Advanced Atmospheric Pressure Synthesis of Lubiprostone for Scalable Pharmaceutical Production
The pharmaceutical landscape for treating chronic idiopathic constipation and irritable bowel syndrome has been significantly shaped by the development of selective chloride channel activators, most notably Lubiprostone. As detailed in patent CN104557845B, recent advancements in synthetic methodology have addressed critical bottlenecks in the manufacturing of this complex prostaglandin derivative. The molecule exists in a dynamic equilibrium between hemiketal and ketone forms, presenting unique challenges in purification and stability that demand precise control over reaction conditions. This technical report analyzes a breakthrough preparation method that utilizes transfer catalytic hydrogenation, offering a robust alternative to traditional high-pressure hydrogenation techniques. By leveraging triethylsilane as a hydrogen source in conjunction with palladium catalysts, this process achieves exceptional purity and yield while operating under atmospheric pressure, marking a significant evolution in the production of high-purity pharmaceutical intermediates.
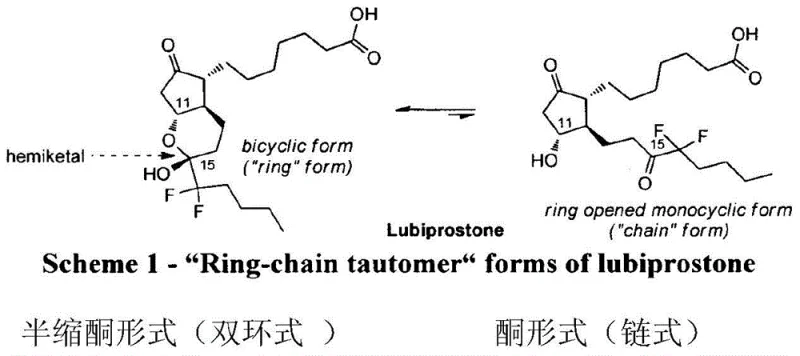
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lubiprostone has relied heavily on catalytic hydrogenation using molecular hydrogen gas under elevated pressures, as documented in prior art such as WO9927934 and US2013184476A. These conventional routes typically require specialized high-pressure autoclaves to facilitate the reduction of olefinic bonds and the simultaneous removal of benzyl protecting groups. The reliance on pressurized hydrogen introduces substantial operational hazards and necessitates rigorous safety protocols, which inherently increases the capital expenditure for manufacturing facilities. Furthermore, comparative data indicates that these high-pressure methods often suffer from prolonged reaction times, frequently exceeding eight hours, which can lead to the formation of dehydration byproducts and other impurities. The resulting crude product often exhibits lower purity profiles, necessitating extensive downstream purification steps that erode overall process efficiency and increase the cost of goods sold for the final API.
The Novel Approach
In stark contrast, the methodology disclosed in CN104557845B introduces a sophisticated transfer catalytic hydrogenation strategy that operates effectively at atmospheric pressure. This novel route employs triethylsilane as a potent hydride donor, replacing the need for gaseous hydrogen and high-pressure containment systems. The reaction proceeds rapidly, typically completing within a window of 10 to 40 minutes, which drastically reduces the thermal stress on the sensitive prostaglandin scaffold. By utilizing alcohol solvents such as methanol or ethanol, the process ensures high solubility of reactants while maintaining a mild environment that preserves stereochemical integrity. This approach not only simplifies the operational workflow but also delivers product purity levels consistently above 97%, demonstrating a clear superiority over legacy methods in terms of both speed and quality control for reliable API intermediate supplier operations.
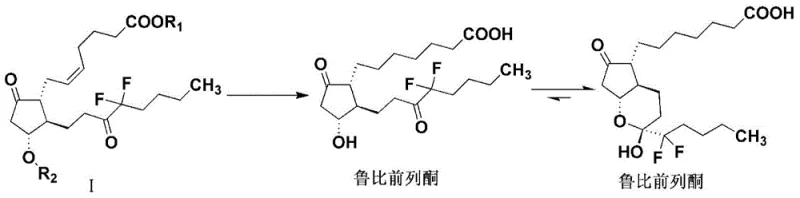
Mechanistic Insights into Triethylsilane-Mediated Transfer Hydrogenation
The core innovation of this synthesis lies in the mechanistic pathway facilitated by the triethylsilane-palladium system. Unlike traditional hydrogenation where molecular hydrogen adsorbs onto the catalyst surface, this transfer hydrogenation involves a concerted mechanism where the silicon-hydrogen bond serves as the source of hydride ions. The reaction initiates with the protonation of the substrate, likely facilitated by trace water or the alcoholic solvent, generating a reactive intermediate. Subsequently, the triethylsilane delivers hydride equivalents to reduce the carbon-carbon double bonds and cleave the benzyl ether linkages simultaneously. This dual functionality is critical for the efficient deprotection and saturation of the side chains without affecting the sensitive cyclopentane ring or the fluorine substituents. The use of palladium on carbon or palladium hydroxide provides the necessary catalytic surface to lower the activation energy, allowing the reaction to proceed swiftly at temperatures ranging from 15°C to 30°C, thereby minimizing thermal degradation pathways.
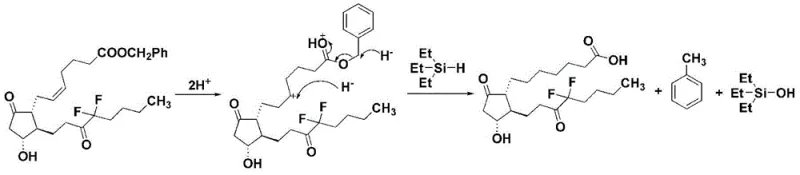
Furthermore, the choice of solvent plays a pivotal role in dictating the reaction trajectory and impurity profile. Experimental data within the patent highlights that alcoholic solvents are superior to ethers like tetrahydrofuran (THF). In THF, the reaction environment may promote radical pathways that lead to unwanted side reactions, resulting in significantly lower yields and purity. In contrast, protic alcoholic solvents stabilize the transition states and facilitate the smooth transfer of protons and hydrides. This mechanistic understanding allows process chemists to fine-tune the reaction parameters, ensuring that the final product meets stringent purity specifications required for clinical applications. The ability to control the tautomeric equilibrium between the ring and chain forms during workup further underscores the precision of this method, ensuring a consistent and high-quality output suitable for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Lubiprostone Efficiently
The implementation of this synthesis route requires careful attention to reagent addition and temperature control to maximize the benefits of the transfer hydrogenation mechanism. The process begins by dissolving the protected precursor in a suitable alcohol, followed by the introduction of the palladium catalyst under an inert atmosphere to prevent oxidation. The triethylsilane is then added, either directly or as a diluted solution, to initiate the reduction. Detailed standard operating procedures regarding stoichiometry, specifically the molar ratio of substrate to silane (optimized at 1:4), are critical for reproducibility. For a comprehensive guide on the specific execution of this protocol, including workup and isolation steps, please refer to the standardized synthesis instructions below.
- Dissolve the protected precursor compound (Compound I) in an alcoholic solvent such as methanol or ethanol under an inert gas atmosphere.
- Add the palladium catalyst (Pd/C or Pd(OH)2) and introduce triethylsilane as the hydrogen source, maintaining the temperature between 15°C and 30°C.
- Stir the reaction mixture for 10 to 40 minutes to complete the reduction and deprotection, then filter and concentrate to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this atmospheric pressure transfer hydrogenation method offers transformative benefits for supply chain resilience and cost management. The elimination of high-pressure hydrogenation equipment removes a significant barrier to entry for manufacturing partners, allowing for production in standard glass-lined or stainless steel reactors that are more readily available and easier to maintain. This shift drastically simplifies the facility requirements, reducing the capital investment needed for plant upgrades and lowering the ongoing maintenance costs associated with high-pressure systems. Additionally, the use of triethylsilane, a liquid reagent, simplifies logistics and storage compared to managing bulk hydrogen gas supplies, thereby enhancing the overall safety profile of the manufacturing site and reducing insurance and compliance overheads.
- Cost Reduction in Manufacturing: The transition to atmospheric conditions fundamentally alters the cost structure of production by removing the need for expensive autoclaves and specialized high-pressure piping. This reduction in capital expenditure is complemented by the operational efficiency gained from shorter reaction cycles, which increases reactor throughput and reduces utility consumption per kilogram of product. The high yield and purity achieved directly translate to reduced waste generation and lower costs associated with purification and recycling, providing a compelling economic argument for adopting this technology in cost reduction in pharmaceutical manufacturing initiatives.
- Enhanced Supply Chain Reliability: By simplifying the reaction conditions and utilizing commercially available reagents like triethylsilane and palladium catalysts, the supply chain becomes more robust against disruptions. The method does not rely on specialized infrastructure that might be a bottleneck in certain regions, allowing for greater flexibility in sourcing and manufacturing locations. This decentralization potential ensures a more continuous supply of high-purity pharmaceutical intermediates, mitigating the risks of production delays that can impact downstream drug formulation and market availability.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of high-pressure hazards make this process inherently safer and easier to scale from pilot batches to multi-ton commercial production. The simplified waste stream, characterized primarily by benign silanol byproducts and alcohol solvents, facilitates easier treatment and disposal, aligning with increasingly stringent environmental regulations. This ease of scale-up ensures that manufacturers can respond rapidly to market demand fluctuations without compromising on safety or environmental standards, securing a sustainable long-term supply.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. Understanding these nuances is essential for process engineers and quality assurance teams looking to integrate this technology into their existing workflows. The answers provided are derived directly from the experimental data and technical disclosures found in the patent literature, ensuring accuracy and relevance for industrial application.
Q: Why is transfer hydrogenation preferred over traditional high-pressure hydrogenation for Lubiprostone?
A: Transfer hydrogenation eliminates the need for high-pressure autoclaves, significantly reducing equipment costs and safety risks while achieving higher purity and shorter reaction times compared to conventional methods.
Q: What is the role of triethylsilane in this synthesis?
A: Triethylsilane acts as a highly active hydride donor that facilitates the simultaneous reduction of double bonds and removal of benzyl protecting groups under mild, atmospheric conditions.
Q: How does solvent choice impact the reaction outcome?
A: Alcoholic solvents like methanol and ethanol are critical for stabilizing the reaction intermediates; using solvents like THF can lead to radical side reactions that drastically lower yield and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lubiprostone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement complex chemistries, such as the transfer hydrogenation described herein, positions us as a strategic partner capable of navigating the intricacies of modern API manufacturing.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this atmospheric pressure method. We encourage you to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality, efficiency, and partnership in the development of high-value pharmaceutical intermediates.
