Advanced Oxidative Coupling Technology for High-Purity Phenanthrene and Biphenyl Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance high purity with environmental sustainability. Patent CN101747126A introduces a transformative approach to the preparation of phenanthrene, binaphthol, and biphenyl derivatives through oxidative coupling mediated by manganese dioxide (MnO2) or m-chloroperoxybenzoic acid (m-CPBA). This technology represents a significant departure from legacy methods that rely on hazardous heavy metal catalysts, offering a cleaner and more efficient pathway for generating complex aromatic scaffolds. For R&D directors and procurement managers alike, this innovation addresses critical pain points regarding impurity profiles and waste management. By leveraging mild reaction conditions ranging from -30°C to 80°C and utilizing readily available oxidants, this process ensures consistent quality while mitigating the supply chain risks associated with specialized toxic reagents. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such proprietary methodologies is essential for securing long-term production stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenanthrene rings and related biaryl structures has been fraught with significant technical and environmental challenges. Traditional bibliographic methods, such as the Pschorr cyclization or coupling reactions mediated by thallium trifluoroacetate and lead acetate, suffer from severe drawbacks that hinder large-scale manufacturing. The primary concern is the inherent toxicity of reagents like thallium and lead, which necessitates rigorous and costly waste disposal protocols to meet modern environmental regulations. Furthermore, these conventional pathways often involve multi-step sequences with harsh reaction conditions that can degrade sensitive functional groups, leading to lower overall yields and complex impurity spectra. The difficulty in separating catalyst residues from the final product often requires additional purification steps, such as column chromatography or extensive recrystallization, which drastically increases production time and operational costs. Consequently, reliance on these outdated technologies creates bottlenecks in the supply chain, making it difficult to achieve the cost reduction in pharmaceutical intermediates manufacturing that global markets demand.
The Novel Approach
In stark contrast, the novel oxidative coupling method described in the patent utilizes manganese dioxide or m-CPBA as the primary oxidants, offering a streamlined and safer alternative. This approach allows for the direct cyclization of (E)- or (Z)-1,2-di(substituted-phenyl) ethylene derivatives into phenanthrene structures with remarkable efficiency. The reaction proceeds smoothly in a variety of organic solvents, with trifluoroacetic acid identified as the optimal medium, facilitating rapid dissolution and reaction kinetics. By operating within a moderate temperature window of 0°C to 20°C, the process minimizes thermal stress on the substrates, thereby preserving stereochemical integrity and reducing the formation of thermal degradation byproducts. The simplicity of the workup procedure, involving basic aqueous washing and solvent evaporation, significantly shortens the production cycle. This technological leap not only enhances the purity of the final API intermediates but also aligns with green chemistry principles by eliminating the need for toxic heavy metals, thus providing a sustainable competitive advantage.
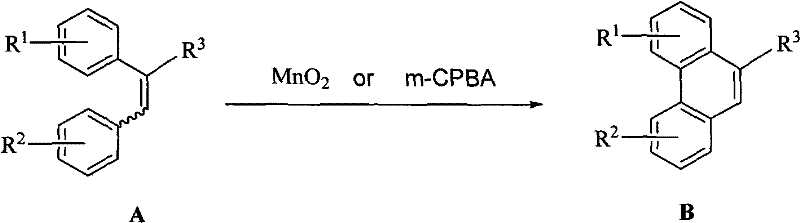
Mechanistic Insights into MnO2/m-CPBA Mediated Oxidative Coupling
The mechanistic underpinning of this transformation involves a sophisticated oxidative radical or electrophilic pathway depending on the specific oxidant employed. When manganese dioxide is utilized, it acts as a heterogeneous oxidant that likely facilitates the generation of radical intermediates on the olefinic bond of the stilbene derivative. These radicals undergo intramolecular cyclization to form the new carbon-carbon bond required for the phenanthrene core, followed by rearomatization to yield the stable polycyclic product. Alternatively, m-CPBA functions as a potent oxygen transfer agent that can epoxidize the double bond or generate cationic species which subsequently rearrange to form the coupled product. The versatility of this mechanism is evidenced by its applicability to a wide range of substrates, including those with electron-donating methoxy groups or electron-withdrawing ester functionalities. Understanding this mechanistic flexibility is crucial for R&D teams aiming to adapt the process for novel analogues, as it suggests a high tolerance for diverse substitution patterns without compromising reaction fidelity.
Impurity control is a paramount concern in the synthesis of high-purity OLED material or pharmaceutical precursors, and this oxidative coupling strategy excels in this regard. The high chemoselectivity of MnO2 and m-CPBA ensures that side reactions, such as over-oxidation of sensitive aldehyde or alcohol groups, are minimized. For instance, the patent data demonstrates successful conversion of substrates containing ester and nitrile groups without hydrolysis or degradation, indicating a gentle yet effective oxidation profile. The use of trifluoroacetic acid as a solvent further aids in suppressing side reactions by stabilizing cationic intermediates and preventing polymerization. This results in a crude product with a significantly cleaner profile, reducing the burden on downstream purification units. For supply chain heads, this translates to higher throughput and reduced solvent consumption, directly impacting the commercial scale-up of complex polymer additives and fine chemicals.
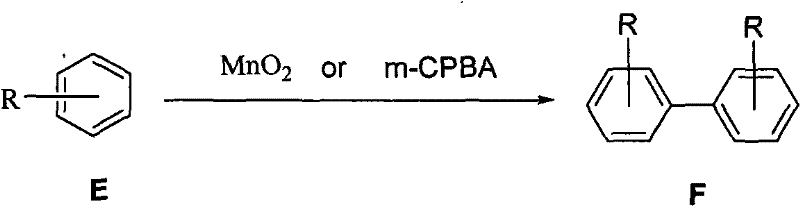
How to Synthesize Phenanthrene Derivatives Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it ideal for industrial adoption. The process begins with the dissolution of the stilbene precursor in a suitable organic solvent, followed by the controlled addition of the oxidant. Reaction monitoring via HPLC confirms complete conversion within 0.5 to 12 hours, depending on the specific substrate reactivity. The subsequent workup involves simple phase separation and drying, avoiding the need for complex extraction protocols. Detailed standard operating procedures for this synthesis are critical for maintaining batch-to-batch consistency.
- Dissolve the (E)- or (Z)-1,2-di(substituted-phenyl) ethylene derivative in an organic solvent such as trifluoroacetic acid.
- Add manganese dioxide (MnO2) or m-chloroperoxybenzoic acid (m-CPBA) with a molar ratio of 1: 0.8-2 relative to the substrate.
- Stir the mixture at temperatures between -30°C and 80°C until reaction completion, then perform aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this MnO2/m-CPBA mediated oxidative coupling technology offers substantial strategic benefits for procurement and supply chain management. The elimination of expensive and regulated heavy metal catalysts like thallium and vanadium removes a significant cost center associated with hazardous material handling and disposal. This shift not only lowers the direct material costs but also reduces the regulatory overhead required for storing and transporting toxic substances. Furthermore, the use of commodity chemicals such as manganese dioxide and m-CPBA ensures a stable supply base, mitigating the risk of raw material shortages that can plague specialty catalyst markets. The robustness of the reaction conditions allows for flexible manufacturing schedules, enhancing the overall agility of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of replacing toxic heavy metal catalysts with benign oxidants is profound. Traditional methods often incur hidden costs related to the remediation of heavy metal waste and the specialized equipment needed for safe handling. By utilizing MnO2 or m-CPBA, manufacturers can bypass these expenses entirely. Additionally, the high conversion rates and simplified workup procedures reduce solvent usage and energy consumption, leading to significant operational savings. The ability to achieve high yields without extensive purification steps further drives down the cost per kilogram, making the final intermediates more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the availability of the key reagents involved in this process. Manganese dioxide and m-CPBA are widely produced industrial chemicals with established global supply networks, unlike niche organometallic catalysts that may have single-source dependencies. This diversification of the supply base reduces the risk of production stoppages due to vendor issues. Moreover, the mild reaction conditions reduce the wear and tear on reactor vessels and ancillary equipment, extending asset life and minimizing unplanned maintenance downtime. This reliability is crucial for meeting the strict delivery timelines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards typically associated with heavy metal oxidations. The reaction can be safely conducted in standard glass-lined or stainless steel reactors without the need for specialized containment systems. From an environmental standpoint, the process generates significantly less hazardous waste, simplifying compliance with increasingly stringent environmental protection laws. This eco-friendly profile enhances the corporate sustainability metrics of the manufacturer, appealing to environmentally conscious partners and investors who prioritize green chemistry initiatives in their sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the oxidative coupling technology described in patent CN101747126A. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. They serve to clarify the practical implications of adopting this novel synthetic route for the production of phenanthrene and biphenyl derivatives. Understanding these details is vital for technical teams evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the advantages of using MnO2 over traditional thallium catalysts?
A: Traditional methods often utilize toxic heavy metals like thallium trifluoroacetate or lead acetate, which pose significant environmental and safety hazards. The use of MnO2 or m-CPBA eliminates these toxic heavy metals, simplifying waste treatment and reducing regulatory compliance burdens while maintaining high conversion efficiency.
Q: What is the optimal temperature range for this oxidative coupling reaction?
A: The reaction can be conducted within a broad temperature range of -30°C to 80°C. However, experimental data indicates that the optimal temperature range for maximizing yield and selectivity is between 0°C and 20°C, allowing for energy-efficient processing without requiring extreme cooling or heating infrastructure.
Q: Can this method be applied to synthesize binaphthol derivatives?
A: Yes, the methodology is highly versatile. By substituting the stilbene substrate with 2-naphthol, the same oxidative coupling conditions using MnO2 or m-CPBA effectively produce binaphthol derivatives, which are critical chiral ligands in asymmetric synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenanthrene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the MnO2-mediated oxidative coupling can be seamlessly transitioned from benchtop to bulk manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex aromatic couplings positions us as a strategic partner for companies seeking to optimize their API intermediate supply chains.
We invite you to collaborate with us to explore how this technology can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply of high-quality intermediates backed by decades of chemical engineering expertise and a steadfast commitment to quality and sustainability.
