Scalable Production Of Enantiopure 1 1 Spirobiindane 6 6 Diol Derivatives Via Diastereomeric Resolution
The chemical industry continuously seeks robust methodologies for producing enantiomerically pure intermediates, particularly for high-value applications in liquid crystals and advanced polymers. Patent CN1989093A introduces a transformative approach for the chemical separation of 1,1'-spirobiindane-6,6'-diol derivatives, addressing critical bottlenecks in traditional chiral synthesis. This technology leverages diastereomeric resolution through carbonate formation, offering a viable pathway for reliable specialty chemical supplier operations aiming to bypass the limitations of chromatographic separation. By converting racemic mixtures into separable diastereomers using chiral auxiliaries such as menthyl chloroformate, the process achieves high optical purity without the prohibitive costs associated with preparative HPLC. This innovation is pivotal for manufacturers targeting the electronic materials and pharmaceutical sectors, where supply continuity and cost efficiency are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of non-racemic 1,1'-spirobiindane-6,6'-diol derivatives has relied heavily on enzymatic resolution or chiral chromatography, both of which present significant operational challenges for large-scale manufacturing. Enzymatic methods often require precise control over reaction temperature and pH over extended periods, sometimes lasting several days, which complicates process management and increases energy consumption. Furthermore, these biological processes frequently necessitate the use of environmentally unfriendly chlorinated solvents like dichloromethane for extraction and purification, raising compliance issues under modern environmental regulations. Preparative chromatography, while effective for analytical quantities, is economically unfeasible for multi-kilogram production due to high solvent consumption, low throughput, and the substantial capital investment required for column packing and maintenance. These factors collectively hinder the ability to achieve cost reduction in electronic chemical manufacturing, forcing producers to seek more efficient alternatives.
The Novel Approach
The methodology disclosed in the patent circumvents these obstacles by employing a chemical resolution strategy based on diastereomer formation and selective crystallization. Instead of relying on subtle differences in adsorption affinity on a chiral stationary phase, this approach chemically modifies the racemic substrate into diastereomeric dicarbonates using a chiral reagent like (-)-menthyl chloroformate. These diastereomers exhibit distinct physical properties, specifically solubility differences, which allow for their separation through simple crystallization techniques using green solvents such as heptane. This shift from chromatography to crystallization represents a fundamental improvement in process economics, drastically simplifying the workflow and eliminating the need for complex equipment. The resulting process is not only faster but also more amenable to standard chemical engineering practices, ensuring that high-purity OLED material or liquid crystal precursors can be produced with consistent quality and reduced environmental impact.
Mechanistic Insights into Diastereomeric Carbonate Formation
The core of this technology lies in the stereoselective derivatization of the racemic diol to create diastereomeric pairs that are physically separable. The reaction initiates with the treatment of the racemic 1,1'-spirobiindane-6,6'-diol with a chiral chloroformate in the presence of a base such as triethylamine and a catalyst like DMAP. This nucleophilic substitution converts the hydroxyl groups into carbonate esters, linking the chiral information of the auxiliary (e.g., menthol) to the spiro-center of the substrate. The resulting mixture contains two diastereomers which, unlike enantiomers, possess different lattice energies and solubilities in organic media. By carefully selecting the crystallization solvent, typically a non-polar alkane like heptane, one diastereomer preferentially precipitates out of the solution while the other remains dissolved. This thermodynamic control allows for the isolation of a single diastereomer with high purity, effectively enriching the desired enantiomeric configuration of the spirobiindane core before the final deprotection step.
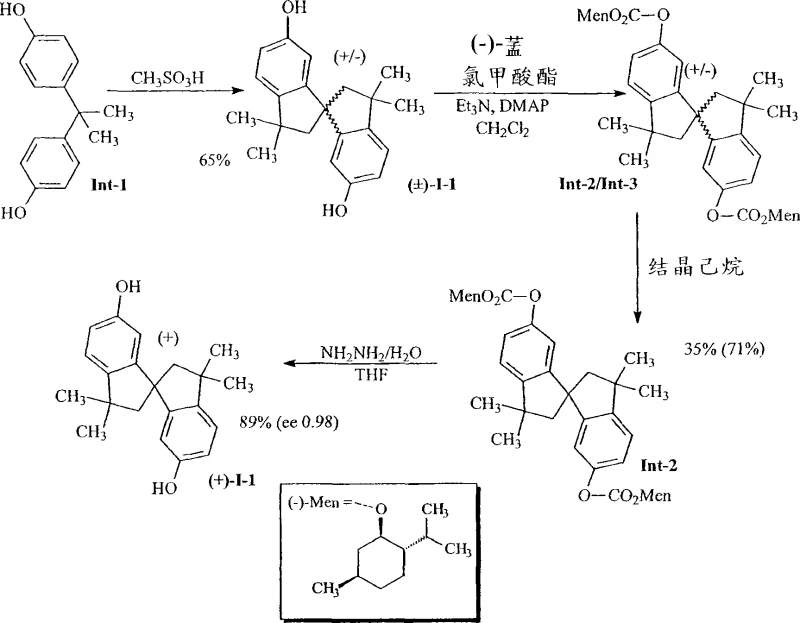
Following the isolation of the pure diastereomeric carbonate, the chiral auxiliary must be removed to regenerate the free diol without racemization. The patent describes effective deprotection strategies using hydrazine hydrate or alkaline hydrolysis under mild conditions. Hydrazinolysis, performed in tetrahydrofuran (THF) under reflux, cleaves the carbonate linkage efficiently, releasing the chiral alcohol and the target spirobiindane diol. Alternatively, basic hydrolysis using sodium hydroxide in a methanol-THF mixture provides a robust route for ester cleavage. Crucially, these deprotection conditions are optimized to preserve the stereochemical integrity established during the crystallization step, ensuring that the final product retains an enantiomeric excess (ee) of greater than 98%. This mechanistic robustness guarantees that the optical purity achieved during separation is not compromised during the final workup, delivering a product suitable for sensitive applications in nematic liquid crystal compositions.
How to Synthesize 1,1'-Spirobiindane-6,6'-diol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from racemic starting materials to enantiopure products through a sequence of derivatization, crystallization, and deprotection. The process begins with the acid-catalyzed cyclization of bisphenol A to form the racemic spiro-compound, followed by reaction with (-)-menthyl chloroformate to generate the diastereomeric mixture. The critical purification step involves dissolving the crude mixture in heptane and inducing crystallization, which selectively isolates the desired diastereomer. Finally, the chiral auxiliary is removed to yield the target diol. For detailed operational parameters, stoichiometry, and specific workup procedures required to replicate this high-efficiency synthesis, please refer to the standardized guide below.
- Synthesize racemic 1,1'-spirobiindane-6,6'-diol derivative via acid-catalyzed cyclization of bisphenol A.
- React the racemic diol with a chiral auxiliary like (-)-menthyl chloroformate to form diastereomeric dicarbonates.
- Separate the diastereomers via selective crystallization in heptane, followed by deprotection using hydrazine or base hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this diastereomeric resolution process offers substantial strategic benefits regarding cost structure and supply reliability. By eliminating the dependency on preparative chromatography, the process removes a major bottleneck that typically limits production volume and inflates operating expenses. The replacement of toxic chlorinated solvents with safer alternatives like heptane and THF reduces hazardous waste disposal costs and simplifies regulatory compliance, contributing to a more sustainable manufacturing footprint. Furthermore, the use of crystallization as the primary purification technique enhances the scalability of the operation, allowing for seamless transition from pilot plant batches to commercial tonnage without the need for specialized separation equipment. These improvements collectively drive significant cost savings and ensure a more resilient supply chain for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps results in a drastic reduction in solvent consumption and processing time, directly lowering the variable costs associated with production. By utilizing inexpensive chiral auxiliaries that can potentially be recovered and recycled, the overall material cost is optimized compared to purchasing pre-resolved starting materials or using expensive enzymatic kits. The simplified workflow also reduces labor hours and utility consumption, as crystallization is a passive separation method that requires less active monitoring than enzymatic reactions or HPLC runs. Consequently, the total cost of goods sold (COGS) for the enantiopure diol is significantly decreased, enhancing profit margins for downstream applications.
- Enhanced Supply Chain Reliability: Relying on standard chemical unit operations like crystallization and filtration ensures that production is not constrained by the availability of specialized chiral columns or enzyme batches, which can suffer from supply disruptions. The robustness of the chemical resolution method means that production schedules can be maintained with greater predictability, reducing lead time for high-purity pharmaceutical intermediates. Additionally, the process tolerance for scale-up implies that suppliers can rapidly respond to increased demand spikes without requiring extensive capital expenditure on new infrastructure. This flexibility is crucial for maintaining continuous supply to global clients in the fast-moving electronics and pharma sectors.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex polymer additives by avoiding the linear scaling issues associated with chromatography, where column size and solvent volume become prohibitive at large scales. The shift away from benzene and dichloromethane towards greener solvents aligns with increasingly strict global environmental regulations, mitigating the risk of regulatory shutdowns or fines. Waste streams are easier to treat and manage, reducing the environmental liability of the manufacturing site. This alignment with green chemistry principles not only safeguards the supply chain but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on purity, scalability, and process mechanics. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines.
Q: What is the primary advantage of this resolution method over enzymatic separation?
A: This chemical resolution method avoids the long reaction times, complex pH control, and extensive chromatographic purification required by enzymatic processes, significantly improving scalability and reducing solvent consumption.
Q: Can this process be scaled for industrial production of liquid crystal dopants?
A: Yes, the process replaces preparative chromatography with crystallization, a unit operation that is inherently scalable from kilograms to multi-ton production levels, making it ideal for industrial supply chains.
Q: What level of enantiomeric purity can be achieved?
A: The patent data demonstrates that following the crystallization and deprotection steps, enantiomeric excess (ee) values greater than 98% can be consistently achieved, meeting stringent specifications for high-performance materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1'-Spirobiindane-6,6'-diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation liquid crystals and functional polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,1'-spirobiindane-6,6'-diol meets the exacting standards required for high-performance applications. Our capability to implement the diastereomeric resolution process described in CN1989093A allows us to offer a competitive, scalable, and environmentally responsible supply solution.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to our resolved intermediates. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, quality, and mutual success.
