Scalable Electrochemical Synthesis of N-Alkyl Amides for High-Purity Pharmaceutical Intermediates
Scalable Electrochemical Synthesis of N-Alkyl Amides for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable and efficient methodologies, driven by the urgent need for greener manufacturing processes in the fine chemical industry. A pivotal development in this arena is detailed in patent CN115584518A, which discloses a novel electrochemical method for the preparation of N-alkyl amides. This technology leverages electric current as a traceless oxidant to drive the condensation of fatty amines and alpha-keto acids, bypassing the traditional reliance on stoichiometric chemical oxidants or transition metal catalysts. For R&D directors and procurement strategists, this represents a significant opportunity to streamline supply chains and reduce the environmental footprint of API intermediate production. By operating under mild conditions—specifically at room temperature and in an open air atmosphere—this electrochemical protocol offers a robust alternative to conventional amidation strategies that often suffer from harsh conditions and complex waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the amide bond, a ubiquitous motif in pharmaceuticals and agrochemicals, has relied heavily on dehydration condensation reactions mediated by coupling reagents such as carbodiimides (e.g., EDC, DCC) or phosphonium salts (e.g., HATU, PyBOP). While effective on a small laboratory scale, these methods present substantial challenges when translated to commercial manufacturing. The primary drawback is the generation of stoichiometric amounts of urea or phosphine oxide byproducts, which are notoriously difficult to remove and contribute significantly to the process mass intensity (PMI). Furthermore, many traditional protocols require the use of transition metal catalysts or strong activating agents that are moisture-sensitive, necessitating expensive inert atmosphere equipment and rigorous drying of solvents. These factors collectively inflate the cost of goods sold (COGS) and complicate the regulatory approval process due to concerns over residual metal impurities and genotoxic potential in the final active pharmaceutical ingredient.
The Novel Approach
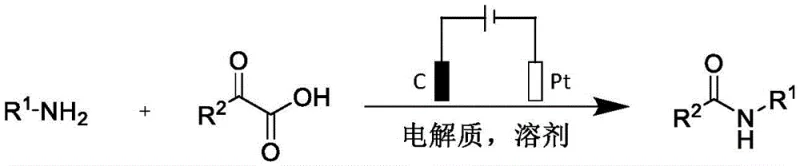
In stark contrast, the electrochemical methodology outlined in the patent utilizes electrons as the primary reagent, fundamentally altering the economic and operational dynamics of amide synthesis. The reaction proceeds in a simple diaphragm-free electrolytic cell equipped with a inexpensive graphite rod anode and a platinum sheet cathode. By applying a constant current of 8 mA in a mixed solvent system of dichloromethane and tetrahydrofuran (4:1), the system achieves efficient conversion without the need for external chemical oxidants. This approach not only eliminates the cost associated with purchasing and disposing of coupling reagents but also simplifies the work-up procedure to a straightforward solvent evaporation followed by column chromatography. The ability to run the reaction under ambient air conditions further reduces capital expenditure on specialized reactor infrastructure, making this a highly attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Electrochemical Oxidative Amidation
The core of this innovation lies in the anodic oxidation mechanism that activates the carboxylic acid derivative for nucleophilic attack by the amine. In the absence of traditional activating agents, the application of an electric potential at the graphite anode facilitates the generation of reactive intermediates from the alpha-keto acid substrate. It is hypothesized that the electrochemical environment promotes the decarboxylation or direct activation of the carbonyl group, rendering it sufficiently electrophilic to react with the nucleophilic amine. The use of tetrabutylammonium fluoride as the supporting electrolyte plays a critical role in maintaining conductivity and potentially stabilizing charged intermediates within the double layer at the electrode surface. This mechanistic pathway avoids the formation of stable metal-ligand complexes that often plague transition-metal catalyzed reactions, thereby ensuring that the final product is free from heavy metal contamination—a critical quality attribute for any reliable pharma intermediate supplier.
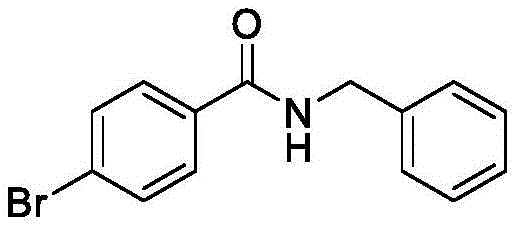
Furthermore, the broad substrate scope demonstrated in the patent examples underscores the versatility of this electrochemical system. The reaction tolerates a wide array of functional groups, including halogens and heterocycles, which are essential structural features in modern drug discovery. For instance, the successful synthesis of N-benzyl-4-bromobenzamide illustrates that the oxidative conditions are mild enough to preserve sensitive carbon-halogen bonds that might otherwise undergo unwanted side reactions under harsher chemical oxidation regimes. This high level of chemoselectivity ensures that the impurity profile of the crude product remains manageable, reducing the burden on downstream purification units. The consistent yields observed across different substrates, ranging from simple alkyl amines to complex heteroaromatic amines, indicate a robust and predictable reaction manifold that is ideal for process optimization and scale-up.
How to Synthesize N-Alkyl Amides Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to maximize yield and reproducibility. The patent specifies a molar ratio of 1:1.2 between the fatty amine and the alpha-keto acid, ensuring that the more valuable or difficult-to-remove component is fully consumed. The choice of solvent is also critical; the 4:1 mixture of dichloromethane and tetrahydrofuran provides the optimal balance of solubility for both organic substrates and the ionic electrolyte. Operators must maintain a constant current of 8 mA for a duration of 2 hours, a parameter that directly correlates with the total charge passed and thus the extent of conversion. Deviating from these optimized conditions could lead to incomplete reaction or over-oxidation of the substrate. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the reaction mixture by combining fatty amine and alpha-keto acid in a 1: 1.2 molar ratio with tetrabutylammonium fluoride electrolyte in a DCM/THF solvent system.
- Insert a graphite rod anode and a platinum sheet cathode into the diaphragm-free cell and apply a constant current of 8 mA under air atmosphere at room temperature.
- After 2 hours of electrolysis, remove the solvent under reduced pressure and purify the crude residue via column chromatography to isolate the target N-alkyl amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic advantages beyond mere technical novelty. The elimination of expensive coupling reagents and transition metal catalysts directly impacts the raw material cost structure, offering a pathway to significant cost savings in high-volume production. Moreover, the simplified reaction setup, which does not require inert gas protection or cryogenic cooling, reduces the energy consumption and operational complexity of the manufacturing facility. This simplicity enhances supply chain reliability by minimizing the risk of batch failures due to equipment malfunction or operator error, ensuring a steady flow of critical intermediates to downstream synthesis units. The use of commercially available and inexpensive starting materials further secures the supply chain against volatility in the pricing of specialized reagents.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the replacement of stoichiometric chemical oxidants and coupling agents with electricity, which is a vastly cheaper reagent. Traditional amidation often generates large volumes of solid waste that requires costly disposal; by contrast, this electrochemical method produces minimal waste, primarily consisting of the electrolyte and solvent which can potentially be recycled. Additionally, the absence of transition metals removes the need for expensive scavenging resins or complex purification steps designed to meet strict residual metal limits, thereby lowering the overall processing cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply security. Because the process operates under air and at room temperature, it is less susceptible to disruptions caused by utility failures or environmental fluctuations compared to sensitive catalytic processes. The starting materials, fatty amines and alpha-keto acids, are commodity chemicals with established global supply networks, reducing the risk of raw material shortages. This stability allows for more accurate forecasting and inventory management, ensuring that production schedules for key pharmaceutical intermediates can be maintained without unexpected delays.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this method offers a clear advantage. The "green" nature of using electrons as reagents aligns perfectly with increasingly stringent environmental regulations regarding solvent use and waste generation. Scaling this process is straightforward, as electrochemical reactors can be expanded by increasing electrode surface area or numbering up cells, avoiding the heat transfer limitations often encountered in large-scale exothermic chemical reactions. This scalability ensures that the technology can support commercial production volumes ranging from pilot plant batches to multi-ton annual campaigns without compromising on safety or environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical amidation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows. The responses cover aspects of reaction sensitivity, substrate compatibility, and purification requirements to give a holistic view of the process capabilities.
Q: What are the advantages of using electrochemical methods over traditional coupling reagents for amide synthesis?
A: Electrochemical methods eliminate the need for expensive and wasteful chemical coupling agents like EDC or HATU, as well as transition metal catalysts. This results in a cleaner reaction profile, simplified downstream purification, and significantly reduced chemical waste, aligning with green chemistry principles.
Q: Is this electrochemical amidation process sensitive to oxygen or moisture?
A: No, the process described in patent CN115584518A is remarkably robust. It operates effectively under an open air atmosphere and does not require stringent anhydrous or anaerobic conditions, which drastically simplifies the operational requirements for large-scale manufacturing.
Q: What types of substrates are compatible with this electrochemical protocol?
A: The method exhibits excellent functional group tolerance, successfully accommodating various substituted aromatic amines, heterocyclic amines, and alpha-keto acids bearing electron-withdrawing or electron-donating groups, making it highly versatile for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115584518A can be successfully translated to industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity N-alkyl amides that serve as critical building blocks for next-generation therapeutics, leveraging our technical expertise to optimize yield and minimize impurity levels.
We invite potential partners to engage with our technical team to explore how this cost-effective and sustainable synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this electrochemical protocol for your supply chain. We encourage you to contact our technical procurement team to obtain specific COA data for our catalog compounds and to discuss route feasibility assessments for your proprietary molecules. Let us collaborate to build a more efficient and sustainable future for chemical manufacturing.
