Advanced Catalytic Synthesis of 3-Phenyl-1,2,4-Benzothiadiazine Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as the backbone for potent therapeutic agents. A significant advancement in this domain is detailed in patent CN108440448B, which discloses a novel synthesis method for 3-phenyl-1,2,4-benzothiadiazine-1,1(2H)-dioxide compounds. These structures are pivotal in medicinal chemistry, particularly known for their diuretic and antihypertensive properties, often functioning as thiazide derivatives. The innovation lies in the strategic utilization of styrene compounds as starting materials, which undergo a palladium-catalyzed transformation to construct the benzothiadiazine core. This approach represents a paradigm shift from traditional methods that often rely on harsher conditions or less accessible precursors. By leveraging the cleavage of carbon-carbon bonds in styrenes, this methodology offers a streamlined pathway that aligns with modern green chemistry principles while maintaining high structural fidelity. For R&D directors and procurement specialists, understanding the nuances of this patent provides a competitive edge in sourcing high-purity pharmaceutical intermediates efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiadiazine derivatives has been fraught with challenges that impede efficient commercial production. Traditional routes often involve multi-step sequences requiring stringent reaction conditions, such as high temperatures and elevated pressures, which necessitate specialized reactor equipment and increase operational risks. Furthermore, conventional methods frequently employ hydrogenation steps using platinum oxide catalysts in acetic acid, which not only introduces safety concerns regarding hydrogen handling but also complicates the removal of heavy metal residues from the final product. The reliance on expensive or difficult-to-source precursors further exacerbates cost structures, making the final API intermediates less economically viable for large-scale manufacturing. Additionally, older methodologies often suffer from poor atom economy and generate significant waste streams, posing environmental compliance hurdles for modern chemical plants striving for sustainability. These cumulative factors create bottlenecks in the supply chain, leading to longer lead times and reduced flexibility in responding to market demands for antihypertensive medications.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a highly efficient oxidative coupling strategy that circumvents many of these historical obstacles. By employing styrene compounds, which are abundant and cost-effective industrial chemicals, the process significantly lowers the raw material entry barrier. The reaction proceeds under an oxygen atmosphere using a palladium catalyst system, eliminating the need for hazardous hydrogen gas and high-pressure vessels. This transition to aerobic oxidation not only enhances operational safety but also simplifies the engineering controls required for production. The mild reaction conditions, typically ranging between 80°C and 110°C, allow for the use of standard glass-lined or stainless-steel reactors commonly found in fine chemical facilities. Moreover, the direct formation of the benzothiadiazine ring through intermolecular condensation reduces the number of isolation steps, thereby minimizing product loss and solvent consumption. This streamlined workflow translates directly into improved throughput and a more resilient supply chain for critical pharmaceutical intermediates.
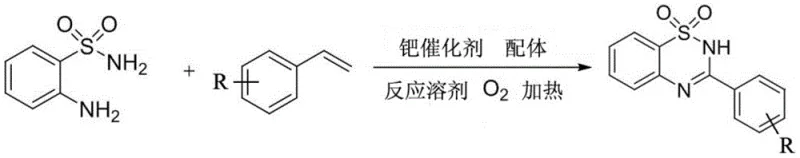
Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The core of this synthetic breakthrough relies on a sophisticated palladium-catalyzed mechanism that facilitates the cleavage of the carbon-carbon double bond in styrene derivatives. Initially, the palladium catalyst, specifically palladium trifluoroacetate, coordinates with the styrene substrate, activating the vinyl group towards oxidative functionalization. In the presence of molecular oxygen, the system promotes the cleavage of the C=C bond to generate an intermediate benzaldehyde species in situ. This transient aldehyde then undergoes a condensation reaction with 2-aminobenzenesulfonamide, driven by the nucleophilic attack of the amine group on the carbonyl carbon. The subsequent cyclization and dehydration steps forge the stable 1,2,4-benzothiadiazine dioxide ring system. The choice of ligand, such as 2,2'-bipyridine, is crucial as it stabilizes the palladium center and modulates its electronic properties to favor the desired oxidative pathway over competing side reactions. This precise control over the catalytic cycle ensures high selectivity, minimizing the formation of regioisomers or over-oxidized byproducts that could compromise the purity profile required for pharmaceutical applications.
Impurity control is inherently built into this mechanistic design, addressing a primary concern for R&D directors overseeing quality assurance. The specificity of the palladium-ligand complex reduces the likelihood of non-selective radical reactions that often plague free-radical oxidative processes. Furthermore, the use of dimethyl sulfoxide (DMSO) as the solvent plays a dual role; it acts as a polar medium that solubilizes the sulfonamide reactant effectively while also participating in the stabilization of polar transition states. Experimental data from the patent indicates that deviating from optimal solvent and catalyst combinations leads to negligible yields, underscoring the robustness of the optimized system against variance. For manufacturing teams, this means that maintaining strict adherence to the specified reagent grades and molar ratios is sufficient to guarantee consistent batch-to-bquality. The resulting product exhibits a clean impurity profile, facilitating easier downstream purification and ensuring that the final intermediate meets the stringent specifications demanded by global regulatory bodies for drug substance manufacturing.
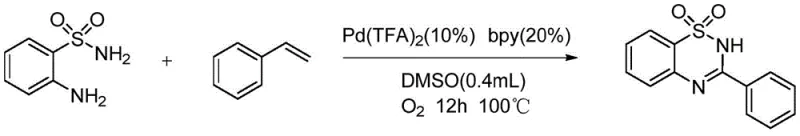
How to Synthesize 3-Phenyl-1,2,4-Benzothiadiazine Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of an aerobic environment to drive the oxidative cycle. The process begins with the charging of the solid reagents, including the sulfonamide, catalyst, and ligand, into a dry reaction vessel to prevent moisture interference. Following this, the system must be rigorously degassed and backfilled with oxygen to ensure that the oxidant concentration is sufficient throughout the reaction duration. The addition of the solvent and styrene substrate initiates the transformation, which is then sustained at elevated temperatures to overcome the activation energy barrier for C-C bond cleavage. While the general procedure is straightforward, the nuances of workup and purification are vital for isolating the product in high purity. Detailed standardized operating procedures regarding stoichiometry, temperature ramping, and chromatographic separation are essential for technology transfer.
- Charge 2-aminobenzenesulfonamide, palladium trifluoroacetate catalyst, and bipyridine ligand into the reaction vessel.
- Perform three cycles of vacuum evacuation and oxygen filling to ensure an aerobic environment.
- Add DMSO solvent and styrene derivative, then heat to 100°C for 12 hours with stirring.
- Remove solvent via rotary evaporation and purify the crude residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers substantial benefits that resonate deeply with procurement managers and supply chain heads focused on cost optimization and reliability. The primary driver of cost reduction lies in the raw material selection; styrene derivatives are commodity chemicals produced on a massive global scale, ensuring price stability and widespread availability compared to specialized heterocyclic precursors. This abundance mitigates the risk of supply disruptions caused by single-source dependencies, a critical factor for maintaining continuous manufacturing operations. Furthermore, the elimination of high-pressure hydrogenation steps reduces capital expenditure on specialized equipment and lowers the ongoing maintenance costs associated with high-risk reactor systems. The mild reaction conditions also contribute to energy efficiency, as heating to 100°C is significantly less energy-intensive than maintaining high-pressure environments or cryogenic conditions required by alternative routes. These operational efficiencies accumulate to provide a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global marketplace for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the avoidance of expensive noble metal catalysts like platinum oxide, which are traditionally used in similar transformations. Instead, the palladium system employed here operates at relatively low loading levels, and the potential for catalyst recovery and recycling further diminishes the effective cost per kilogram of product. Additionally, the simplified workup procedure, which involves standard solvent removal and chromatography, reduces the consumption of auxiliary materials and labor hours associated with complex extraction or crystallization protocols. By streamlining the synthesis into fewer operational steps, manufacturers can achieve higher overall throughput without proportional increases in overhead, resulting in substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents such as 2-aminobenzenesulfonamide and various substituted styrenes. These materials are not subject to the same geopolitical or logistical constraints as exotic reagents, ensuring a steady flow of inputs for production scheduling. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility supplies, such as cooling water temperature or steam pressure, which can often cause batch failures in more sensitive chemistries. This reliability translates to predictable lead times and the ability to commit to larger volume contracts with confidence. For supply chain planners, this predictability is invaluable for inventory management and meeting the just-in-time delivery requirements of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous reagents and extreme conditions. The use of molecular oxygen as the terminal oxidant generates water as the primary byproduct, aligning with green chemistry metrics and reducing the burden on waste treatment facilities. This environmental compatibility simplifies the permitting process for new production lines and ensures compliance with increasingly stringent environmental regulations globally. The scalability is further supported by the liquid-phase nature of the reaction, which allows for efficient heat and mass transfer in large-scale stirred tank reactors. Consequently, manufacturers can ramp up production volumes to meet surging market demand for antihypertensive intermediates without encountering the technical barriers often associated with scaling heterogeneous or high-pressure reactions.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled answers based on the specific data and claims within the patent documentation. These responses clarify the operational parameters and the strategic advantages of adopting this route for pharmaceutical intermediate production. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: What are the primary advantages of this styrene-based synthesis route?
A: This method utilizes cheap and readily available styrene compounds as raw materials, avoiding expensive precursors. It operates under mild conditions without high pressure, significantly simplifying equipment requirements and post-treatment processes compared to traditional hydrogenation methods.
Q: How does the catalyst system impact reaction yield?
A: The selection of Palladium Trifluoroacetate (Pd(TFA)2) combined with 2,2'-bipyridine is critical. Experimental data indicates that substituting this specific catalyst-ligand combination with other palladium salts or ligands results in drastically reduced yields or complete reaction failure, highlighting the specificity of this catalytic cycle.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process avoids high-temperature and high-pressure conditions, using molecular oxygen as the oxidant. This enhances safety profiles for scale-up. Furthermore, the use of common solvents like DMSO and standard purification techniques like column chromatography ensures compatibility with existing GMP production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Phenyl-1,2,4-Benzothiadiazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of life-saving medications. Our technical team has thoroughly analyzed the potential of the palladium-catalyzed styrene cleavage method and is well-equipped to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and compliant. Our facilities are designed to handle sensitive catalytic reactions with precision, featuring rigorous QC labs that enforce stringent purity specifications for every batch released. We understand that consistency is key in the pharmaceutical supply chain, and our quality management systems are aligned with international standards to guarantee that our intermediates meet the exacting requirements of global drug manufacturers.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner committed to innovation, reliability, and the highest standards of chemical manufacturing excellence, ensuring your projects proceed without interruption.
