Advanced Semi-Synthetic Route for Chenodeoxycholic Acid: Scalable Manufacturing and Cost Efficiency
Advanced Semi-Synthetic Route for Chenodeoxycholic Acid: Scalable Manufacturing and Cost Efficiency
The pharmaceutical industry is constantly seeking robust, scalable, and ethically sound methods for producing critical bile acid intermediates. Patent CN112724189A introduces a groundbreaking preparation method for Chenodeoxycholic Acid (CDCA), a pivotal precursor for Ursodeoxycholic Acid (UDCA) and various other therapeutic agents. This innovation shifts the paradigm from reliance on limited animal extraction to a sophisticated semi-synthetic approach that utilizes waste components from duck bile paste processing. By transforming what was previously considered industrial waste into a high-value active pharmaceutical ingredient, this technology addresses both economic and environmental challenges simultaneously. For R&D directors and procurement specialists, this represents a significant opportunity to secure a more stable and cost-effective supply chain for bile acid derivatives, ensuring continuity in the production of hepatoprotective drugs without compromising on purity or yield standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Chenodeoxycholic Acid and its derivative Ursodeoxycholic Acid has been heavily dependent on the direct extraction from animal bile, particularly from bears and poultry. This traditional reliance presents severe bottlenecks for modern pharmaceutical manufacturing. Firstly, the yield from direct extraction is inherently limited by the biological availability of the source material, creating a supply ceiling that cannot easily be expanded to meet growing global demand. Secondly, the ethical implications of extracting bile from live animals have led to stringent regulations and public scrutiny, forcing manufacturers to seek alternative, cruelty-free production methods. Furthermore, the purification of natural extracts often involves complex chromatography steps to remove impurities and other bile acid congeners, resulting in lower overall recovery rates and higher processing costs. These factors combine to create a volatile market where price fluctuations are common, and supply continuity is frequently at risk due to seasonal or regulatory constraints on animal sourcing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112724189A leverages a waste valorization strategy that fundamentally alters the economic landscape of CDCA production. By isolating specific bile acid components, referred to as PhCA (Phocaean cholic acid or similar waste fractions), from the residue of duck bile paste extraction, the process turns a disposal problem into a revenue stream. This semi-synthetic route employs a series of well-defined organic transformations including propylidene protection, acetylation, and stereoselective inversion to construct the target molecule. The beauty of this approach lies in its modularity; each step is optimized for high conversion and ease of workup, utilizing common reagents like acetone, acetic anhydride, and lithium bromide. This not only simplifies the operational complexity but also ensures that the process can be seamlessly integrated into existing fine chemical manufacturing facilities without requiring exotic catalysts or extreme reaction conditions, thereby facilitating rapid industrial adoption.
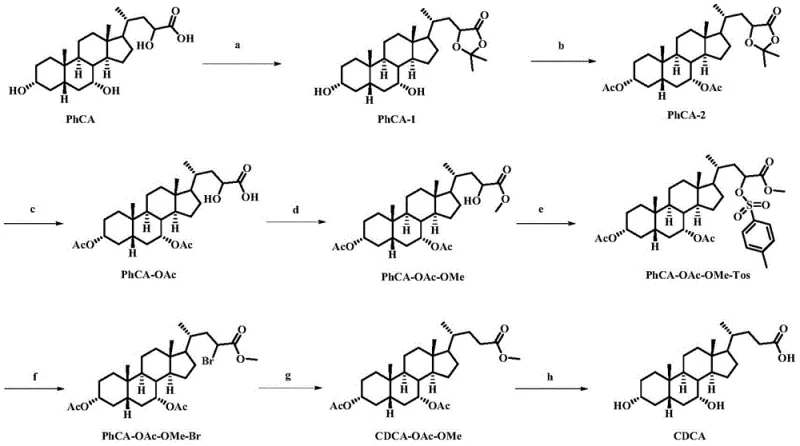
Mechanistic Insights into Stereoselective Inversion and Protection Strategies
The core chemical ingenuity of this patent lies in the precise manipulation of the steroid backbone to achieve the correct stereochemistry at the C-7 position. The synthesis begins with a propylidene protection step, where acetone and concentrated sulfuric acid are used to form a cyclic ketal protecting group. This is a critical strategic move that masks the reactivity of the 3-alpha and 7-alpha hydroxyl groups, allowing for selective modification elsewhere on the molecule without unwanted side reactions. Following this, acetylation is performed to introduce acetate groups, which serve as temporary protecting groups that are easier to manipulate than free hydroxyls. The subsequent removal of the propylidene group under acidic conditions reveals the specific hydroxyl functionality required for the next stage of transformation. This careful orchestration of protection and deprotection ensures that the reactive sites are exposed only when necessary, minimizing the formation of regioisomers and simplifying the purification profile of the intermediates.
The most chemically significant phase of the synthesis is the inversion of configuration at the C-7 position to generate the desired CDCA structure from the starting material. This is achieved through a classic activation-substitution-reduction sequence. The hydroxyl group is first activated by reaction with p-toluenesulfonyl chloride to form a tosylate, a superb leaving group. This is followed by a nucleophilic substitution with lithium bromide, which displaces the tosylate and introduces a bromine atom with inverted stereochemistry. Finally, a debromination step using sodium borohydride reduces the carbon-bromine bond, effectively locking in the new stereochemical configuration while removing the halogen. This multi-step inversion strategy is far superior to direct oxidation-reduction methods which often suffer from poor selectivity. The final deprotection step utilizes alkaline hydrolysis to remove the acetate and methyl ester groups, yielding the free acid form of Chenodeoxycholic Acid with high purity and the correct physiological configuration required for downstream pharmaceutical applications.
How to Synthesize Chenodeoxycholic Acid Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters outlined in the patent to maximize yield and minimize impurity formation. The process is designed to be operationally simple, avoiding the need for cryogenic temperatures or high-pressure equipment in most steps. Key to success is the control of reaction times during the protection and acetylation phases, as well as the precise stoichiometric addition of reagents during the inversion sequence. The following guide summarizes the critical operational phases derived from the patent examples, providing a roadmap for process chemists to replicate this high-yielding pathway in a pilot or production setting.
- Perform propylidene protection on the starting material (PhCA) using acetone and acid catalyst to protect diol functionalities.
- Execute acetylation and subsequent depropylene removal to isolate the mono-acetylated intermediate.
- Conduct methyl esterification followed by tosylation and bromination to invert the stereochemistry at the C-7 position.
- Finalize the synthesis through debromination reduction and hydrolytic deprotection to yield pure Chenodeoxycholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this semi-synthetic technology offers profound strategic advantages that extend beyond simple unit cost metrics. The primary value proposition is the decoupling of production capacity from the limitations of animal farming cycles. By utilizing waste streams that are already generated in large volumes by the poultry industry, manufacturers can secure a raw material supply that is both abundant and consistent. This stability is crucial for long-term contract planning and mitigates the risk of price spikes associated with seasonal shortages of natural bile. Furthermore, the chemical steps involved are robust and utilize commodity chemicals, reducing dependency on specialized or single-source reagents that could become supply chain bottlenecks.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the low cost of the starting material, which is essentially a byproduct of another industry. By valorizing waste, the input cost basis is significantly lower than purchasing purified natural bile acids. Additionally, the synthetic route avoids the use of expensive transition metal catalysts or enzymes, relying instead on inexpensive reagents like sulfuric acid and lithium bromide. The high overall yield reported in the patent means that less raw material is wasted per kilogram of finished product, further driving down the cost of goods sold. This efficiency allows for competitive pricing strategies in the global API market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved because the feedstock is derived from the massive duck farming industry, which operates on a much larger and more consistent scale than bear farming or specialized bile collection. This abundance ensures that raw material availability is not a limiting factor for scaling up production to meet surging demand for liver disease medications. Moreover, the synthetic nature of the process allows for production to be located closer to key pharmaceutical markets, reducing logistics lead times and freight costs. The ability to produce CDCA independently of biological extraction cycles provides a buffer against biological variability and regulatory changes affecting animal-derived products.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with green chemistry principles by reducing waste and maximizing atom economy. The solvents used, such as ethyl acetate and methanol, are readily recoverable and recyclable, minimizing the environmental footprint of the manufacturing process. The absence of heavy metals or toxic biological residues simplifies the waste treatment protocols and reduces the cost of environmental compliance. This makes the facility more sustainable and less prone to shutdowns due to environmental violations. The simplicity of the reaction conditions also means that the process can be scaled from pilot batches to multi-ton production with minimal re-engineering, ensuring a smooth transition from R&D to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CDCA synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in patent CN112724189A, providing clarity on the feasibility and advantages of this route for potential partners and licensees.
Q: What is the primary advantage of this CDCA synthesis method over traditional extraction?
A: The primary advantage is the utilization of waste streams from duck bile paste extraction as raw materials. This approach transforms low-value byproducts into high-value APIs, significantly reducing raw material costs and alleviating pressure on animal resources compared to direct extraction methods.
Q: How does the process ensure high stereochemical purity at the C-7 position?
A: The process employs a strategic functional group inversion sequence involving tosylation followed by bromine substitution and subsequent reduction. This specific chemical transformation allows for precise control over the stereochemistry at the C-7 position, ensuring the formation of the desired beta-hydroxyl configuration essential for biological activity.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the simplicity of the reaction conditions, such as ambient temperature operations for several steps and the use of common solvents like ethyl acetate and methanol. These factors, combined with high overall yields exceeding 50%, indicate strong feasibility for commercial scale-up and industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chenodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN112724189A for the global bile acid market. As a leading CDMO and fine chemical manufacturer, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to fruition. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions outlined in the patent, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to monitor the stereochemical integrity of the C-7 position and the removal of all protecting groups, guaranteeing a product that exceeds pharmacopeial standards for API intermediates.
We invite pharmaceutical companies and bulk drug manufacturers to collaborate with us to leverage this cost-effective and sustainable production method. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in steroid chemistry can optimize your supply chain and reduce your overall manufacturing costs for Chenodeoxycholic Acid and its derivatives.
