Advanced Chiral Resolution Technology for High-Purity Rotigotine Intermediates and Commercial Scalability
Introduction to Novel Chiral Synthesis Strategies
The pharmaceutical landscape for Parkinson's disease treatment continues to evolve, driven by the demand for high-purity dopaminergic agonists like Rotigotine. A pivotal advancement in this domain is documented in patent CN102010400A, which discloses a sophisticated method for preparing S-5-substituted-N-2'-(thien-2-yl)ethyl-1,2,3,4-tetrahydronaphthalen-2-amine and its subsequent conversion into Rotigotine. This technology represents a significant departure from traditional synthetic pathways, addressing critical bottlenecks related to stereochemical control and process efficiency. By leveraging a novel chiral resolution strategy using conventional chiral acids such as L-(+)-tartaric acid, the inventors have established a route that delivers optical purity exceeding 96% through straightforward recrystallization techniques. For R&D directors and process chemists, this patent offers a compelling blueprint for optimizing the manufacture of complex tetrahydronaphthalene derivatives, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards for enantiomeric excess while maintaining economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Rotigotine has been plagued by inefficiencies inherent in multi-step linear sequences. Traditional routes often commence with 5-methoxy-2-tetralone, proceeding through reductive amination, followed by cumbersome acylation and reduction steps to introduce the necessary side chain. These classical methodologies suffer from several distinct disadvantages: the acylation-reduction sequence is chemically complex, prone to generating impurities, and carries a significant risk of partial racemization of the chiral center, thereby compromising the optical purity of the intermediate. Furthermore, alternative optimization attempts involving direct alkylation with leaving-group-functionalized thiophene ethyl derivatives have yielded disappointing results, with reported yields rarely exceeding 55% and necessitating purification via column chromatography. Such reliance on chromatographic separation is a major deterrent for commercial scale-up, as it introduces substantial operational costs, solvent waste, and throughput limitations that are unacceptable for high-volume API manufacturing.
The Novel Approach
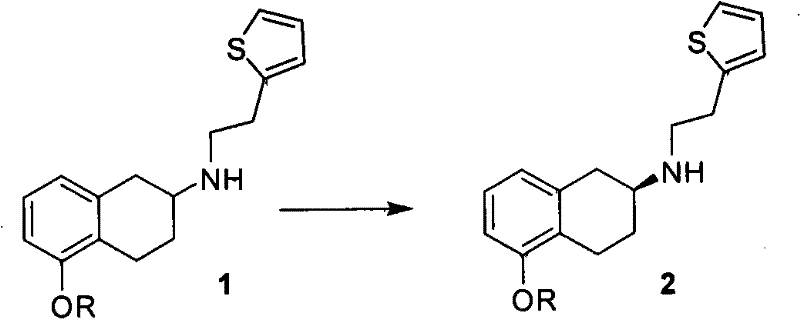
In stark contrast, the methodology outlined in CN102010400A introduces a paradigm shift by prioritizing chiral resolution at an earlier stage of the synthesis. Instead of struggling to maintain chirality through multiple transformations, this approach synthesizes the racemic amine first and then employs a highly selective resolution process. By reacting the racemic 5-substituted-N-2'-(thien-2-yl)ethyl-1,2,3,4-tetrahydronaphthalen-2-amine (Compound 1) with chiral acids like L-(+)-tartaric acid or L-(+)-mandelic acid, the process forms diastereomeric salts that can be easily separated via crystallization. This strategy effectively bypasses the need for expensive chiral catalysts or difficult asymmetric syntheses. The resulting S-configuration intermediate (Compound 2) is obtained with high optical purity after a simple recrystallization step, setting a robust foundation for the subsequent alkylation and deprotection reactions. This streamlined workflow not only simplifies the operational complexity but also drastically improves the overall mass balance and yield of the final Rotigotine product.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technological breakthrough lies in the thermodynamics and kinetics of diastereomeric salt formation. When the racemic amine (Compound 1) interacts with a single enantiomer of a chiral acid, such as L-(+)-tartaric acid, two distinct diastereomeric salts are formed: one involving the (S)-amine and the other involving the (R)-amine. Due to the differences in their crystal lattice energies and solubility profiles in specific solvent systems—preferably a mixture of acetone and water—the less soluble diastereomer precipitates selectively from the solution. The patent specifies that controlling the molar ratio of chiral acid to amine between 0.5:1 and 0.7:1, along with maintaining temperatures between 20°C and 30°C, is critical for maximizing the enrichment of the desired S-enantiomer. This precise control over crystallization conditions allows for the isolation of the S-configuration tartrate salt with an initial optical purity of around 70%, which can be further upgraded to greater than 96% through subsequent recrystallization cycles.
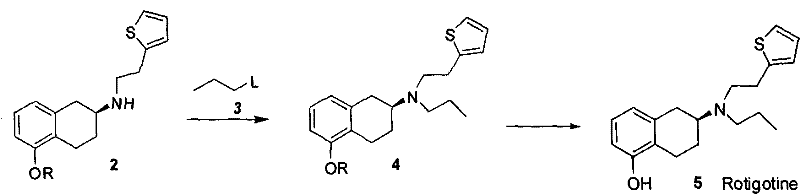
Following the isolation of the chiral salt, the free base is liberated under alkaline conditions, typically using sodium hydroxide or potassium carbonate in a biphasic system of dichloromethane and water. This step is crucial as it regenerates the nucleophilic amine without compromising its stereochemical integrity. The resulting optically pure S-amine (Compound 2) serves as a versatile building block. Its high enantiomeric purity ensures that downstream reactions, specifically the N-alkylation with propyl halides and the final demethylation using boron tribromide, proceed without generating significant amounts of the unwanted R-enantiomer impurity. Consequently, the final Rotigotine product achieves an optical purity of not less than 99.5%, satisfying the rigorous quality requirements for neurological therapeutics.
How to Synthesize S-5-Methoxy-N-2'-(Thien-2-yl)ethyl-1,2,3,4-tetrahydronaphthalen-2-amine Efficiently
The practical implementation of this synthesis route requires careful attention to solvent selection and temperature control during the resolution phase. The process begins with the preparation of the racemic amine via reductive amination, utilizing sodium triacetoxyborohydride in dichloromethane, which provides a high-yielding entry point to the chiral pool. Once the racemate is secured, the critical resolution step is executed by dissolving the amine in an acetone-water mixture and adding the chiral acid solution dropwise. The choice of acetone as the primary solvent is strategic, as it modulates the solubility of the tartrate salt to favor precipitation of the desired isomer. After filtration and washing, the crude chiral salt undergoes recrystallization to boost purity. Finally, the free base is generated and immediately subjected to alkylation. Detailed standardized synthesis steps and specific operating parameters for this high-efficiency route are provided in the guide below.
- Prepare racemic 5-methoxy-N-2'-(thien-2-yl)ethyl-1,2,3,4-tetrahydronaphthalen-2-amine (Compound 1) via reductive amination of 5-methoxy-2-tetralone and 2-thiopheneethylamine.
- Perform chiral resolution by reacting Compound 1 with L-(+)-tartaric acid in an acetone/water solvent system to precipitate the optically pure S-configuration tartrate salt.
- Liberate the free base (Compound 2) using alkaline dissociation, followed by N-alkylation with propyl halide and final deprotection to synthesize Rotigotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN102010400A translates into tangible strategic benefits regarding cost structure and supply reliability. The most significant advantage is the elimination of chromatographic purification steps, which are notoriously resource-intensive and difficult to scale. By replacing column chromatography with crystallization, the process drastically reduces solvent consumption and waste generation, leading to substantially lower operational expenditures. Furthermore, the raw materials required for this synthesis, such as 5-methoxy-2-tetralone and 2-thiopheneethylamine, are commercially available commodity chemicals, reducing the risk of supply chain disruptions associated with specialized or custom-synthesized reagents. This accessibility ensures a stable and continuous supply of the intermediate, which is critical for maintaining uninterrupted API production schedules.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route directly impacts the cost of goods sold (COGS) by minimizing the number of unit operations. Conventional methods often require multiple protection and deprotection steps, as well as expensive reducing agents for acylation reductions. In contrast, this novel approach utilizes a direct resolution strategy that avoids these costly transformations. The ability to achieve high purity through recrystallization rather than preparative HPLC or flash chromatography removes a major cost driver from the manufacturing budget. Additionally, the high yields reported in the patent examples for the alkylation and deprotection steps contribute to better overall material efficiency, ensuring that a greater proportion of input raw materials are converted into valuable final product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical process. The use of common solvents like acetone, water, and dichloromethane means that the manufacturing process is not dependent on exotic or hard-to-source reagents that might face geopolitical or logistical constraints. The simplicity of the workup procedures, involving standard filtration and phase separation, allows for faster batch turnover times in the production facility. This increased throughput capacity enables manufacturers to respond more agilely to market demand fluctuations, reducing lead times for high-purity pharmaceutical intermediates and ensuring that downstream API production lines remain fully stocked.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for commercial scale-up. Crystallization is a unit operation that scales linearly and predictably from the laboratory to the plant floor, unlike chromatography which faces significant engineering challenges at large volumes. The reduction in solvent usage and the avoidance of heavy metal catalysts or hazardous reducing agents align with green chemistry principles, simplifying waste treatment and regulatory compliance. This environmental compatibility not only reduces disposal costs but also enhances the sustainability profile of the supply chain, a factor of increasing importance to global pharmaceutical partners and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of the chiral resolution method described in CN102010400A?
A: The primary advantage is the ability to achieve high optical purity (>96% ee) through simple recrystallization of the diastereomeric tartrate salt, avoiding the need for complex chiral chromatography or enzymatic resolution which are often cost-prohibitive at scale.
Q: How does this route improve upon conventional Rotigotine synthesis methods?
A: Conventional routes often involve lengthy sequences including acylation and reduction steps that risk racemization and lower overall yields. This novel approach utilizes a direct resolution of the racemic amine followed by efficient alkylation, significantly shortening the production cycle and reducing raw material consumption.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process relies on readily available starting materials like 5-methoxy-2-tetralone and uses standard unit operations such as crystallization and filtration. The elimination of column chromatography and the use of robust solvents like acetone and water make it highly amenable to multi-kilogram and ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rotigotine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications like Rotigotine. Our technical team has thoroughly analyzed the methodology disclosed in CN102010400A and possesses the expertise to execute this chiral resolution process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical purity of every batch of S-5-methoxy-N-2'-(thien-2-yl)ethyl-1,2,3,4-tetrahydronaphthalen-2-amine we produce.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for your Rotigotine supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and economics of your Parkinson's disease therapeutic portfolio.
