Advanced Gold-Catalyzed Synthesis of Indanone Imine Derivatives for Oncology Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that can address the growing challenge of drug-resistant malignancies. Patent CN113292448B introduces a significant breakthrough in this domain with the disclosure of a series of indanone imine derivatives. These compounds are not merely structural variations but represent a strategic advancement in medicinal chemistry, specifically targeting solid tumors such as colorectal adenocarcinoma, osteosarcoma, and breast cancer. The core innovation lies in the efficient construction of the indanone imine skeleton, a pharmacophore known for its diverse biological activities ranging from analgesic to anti-inflammatory properties. By leveraging a sophisticated gold-catalyzed cyclization strategy, this technology overcomes the synthetic bottlenecks often associated with complex heterocyclic systems. For R&D directors and procurement specialists alike, understanding the underlying chemistry of this patent is crucial, as it offers a pathway to high-purity intermediates that are essential for the next generation of oncology therapeutics. The structural versatility allowed by the substituents R1 and R2 further enhances the potential for structure-activity relationship (SAR) optimization, ensuring that these molecules can be fine-tuned for maximum efficacy and minimal toxicity in clinical settings.
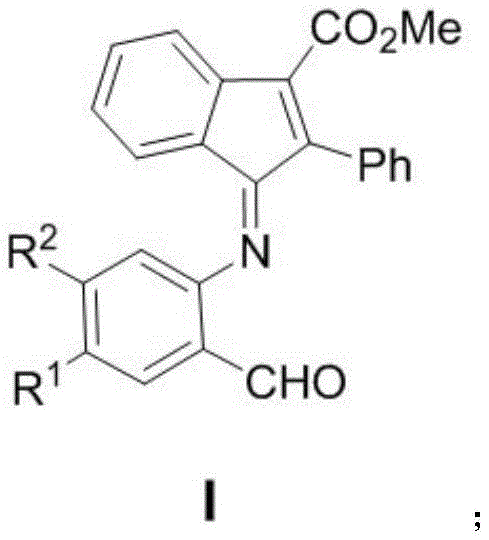
The development of effective anti-tumor agents often hinges on the ability to synthesize complex molecular architectures reliably. Historically, the construction of indanone derivatives has been plagued by limitations inherent in conventional synthetic methodologies. Traditional routes frequently involve multi-step sequences that require harsh reaction conditions, such as strong acids or high temperatures, which can lead to the degradation of sensitive functional groups and the formation of difficult-to-remove impurities. Furthermore, older methods often suffer from poor atom economy, generating substantial amounts of chemical waste that complicate downstream processing and increase environmental compliance costs. These inefficiencies translate directly into higher production costs and longer lead times, creating significant friction in the supply chain for critical pharmaceutical intermediates. The reliance on stoichiometric reagents rather than catalytic systems in legacy processes also limits the scalability, making it challenging to transition from gram-scale laboratory experiments to kilogram or ton-scale commercial manufacturing without extensive process re-engineering.
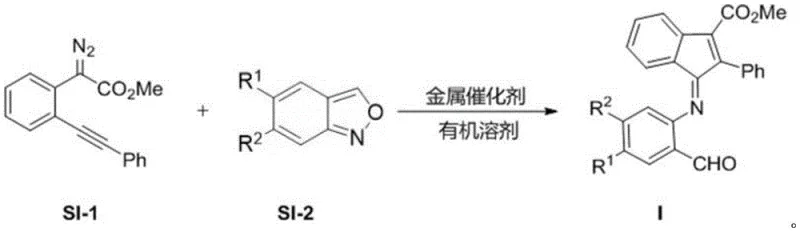
In stark contrast, the novel approach detailed in the patent utilizes a transformative gold-catalyzed reaction between a diazo compound (SI-1) and a benzisoxazole derivative (SI-2). This methodology represents a paradigm shift towards green and sustainable chemistry in pharmaceutical manufacturing. The reaction proceeds through a highly selective mechanism that constructs the target indanone imine framework in a single operational step. By employing a specialized gold complex catalyst, specifically [2-(dicyclohexylphosphine)-3,6-dimethoxy-2',4',6'-triisopropyl-1,1'-biphenyl]bis(trifluoromethanesulfonimide)gold, the process achieves exceptional conversion rates under mild thermal conditions. This eliminates the need for aggressive reagents and significantly reduces the energy footprint of the synthesis. The result is a streamlined workflow that not only accelerates the timeline for drug discovery but also ensures a consistent supply of high-quality material. For a reliable pharmaceutical intermediate supplier, adopting such advanced catalytic technologies is key to maintaining competitiveness and meeting the rigorous quality standards demanded by global regulatory bodies.
Mechanistic Insights into Gold-Catalyzed Cyclization
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic intricacies of the gold-catalyzed transformation. The reaction initiates with the activation of the diazo compound (SI-1) by the cationic gold(I) species. This interaction facilitates the extrusion of nitrogen gas, generating a highly reactive gold-carbene intermediate. This electrophilic species is then poised for nucleophilic attack by the nitrogen atom of the benzisoxazole ring (SI-2). The subsequent rearrangement and cyclization steps are meticulously orchestrated by the ligand environment of the gold catalyst, which stabilizes the transition states and directs the regioselectivity of the bond formation. This precise control is vital for preventing the formation of regioisomers or side products that could compromise the purity of the final API intermediate. The robustness of this catalytic cycle ensures that even with varying electronic properties of the substituents R1 and R2, the reaction maintains high efficiency. Such mechanistic reliability is a critical factor for R&D teams aiming to explore a broad chemical space without encountering unpredictable synthetic failures.
Furthermore, the impurity profile of the resulting indanone imine derivatives is markedly superior compared to those produced via non-catalytic routes. The high selectivity of the gold catalyst minimizes the generation of polymeric byproducts or decomposition fragments that are common in diazo chemistry. This inherent cleanliness of the reaction simplifies the purification process, often allowing for straightforward crystallization or basic chromatographic separation to achieve the stringent purity specifications required for clinical trials. From a quality control perspective, this means fewer batches are rejected due to out-of-specification impurities, thereby enhancing the overall yield and reducing the cost of goods sold. The ability to consistently produce material with a well-defined impurity profile also facilitates the regulatory filing process, as the characterization of degradation products becomes more manageable. This level of process control is indispensable for manufacturers aiming to secure long-term contracts with top-tier pharmaceutical companies.
How to Synthesize Indanone Imine Derivatives Efficiently
The practical implementation of this synthesis protocol is designed to be accessible yet rigorous, ensuring reproducibility across different laboratory and production environments. The process begins with the precise weighing of the diazo precursor and the benzisoxazole substrate, which are then dissolved in anhydrous 1,2-dichloroethane. The choice of solvent is critical, as it must effectively solubilize both reactants while remaining inert under the reaction conditions. Once the solution is prepared, the gold catalyst is introduced in a specific molar ratio to optimize turnover frequency without excessive metal loading. The reaction mixture is then heated to a controlled temperature of 60°C and maintained under stirring for approximately 24 hours. Monitoring the consumption of the starting diazo compound is essential to determine the endpoint, ensuring complete conversion before workup. Following the reaction, the crude product is subjected to filtration and purification, typically via column chromatography, to isolate the pure indanone imine derivative. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-yielding process.
- Prepare the reaction mixture by combining the diazo compound (SI-1) and benzisoxazole compound (SI-2) in anhydrous 1,2-dichloroethane solvent.
- Add the gold catalyst, specifically [2-(dicyclohexylphosphine)-3,6-dimethoxy-2',4',6'-triisopropyl-1,1'-biphenyl]bis(trifluoromethanesulfonimide)gold, to the solution.
- Heat the reaction system to 60°C and stir for 24 hours until the diazo compound is completely consumed, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing process. By condensing what would traditionally be a multi-step sequence into a single catalytic pot, the technology significantly reduces the consumption of solvents, reagents, and labor hours. This consolidation of steps directly translates to substantial cost savings in raw material procurement and waste disposal. Moreover, the use of a catalytic amount of gold, rather than stoichiometric quantities of expensive reagents, optimizes the cost structure of the synthesis. Although gold is a precious metal, its recovery and recycling in modern catalytic processes are well-established, further mitigating cost concerns. The operational simplicity also reduces the risk of human error during production, leading to higher batch consistency and reliability in delivery schedules.
- Cost Reduction in Manufacturing: The streamlined nature of this gold-catalyzed process eliminates the need for multiple isolation and purification stages that are typical in conventional synthesis. Each skipped step represents a saving in time, energy, and consumable materials. Additionally, the high atom economy ensures that a greater proportion of the starting mass is incorporated into the final product, minimizing waste generation. This efficiency is crucial for cost reduction in pharmaceutical intermediate manufacturing, allowing suppliers to offer competitive pricing without compromising on quality. The reduced complexity also lowers the capital expenditure required for equipment, as fewer reactors and separation units are needed to achieve the same output volume.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability. Because the process tolerates a range of substituents and operates under mild conditions, it is less susceptible to fluctuations in raw material quality or minor variations in process parameters. This resilience ensures a continuous and predictable supply of critical intermediates, reducing the risk of production delays that can impact downstream drug formulation. Furthermore, the availability of the starting materials, such as benzisoxazoles and diazo compounds, from established chemical vendors supports a resilient supply network. This reliability is paramount for pharmaceutical companies managing tight development timelines and needing to secure long-term sources for their pipeline candidates.
- Scalability and Environmental Compliance: Transitioning from laboratory benchtop to commercial scale is often a hurdle for new synthetic methods, but this protocol is inherently scalable. The use of standard organic solvents and moderate temperatures aligns well with existing infrastructure in fine chemical plants. The reduction in waste generation aligns with increasingly stringent environmental regulations, simplifying the permitting process and reducing the liability associated with hazardous waste management. The high yield and selectivity mean that less material needs to be processed to obtain the required quantity of API, further reducing the environmental footprint. This alignment with green chemistry principles not only meets compliance requirements but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indanone imine derivatives. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding the integration of this technology into their development pipelines. The answers reflect the current state of the art as presented in the intellectual property, providing a clear picture of the capabilities and limitations of the synthesis method.
Q: What are the primary therapeutic applications of these indanone imine derivatives?
A: These derivatives exhibit potent anti-tumor activity, specifically showing strong inhibitory effects against colorectal adenocarcinoma (HCT-116), osteosarcoma (SJSA-1), and breast cancer (MCF-7) cell lines, making them valuable candidates for oncology drug development.
Q: How does the gold-catalyzed method improve upon traditional synthesis routes?
A: The novel method utilizes a one-pot reaction strategy that significantly reduces the number of synthetic steps compared to conventional multi-step approaches. It operates under mild conditions (60°C) and offers high atom economy, minimizing waste generation and simplifying the purification process.
Q: Is this synthesis protocol scalable for commercial production?
A: Yes, the process is designed for scalability. It uses commercially available raw materials and standard organic solvents like 1,2-dichloroethane. The operational simplicity and safety profile support transition from laboratory scale to industrial manufacturing without complex engineering modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Imine Derivatives Supplier
The potential of indanone imine derivatives in oncology treatment is immense, yet realizing this potential requires a manufacturing partner with the technical acumen to execute complex synthetic routes with precision. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand that in the race to bring life-saving drugs to market, the reliability of your supply chain is as critical as the efficacy of the molecule itself. Our team of expert chemists is ready to collaborate with you to optimize this gold-catalyzed process for your specific needs, ensuring a seamless transition from research to commercial supply.
We invite you to engage with our technical procurement team to discuss how we can support your project goals. Whether you require a Customized Cost-Saving Analysis for your specific volume requirements or need to review specific COA data and route feasibility assessments, we are equipped to provide the detailed information necessary for your decision-making process. By partnering with us, you gain access to a robust supply network and a dedication to innovation that drives the pharmaceutical industry forward. Let us help you secure a stable, high-quality source of these critical anti-tumor intermediates for your next breakthrough therapy.
