Scalable Enzymatic Production of Cefcapene Precursor Acid for Global Pharmaceutical Supply Chains
Scalable Enzymatic Production of Cefcapene Precursor Acid for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for producing critical antibiotic intermediates. A significant breakthrough in this domain is detailed in patent CN113025679B, which discloses an enzymatic preparation process for t-butyloxycarbonyl cefcapene precursor acid. This compound serves as a vital building block in the synthesis of cefcapene pivoxil, a broad-spectrum cephalosporin antibiotic used globally for treating respiratory and urinary tract infections. The patented method represents a paradigm shift from traditional chemical condensation to a biocatalytic approach, utilizing immobilized acylase to couple D-7-ACA with the specific side chain acid. For R&D directors and procurement strategists, this technology offers a compelling value proposition by addressing long-standing issues regarding environmental toxicity, energy intensity, and operational complexity inherent in legacy manufacturing routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefcapene intermediates has been plagued by chemically intensive processes that pose significant safety and environmental challenges. As illustrated in prior art such as Patent WO2008/155615, traditional routes often necessitate the use of highly toxic silane-based protecting groups to mask reactive functional groups on the cephalosporin nucleus. 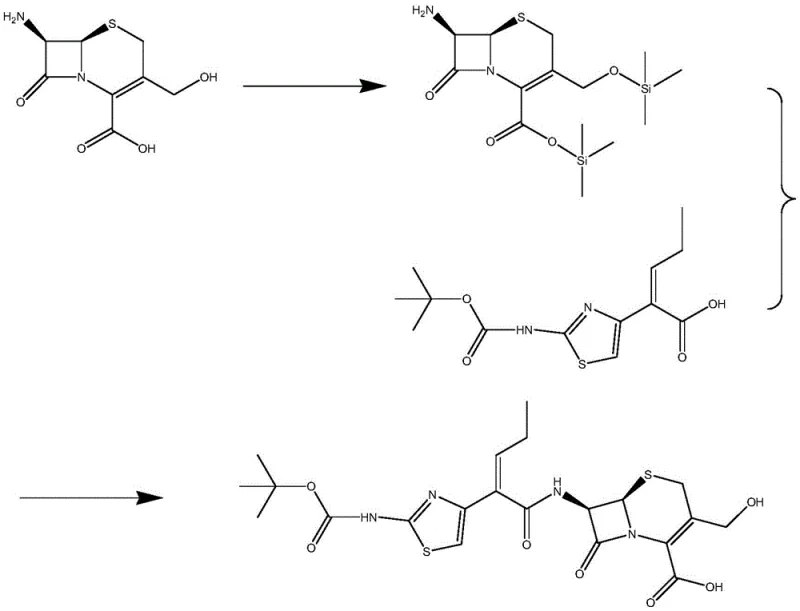 Furthermore, these chemical condensation reactions typically require deep cooling conditions, often dropping to minus 20 degrees Celsius, which imposes a massive energy burden on manufacturing facilities. The reliance on volatile organic solvents like triethylamine and ethyl acetate not only increases the risk of worker exposure to hazardous vapors but also complicates the purification process, often leaving residual solvent impurities that are difficult to remove to acceptable pharmacopeial standards. These factors collectively drive up production costs and create bottlenecks in supply chain continuity due to the stringent handling requirements of the reagents involved.
Furthermore, these chemical condensation reactions typically require deep cooling conditions, often dropping to minus 20 degrees Celsius, which imposes a massive energy burden on manufacturing facilities. The reliance on volatile organic solvents like triethylamine and ethyl acetate not only increases the risk of worker exposure to hazardous vapors but also complicates the purification process, often leaving residual solvent impurities that are difficult to remove to acceptable pharmacopeial standards. These factors collectively drive up production costs and create bottlenecks in supply chain continuity due to the stringent handling requirements of the reagents involved.
The Novel Approach
In stark contrast, the novel enzymatic process described in the patent streamlines the synthesis by leveraging the high specificity of immobilized penicillin acylase. This biological catalyst enables the direct coupling of the cefcapene side chain acid with D-7-ACA in an aqueous medium, effectively bypassing the need for cumbersome protection and deprotection steps. 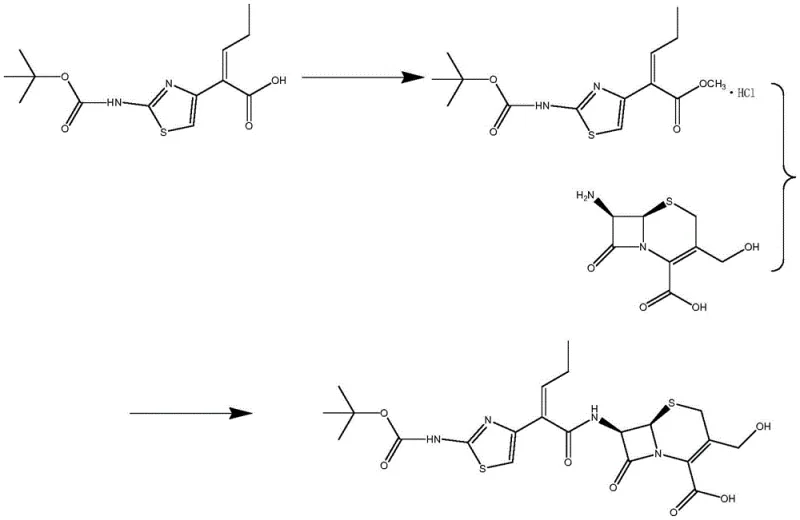 The reaction proceeds under remarkably mild conditions, with temperatures maintained between 11 and 25 degrees Celsius and a neutral pH range of 6.0 to 7.0. This elimination of cryogenic requirements drastically reduces the facility's energy footprint. Moreover, shifting the reaction medium from organic solvents to water fundamentally alters the safety profile of the operation, removing the fire hazards and toxicity risks associated with traditional organic synthesis. The result is a cleaner, more direct pathway that yields the target precursor acid with high efficiency while minimizing the generation of hazardous chemical waste.
The reaction proceeds under remarkably mild conditions, with temperatures maintained between 11 and 25 degrees Celsius and a neutral pH range of 6.0 to 7.0. This elimination of cryogenic requirements drastically reduces the facility's energy footprint. Moreover, shifting the reaction medium from organic solvents to water fundamentally alters the safety profile of the operation, removing the fire hazards and toxicity risks associated with traditional organic synthesis. The result is a cleaner, more direct pathway that yields the target precursor acid with high efficiency while minimizing the generation of hazardous chemical waste.
Mechanistic Insights into Immobilized Acylase Catalysis
The core of this technological advancement lies in the mechanistic action of the immobilized acylase, which facilitates the formation of the amide bond between the beta-lactam nucleus and the side chain. Unlike chemical activators that may attack the sensitive beta-lactam ring or cause epimerization at the chiral centers, the enzyme provides a highly stereoselective environment. The process begins with the activation of the side chain acid using thionyl chloride in methanol, followed by hydrolysis to generate the reactive species in situ. When introduced to the reactor containing D-7-ACA and the immobilized enzyme, the acylase selectively recognizes the specific substrate configuration. The maintenance of the pH between 6.0 and 7.0 is critical, as it ensures the optimal ionization state of the enzyme's active site while preventing the acid-catalyzed degradation of the cephalosporin structure. This precise control over the reaction microenvironment is what allows for the high fidelity of the product structure.
From an impurity control perspective, the enzymatic route offers superior selectivity which translates directly to a cleaner crude product. In chemical synthesis, non-specific acylation can lead to regio-isomers or over-acylated byproducts that are structurally similar to the target molecule and difficult to separate. The biological catalyst, however, restricts the reaction to the desired 7-amino position on the D-7-ACA nucleus. Furthermore, the subsequent crystallization step, achieved by adjusting the pH to 3.5-4.0, leverages the solubility characteristics of the zwitterionic product to further purify the material. This dual mechanism of enzymatic selectivity followed by pH-controlled crystallization ensures that the final API intermediate meets rigorous quality specifications, reducing the burden on downstream purification units and increasing the overall yield of the usable product.
How to Synthesize Cefcapene Precursor Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for manufacturing this critical intermediate. The process is divided into three distinct operational phases: activation of the side chain, enzymatic coupling, and product isolation. The initial activation step involves dissolving the cefcapene tert-butoxycarbonyl side chain acid in methanol and reacting it with thionyl chloride under controlled ice-water bath conditions, followed by a gentle reflux. This prepares the acyl donor for the subsequent biocatalytic step. The detailed standardized synthesis steps, including specific molar ratios and equipment configurations required for GMP compliance, are provided in the guide below.
- Activate the cefcapene tert-butoxycarbonyl side chain acid by dissolving it in methanol and reacting with thionyl chloride under controlled temperature conditions followed by reflux.
- Perform the key coupling reaction in an aqueous reactor by adding D-7-ACA, the activated side chain solution, and immobilized acylase, maintaining pH between 6.0 and 7.0 at mild temperatures.
- Isolate the final product by adjusting the feed liquid pH to 3.5-4.0 to induce crystallization, followed by filtration, washing, and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic process offers tangible strategic benefits beyond mere technical elegance. The removal of exotic and hazardous reagents from the bill of materials simplifies the sourcing strategy and reduces the regulatory burden associated with transporting and storing dangerous chemicals. By eliminating the need for deep cooling infrastructure, manufacturing sites can repurpose energy resources and reduce utility costs significantly. The aqueous nature of the reaction also simplifies waste management, as the effluent is primarily water-based rather than a complex mixture of halogenated or aromatic solvents, leading to substantial cost savings in environmental compliance and waste disposal. These factors combine to create a more resilient and cost-effective supply chain for cefcapene intermediates.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the requirement for expensive silane protecting agents and reduces the consumption of organic solvents like triethylamine and ethyl acetate. By operating at ambient temperatures rather than cryogenic conditions, the facility sees a drastic reduction in energy consumption for refrigeration. The use of immobilized enzymes also opens the possibility for catalyst recycling, further driving down the variable cost per kilogram of the produced intermediate. These cumulative efficiencies result in a leaner cost structure that protects margins against raw material price volatility.
- Enhanced Supply Chain Reliability: Sourcing high-purity D-7-ACA and side chain acids is straightforward, but the reliance on specialized chemical reagents in older methods often created single points of failure. The new method relies on widely available bulk chemicals and robust biocatalysts, diversifying the supply base. Additionally, the milder reaction conditions reduce the risk of batch failures due to thermal runaway or equipment malfunction, ensuring more consistent delivery schedules. This reliability is crucial for maintaining continuous production lines for the final antibiotic formulation, preventing costly stockouts in the pharmaceutical market.
- Scalability and Environmental Compliance: The shift to an aqueous system inherently reduces the volume of volatile organic compounds (VOCs) emitted during production, facilitating easier compliance with increasingly strict environmental regulations. The process is highly scalable, as the heat transfer requirements are minimal compared to exothermic chemical condensations, allowing for larger batch sizes without proportional increases in cooling capacity. This scalability ensures that suppliers can rapidly ramp up production to meet surges in demand for cefcapene pivoxil, providing a secure supply foundation for global health needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of adopting this greener manufacturing technology.
Q: What are the primary advantages of the enzymatic route over traditional chemical synthesis for this intermediate?
A: The enzymatic process eliminates the need for toxic silane protecting groups and hazardous organic solvents like triethylamine. It operates in an aqueous phase at mild temperatures (11-25°C), significantly reducing energy consumption compared to the deep cooling (-20°C) required by conventional methods.
Q: How does this process impact the purity profile of the cefcapene precursor?
A: By utilizing immobilized acylase with high specificity, the reaction minimizes side reactions often associated with harsh chemical condensing agents. The mild pH control (6.0-7.0) during the reaction and precise crystallization pH (3.5-4.0) ensure a cleaner impurity profile and higher structural integrity of the beta-lactam ring.
Q: Is this enzymatic method suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. The use of immobilized enzymes allows for potential reuse, and the aqueous reaction medium simplifies downstream processing and waste treatment, making it highly viable for industrial production ranging from pilot batches to multi-ton annual capacity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefcapene Precursor Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics. Our technical team has extensively analyzed the enzymatic pathways described in recent patents and possesses the expertise to implement these advanced green chemistry solutions. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material regardless of market fluctuations. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cefcapene precursor acid meets the highest international standards for pharmaceutical use.
We invite pharmaceutical partners to collaborate with us to leverage these technological advancements for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized enzymatic processes can enhance your production efficiency and reduce your overall environmental footprint.
