Advanced Synthesis of 3,4-Dichloro-2-Amino-5-Fluorobiphenyl for Scalable Agrochemical Production
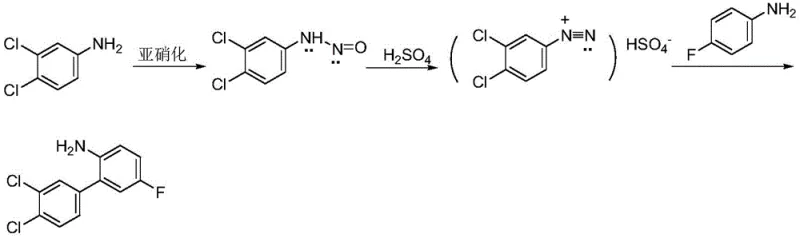
The global demand for high-performance fungicides continues to drive innovation in the synthesis of key agrochemical intermediates. A pivotal development in this sector is detailed in patent CN111072492A, which discloses a robust and environmentally superior method for synthesizing 3,4-dichloro-2-amino-5-fluorobiphenyl. This compound serves as a critical building block for Bixafen, a potent succinate dehydrogenase inhibitor (SDHI) widely used in crop protection. The disclosed technology represents a significant departure from legacy manufacturing routes, addressing long-standing challenges related to heavy metal contamination, hazardous waste generation, and operational complexity. By leveraging a specialized diazotization protocol using nitrosyl sulfuric acid, the process achieves high selectivity and conversion rates while fundamentally altering the waste profile of the reaction. For R&D directors and procurement strategists, understanding this shift is essential for optimizing supply chains and reducing the total cost of ownership in agrochemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 3,4-dichloro-2-amino-5-fluorobiphenyl has relied heavily on transition-metal catalyzed cross-coupling reactions, specifically the Suzuki-Miyaura coupling. This traditional route typically involves the preparation of 3,4-dichlorophenylboronic acid via a Grignard reaction, followed by coupling with a brominated fluoroaniline derivative. While chemically viable, this approach presents severe drawbacks for industrial scale-up. The reliance on palladium catalysts introduces substantial raw material costs and necessitates complex downstream purification steps to remove trace heavy metals to meet regulatory standards. Furthermore, the Grignard step requires strictly anhydrous conditions and the use of tetrahydrofuran (THF), a solvent that poses significant safety risks due to peroxide formation and flammability. Additionally, alternative diazotization methods using sodium nitrite generate stoichiometric amounts of sodium chloride waste, which often contains organic impurities that render it unsuitable for reuse, forcing manufacturers to incur high landfill disposal fees and environmental compliance costs.
The Novel Approach
The methodology outlined in the patent offers a transformative solution by replacing the palladium-catalyzed pathway with a direct diazotization and coupling sequence. Instead of utilizing sodium nitrite, the process employs nitrosyl sulfuric acid as the diazotizing agent in a concentrated sulfuric acid medium. This strategic substitution is crucial because it shifts the inorganic byproduct from sodium chloride to sodium sulfate. Unlike the contaminated salt waste from traditional methods, the sodium sulfate generated here can be effectively decolorized and crystallized into a marketable industrial-grade product. The reaction conditions are notably milder, operating between 0°C and 90°C, which drastically reduces energy consumption compared to the high-temperature requirements of some coupling protocols. This approach not only simplifies the operational workflow by eliminating the need for inert atmosphere handling of pyrophoric reagents but also enhances the overall atom economy of the synthesis, making it a far more sustainable choice for modern chemical production.
Mechanistic Insights into Nitrosyl Sulfuric Acid Diazotization
The core of this synthetic breakthrough lies in the precise generation and utilization of the diazonium intermediate. In the first stage, 3,4-dichloroaniline is dissolved in concentrated sulfuric acid and water, creating a highly acidic environment conducive to protonation. Upon the dropwise addition of nitrosyl sulfuric acid at low temperatures (0-5°C), the nitrosonium ion (NO+) is released, which attacks the amino group of the aniline derivative to form the diazonium salt. This species is stabilized in the acidic medium, preventing premature decomposition. Subsequently, the pH is carefully adjusted using sodium hydroxide to facilitate the coupling phase without isolating the unstable diazonium salt, thereby minimizing safety risks associated with dry diazonium compounds. The mechanistic elegance of this system ensures that the reactive intermediate is consumed immediately in the next step, maintaining high throughput and minimizing side reactions such as phenol formation which often plague aqueous diazotization processes.
Following the formation of the diazonium species, the reaction proceeds to the aryl-aryl bond formation through a coupling mechanism with 4-fluoroaniline. The diazonium salt solution is introduced into an excess of 4-fluoroaniline preheated to 70-90°C. Under these thermal conditions, the diazonium group acts as a leaving group, facilitating a radical or cationic aromatic substitution that links the two aromatic rings. The use of a significant excess of the coupling partner (4-fluoroaniline) drives the equilibrium towards the desired biphenyl product, ensuring high conversion of the valuable dichloro-aniline substrate. Crucially, the absence of transition metals means that the impurity profile is dominated by organic byproducts rather than metal complexes, simplifying the purification strategy. The unreacted 4-fluoroaniline can be easily recovered via reduced pressure distillation and recycled, further enhancing the economic efficiency of the process.
How to Synthesize 3,4-Dichloro-2-Amino-5-Fluorobiphenyl Efficiently
Implementing this synthesis requires careful control of temperature and stoichiometry to maximize yield and safety. The process begins with the preparation of the diazonium salt under inert gas protection to prevent oxidation side reactions. Once the diazonium solution is ready, it is transferred to the coupling vessel containing the heated amine. Post-reaction workup involves standard liquid-liquid extraction using ethyl acetate, followed by washing and drying. A key feature of this protocol is the integrated recovery of the excess amine, which allows for a closed-loop material flow. For detailed operational parameters, including specific mass ratios and temperature ramping rates, please refer to the standardized synthesis guide below.
- Prepare the diazonium salt by reacting 3,4-dichloroaniline with nitrosyl sulfuric acid in concentrated sulfuric acid at 0-5°C, followed by neutralization with sodium hydroxide.
- Perform the coupling reaction by adding the diazonium salt solution to preheated 4-fluoroaniline at 70-90°C under stirring.
- Execute post-treatment via ethyl acetate extraction, washing, drying, and distillation to recover excess amine and isolate the pure biphenyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of palladium catalysts removes a major cost driver and supply bottleneck, as precious metal prices are subject to significant market volatility. Moreover, the transformation of waste salt from a disposal liability into a sellable commodity fundamentally changes the cost structure of the manufacturing process. By producing usable sodium sulfate, the facility can offset waste treatment expenses and potentially generate auxiliary revenue, leading to substantial cost savings in agrochemical intermediate manufacturing. The simplified operational requirements also reduce the need for specialized equipment capable of handling pyrophoric materials or high-pressure hydrogenation, lowering capital expenditure barriers for scale-up.
- Cost Reduction in Manufacturing: The removal of expensive palladium catalysts and the avoidance of anhydrous solvent systems like THF significantly lower raw material expenditures. Furthermore, the ability to recycle the excess 4-fluoroaniline reduces the net consumption of starting materials. The conversion of waste sodium chloride into marketable sodium sulfate eliminates landfill fees, which can be a significant hidden cost in traditional diazotization processes, thereby driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: By avoiding the use of Grignard reagents and other moisture-sensitive organometallics, the process becomes more robust against variations in raw material quality and environmental conditions. This stability ensures consistent batch-to-batch quality and reduces the risk of production delays caused by safety incidents or reagent degradation. The reliance on commodity chemicals like sulfuric acid and sodium hydroxide further secures the supply chain against shortages of specialized reagents.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-compatible workup make this process highly scalable from pilot plant to commercial tonnage without the need for complex engineering controls required for high-pressure or cryogenic reactions. The generation of non-hazardous sodium sulfate waste aligns with increasingly stringent environmental regulations, facilitating easier permitting and reducing the carbon footprint of the production facility, which is a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: How does this method improve upon traditional Suzuki coupling for this intermediate?
A: Unlike Suzuki coupling which requires expensive palladium catalysts and hazardous Grignard reagents, this patented method utilizes a cost-effective diazotization pathway that eliminates heavy metal contamination risks and simplifies the operational workflow.
Q: What is the environmental advantage regarding waste salt management?
A: Traditional sodium nitrite diazotization generates large quantities of sodium chloride waste requiring landfill disposal. This novel process produces sodium sulfate as a byproduct, which can be purified and sold as an industrial commodity, effectively turning a waste liability into a revenue stream.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are mild (0-90°C) and avoid high-pressure or cryogenic requirements typical of Grignard reactions, making it highly adaptable for multi-ton production with enhanced safety profiles and lower energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dichloro-2-Amino-5-Fluorobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global agrochemical supply chain. Our technical team has extensively analyzed the pathway described in CN111072492A and possesses the expertise to optimize this route for maximum efficiency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3,4-dichloro-2-amino-5-fluorobiphenyl meets the exacting standards required for downstream fungicide synthesis.
We invite you to collaborate with us to leverage this advanced technology for your production needs. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this vital agrochemical intermediate.
