Scalable Catalyst-Free Synthesis of 1,2-Diketone Imidazole Heterocycles for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways to construct complex heterocyclic scaffolds, particularly those with proven biological activity. Patent CN109776529B introduces a groundbreaking synthesis method for 1,2-diketone imidazole heterocyclic compounds, a structural motif frequently found in bioactive molecules and potential oncology therapeutics. This technology represents a significant paradigm shift by achieving direct C-C bond construction through a double C-H bond activation mode without the necessity of any external catalyst or additive. The core innovation lies in the utilization of simple, commercially available starting materials—specifically imidazo[1,2-a]pyridine compounds and phenylglyoxal hydrates—reacting in a xylene solvent under mild thermal conditions. 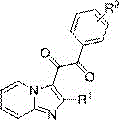 By eliminating the reliance on transition metal catalysts, this process not only adheres to the principles of green chemistry but also drastically simplifies the downstream purification workflow, addressing a critical pain point in the manufacturing of high-purity pharmaceutical intermediates.
By eliminating the reliance on transition metal catalysts, this process not only adheres to the principles of green chemistry but also drastically simplifies the downstream purification workflow, addressing a critical pain point in the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2-diketone imidazole heterocyclic systems has heavily relied on transition metal catalysis or harsh halogenated substrates. Prior art literature, such as studies by Shakoor et al. and Samanta et al., describes protocols utilizing copper bromide with bipyridine ligands or non-metallic iodine catalysts in polar solvents like DMSO. While these methods can achieve reasonable yields, they introduce significant complications for industrial scale-up. The presence of metal catalysts necessitates rigorous and costly purification steps to reduce residual metal content to ppm levels, a requirement strictly enforced by regulatory bodies for Active Pharmaceutical Ingredients (APIs). Furthermore, the use of stoichiometric oxidants or activated halogenated precursors often results in poor atom economy and generates substantial hazardous waste, increasing both the environmental footprint and the overall production cost. These traditional routes often suffer from limited substrate tolerance and require inert atmospheres, adding operational complexity to the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN109776529B offers a streamlined, catalyst-free alternative that leverages aerobic oxidation in a non-polar solvent system. The reaction proceeds efficiently in xylenes at 80°C under ambient air, utilizing the oxygen in the air as the sole oxidant. 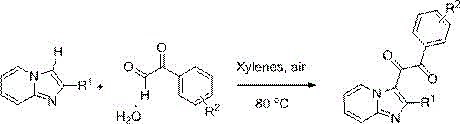 This approach completely bypasses the need for expensive metal catalysts, ligands, or pre-functionalized substrates, thereby enhancing the atom economy and reducing the E-factor of the process. Experimental optimization data within the patent indicates that xylene is superior to other solvents like toluene, DMF, or acetonitrile, delivering yields up to 82% without any additives. The mild reaction conditions and the absence of toxic reagents make this protocol exceptionally suitable for the commercial scale-up of complex pharmaceutical intermediates, offering a cleaner and more cost-effective route to valuable heterocyclic building blocks.
This approach completely bypasses the need for expensive metal catalysts, ligands, or pre-functionalized substrates, thereby enhancing the atom economy and reducing the E-factor of the process. Experimental optimization data within the patent indicates that xylene is superior to other solvents like toluene, DMF, or acetonitrile, delivering yields up to 82% without any additives. The mild reaction conditions and the absence of toxic reagents make this protocol exceptionally suitable for the commercial scale-up of complex pharmaceutical intermediates, offering a cleaner and more cost-effective route to valuable heterocyclic building blocks.
Mechanistic Insights into Catalyst-Free Double C-H Activation
The mechanistic elegance of this transformation lies in its ability to activate two distinct C-H bonds simultaneously without external promotion. The reaction likely proceeds through a radical pathway initiated by thermal activation in the presence of molecular oxygen. The imidazo[1,2-a]pyridine nucleus, being electron-rich, undergoes selective C-H activation at the C-3 position, while the phenylglyoxal hydrate serves as the electrophilic coupling partner. The absence of a catalyst suggests that the inherent reactivity of the substrates, combined with the specific solvation effects of xylene and the thermal energy provided at 80°C, is sufficient to overcome the activation barrier for C-C bond formation. This double C-H activation mode is highly atom-economical, as the only byproduct of the oxidative coupling is water, derived from the dehydration of the glyoxal hydrate and the oxidation process. Such a mechanism minimizes side reactions typically associated with metal-mediated pathways, such as homocoupling or over-oxidation, leading to a cleaner crude reaction profile.
From an impurity control perspective, this catalyst-free system offers distinct advantages for maintaining high product purity. In metal-catalyzed reactions, trace metals can coordinate with the product or intermediates, forming stable complexes that are difficult to remove and may catalyze degradation during storage. By removing the metal entirely, the risk of metal-induced decomposition is eliminated. Furthermore, the use of xylene, a high-boiling non-polar solvent, facilitates easy removal via distillation or crystallization, unlike high-boiling polar solvents like DMSO or NMP which are notoriously difficult to strip completely. The patent data highlights that varying the oxygen concentration or reaction time can fine-tune the yield, indicating a robust process window where the reaction kinetics can be managed to suppress the formation of over-oxidized byproducts, ensuring a consistent and high-quality output suitable for sensitive pharmaceutical applications.
How to Synthesize 1,2-Diketone Imidazole Heterocycles Efficiently
The synthetic protocol outlined in the patent provides a clear and reproducible pathway for generating these valuable heterocycles. The process begins with the preparation of the imidazo[1,2-a]pyridine precursor, followed by the key oxidative coupling step. The operational simplicity is a major highlight, as the second step requires merely mixing the substrates in xylene and heating under air, avoiding the need for gloveboxes or specialized pressure equipment. This ease of execution makes the technology highly accessible for process chemists looking to implement this route in pilot or production plants. For detailed standard operating procedures and specific molar ratios optimized for maximum yield, please refer to the structured guide below.
- Prepare the imidazo[1,2-a]pyridine precursor by reacting 2-aminopyridine with acetophenone derivatives using cuprous iodide and boron trifluoride diethyl etherate in DMF at 60°C.
- Mix the prepared imidazo[1,2-a]pyridine with phenylglyoxal hydrate in xylene solvent without adding any external catalyst or additive.
- Heat the reaction mixture to 80°C under air atmosphere for 6 hours, then purify the resulting 1,2-diketone product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalyst-free synthesis route translates into tangible strategic benefits regarding cost stability and supply reliability. The elimination of transition metal catalysts removes a volatile cost component from the Bill of Materials (BOM), as prices for metals like copper or palladium can fluctuate significantly based on global market dynamics. Moreover, the reliance on commodity chemicals like xylene and air as reagents ensures a stable and abundant supply chain, reducing the risk of raw material shortages that often plague specialty chemical manufacturing. The simplified workup procedure, which avoids complex metal scavenging steps, significantly reduces the consumption of auxiliary materials and processing time, leading to substantial cost savings in the overall manufacturing operation.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of catalyst and ligand costs. In traditional methods, the expense of the catalyst system plus the subsequent purification media (such as specialized scavengers or extensive chromatography) can account for a significant portion of the COGS. By running the reaction in xylene with air, the process utilizes some of the cheapest and most readily available reagents in the chemical industry. Additionally, the high atom economy means less raw material is wasted as byproducts, further driving down the effective cost per kilogram of the final API intermediate. This efficiency allows for more competitive pricing strategies when supplying high-purity pharmaceutical intermediates to downstream clients.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of non-hazardous, widely available solvents and reagents. Unlike processes requiring specialized anhydrous conditions or inert gases, this method operates under ambient air, simplifying the engineering controls required in the production facility. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures related to complex atmosphere control systems. Furthermore, the avoidance of regulated heavy metals simplifies the regulatory compliance landscape, reducing the administrative burden and potential delays associated with environmental reporting and waste disposal, ensuring a smoother and more continuous flow of goods.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the absence of exothermic risks often associated with strong oxidants or reactive metal species. The use of xylene allows for easy solvent recovery and recycling, aligning with modern sustainability goals and reducing the volume of hazardous waste generated. This environmental compatibility is increasingly critical for maintaining social license to operate and meeting the stringent ESG (Environmental, Social, and Governance) criteria set by major pharmaceutical partners. The process design supports the commercial scale-up of complex pharmaceutical intermediates with minimal environmental impact, positioning suppliers who adopt this technology as preferred partners for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits and scope of the method for potential partners and technical stakeholders.
Q: Why is the catalyst-free approach superior for pharmaceutical intermediates?
A: Traditional methods often rely on transition metals like copper or iron, which leave toxic residues requiring expensive removal steps. This catalyst-free method eliminates heavy metal contamination risks, simplifying purification and ensuring higher purity standards essential for API manufacturing.
Q: What are the optimal reaction conditions for this synthesis?
A: The patent identifies xylene as the optimal solvent, providing an 82% yield at 80°C. The reaction utilizes air as the oxidant, avoiding the need for hazardous stoichiometric oxidants, and proceeds efficiently without any additional catalysts or additives.
Q: Does this method support structural diversity for drug discovery?
A: Yes, the method demonstrates excellent substrate scope. It tolerates various substituents on both the imidazo[1,2-a]pyridine ring (such as n-pentyl, cyclopropyl, or phenyl groups) and the phenylglyoxal component (including methoxy and halogen groups), facilitating the rapid generation of diverse libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Diketone Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of catalyst-free technologies in modernizing the supply chain for critical pharmaceutical building blocks. Our team of expert process chemists has extensively evaluated the route described in CN109776529B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,2-diketone imidazole intermediates meets the highest quality standards required for drug development and manufacturing.
We invite R&D directors and procurement leaders to collaborate with us to leverage this advanced synthesis platform for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to green chemistry and operational excellence can drive value and efficiency in your supply chain.
