatures (below 10°C) using inorganic protonic acids like hydrochloric acid, ensuring stable intermediate formation without premature decomposition. The introduction of potassium iodide then facilitates a smooth nucleophilic substitution, where the iodide ion displaces the diazonium group with high fidelity. This step is critical because any deviation in temperature or pH can lead to the formation of phenolic by-products or de-halogenated species, which would complicate downstream purification. The robustness of this Sandmeyer variant allows for the consistent production of the iodo-nitro intermediate with yields exceeding 84%, providing a solid foundation for the subsequent transformations.
Following the reduction of the nitro group to an amine, the final cyclization mechanism employs a unique combination of acetic anhydride and isoamyl nitrite. This reagent system generates the necessary nitrosating species in situ, which reacts with the acetylated amine to form the indazole ring closure. The use of isoamyl nitrite offers a safer and more controllable alternative to traditional nitrous acid gas, allowing for better heat management during the exothermic cyclization event. The final base-mediated deacetylation and aromatization step ensures that the product precipitates in a highly crystalline form, facilitating easy isolation via filtration. This mechanistic understanding allows process chemists to fine-tune parameters such as reagent molar ratios and addition rates to maximize throughput while maintaining the strict impurity profiles required for pharmaceutical grade materials.
How to Synthesize 6-Chloro-4-Iodoindazole Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity, making it accessible for both laboratory scale optimization and pilot plant campaigns. The process begins with the careful preparation of the diazonium species, followed by a straightforward workup that isolates the iodo-intermediate in high purity. The reduction step utilizes catalytic systems that can be easily filtered and recycled, aligning with green chemistry principles. Finally, the cyclization is performed in common solvents like glacial acetic acid or toluene, avoiding the need for exotic or highly toxic reagents. For detailed standardized operating procedures and specific parameter ranges, please refer to the technical guide below.
- Diazotization and Iodination: Treat 3-nitro-5-chloro-2-methylaniline with sodium nitrite in acidic conditions followed by potassium iodide to form the iodo-nitro intermediate.
- Catalytic Reduction: Reduce the nitro group to an amine using hydrazine hydrate and Raney-Ni or catalytic hydrogenation to yield the iodo-aniline derivative.
- Ring Closure Cyclization: React the amine with acetic anhydride and isoamyl nitrite, followed by base-mediated cyclization to form the final indazole core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost stability and sourcing reliability. The reliance on commodity chemicals such as sodium nitrite, potassium iodide, and hydrazine hydrate means that the supply chain is not vulnerable to the volatility associated with specialized reagents. This abundance of raw materials ensures consistent pricing and availability, mitigating the risk of production delays due to material shortages. Furthermore, the high yield and purity achieved in each step reduce the burden on quality control laboratories, as less time is spent troubleshooting failed batches or reprocessing off-spec material. This efficiency translates directly into lower operational expenditures and a more predictable budget for R&D projects.
- Cost Reduction in Manufacturing: The elimination of complex protection groups and the use of cost-effective reagents drastically lowers the raw material costs per kilogram of finished product. By streamlining the process to just three steps, labor costs and utility consumption are significantly reduced compared to legacy methods. The high recovery rates mean that less starting material is wasted, optimizing the overall material balance and improving the return on investment for every batch produced. Additionally, the simplified purification requirements reduce the consumption of expensive chromatography media and solvents, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: Sourcing 3-nitro-5-chloro-2-methylaniline is straightforward due to its widespread availability in the global chemical market, ensuring a secure supply line for long-term projects. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain fluctuations. This resilience allows manufacturers to maintain steady production schedules even during periods of market instability, ensuring that downstream drug development timelines are not compromised by intermediate shortages.
- Scalability and Environmental Compliance: The process is explicitly designed for scale-up, utilizing unit operations that are standard in modern chemical manufacturing facilities. The avoidance of heavy metal catalysts in certain steps and the use of recyclable solvents align with increasingly stringent environmental regulations, reducing the cost and complexity of waste disposal. The ability to run this synthesis in continuous mode or large batch reactors enables rapid expansion of capacity to meet surging demand from clinical trials or commercial launch phases without the need for significant capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-chloro-4-iodoindazole. These insights are derived directly from the patented technology and practical manufacturing experience, providing clarity for stakeholders evaluating this intermediate for their pipeline. Understanding these details is crucial for making informed decisions about process integration and supplier selection.
Q: What is the primary advantage of this synthesis route for 6-chloro-4-iodoindazole?
A: The primary advantage lies in the use of inexpensive, commercially available starting materials (3-nitro-5-chloro-2-methylaniline) and a streamlined three-step process that avoids complex purification steps, resulting in yields up to 99% purity suitable for GMP manufacturing.
Q: How does this intermediate support oncology drug development?
A: The 4-iodo and 6-chloro substitution pattern allows for versatile cross-coupling reactions, such as Suzuki coupling, enabling the rapid construction of diverse kinase inhibitor libraries targeting VEGFR and other critical oncology pathways.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly highlights the method's suitability for large-scale continuous production, utilizing standard reagents like acetic anhydride and isoamyl nitrite which are easily managed in industrial reactor settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Chloro-4-Iodoindazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the success of your drug discovery program depends on the quality and reliability of your starting materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage research through to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-chloro-4-iodoindazole meets the highest international standards. Our commitment to technical excellence means we can rapidly adapt the patented process to fit your specific timeline and volume requirements.
We invite you to collaborate with us to optimize your supply chain and accelerate your project milestones. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can add value to your organization. Let us handle the complexities of chemical synthesis so you can focus on delivering life-saving therapies to patients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
