Advanced Manufacturing of Telmisartan Intermediates via Novel Catalytic Cyclization Routes
Advanced Manufacturing of Telmisartan Intermediates via Novel Catalytic Cyclization Routes
The pharmaceutical industry continuously seeks robust synthetic pathways for antihypertensive agents, particularly for blockbuster drugs like Telmisartan. A pivotal development in this domain is documented in patent CN113045501B, which discloses a highly efficient preparation method for 7-methyl-2-n-propyl-3H-benzimidazole-5-aldehyde. This specific compound serves as a critical building block in the assembly of the Telmisartan molecule, acting as the key electrophile that condenses with N-methyl-o-phenylenediamine. The significance of this patent lies not merely in the creation of a new molecule, but in the radical restructuring of the synthetic logic to favor industrial viability. By shifting away from traditional multi-step sequences that plague older methodologies, this innovation offers a streamlined approach that addresses the triad of modern chemical manufacturing concerns: cost efficiency, environmental sustainability, and operational simplicity. For global procurement teams and R&D directors, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of generic drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-methyl-2-n-propyl-3H-benzimidazole-5-aldehyde has been fraught with inefficiencies that hinder large-scale commercialization. Prior art, such as the methodology developed by Northeast Agricultural University, relies on o-benzylamine as the starting material. This legacy route necessitates a cumbersome sequence involving acylation, a Blanc chloromethylation reaction, followed by a Sommelet reaction using hexamethylenetetramine to introduce the aldehyde functionality. Subsequent steps involve nitration and a reduction-cyclization sequence utilizing stoichiometric amounts of iron powder in glacial acetic acid. The drawbacks of this approach are manifold and severe from an industrial perspective. Firstly, the reaction steps are excessively long, leading to cumulative yield losses that erode profit margins. Secondly, and perhaps more critically, the use of stoichiometric iron powder for reduction generates massive quantities of iron sludge. This hazardous waste presents a significant environmental burden, requiring costly disposal protocols and complicating regulatory compliance. Furthermore, the overall yield of such convoluted pathways is often suboptimal, making the final API cost-prohibitive in competitive markets.
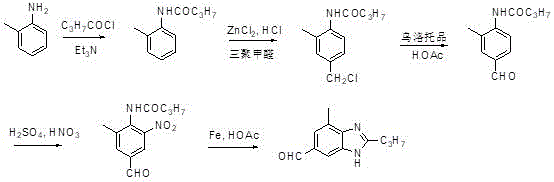
The Novel Approach
In stark contrast to the archaic methods described above, the methodology outlined in CN113045501B introduces a paradigm shift towards convergent and catalytic synthesis. The new route begins with 3-methyl-4-n-butyramido-5-nitrobenzoic acid, a readily available and inexpensive precursor. The innovation here is the strategic use of a sulfonyl hydrazide moiety as a masked functionality that facilitates downstream transformations. Instead of a linear march through protecting group manipulations, this novel approach employs a series of telescoped, one-pot reactions. The initial conversion to the acyl chloride and subsequent coupling with benzenesulfonyl hydrazide is achieved in a single vessel, minimizing solvent usage and handling time. The subsequent reduction and cyclization utilize a catalytic system rather than stoichiometric metals, fundamentally altering the waste profile of the process. This transition from stoichiometric to catalytic reagents is a hallmark of green chemistry, directly translating to reduced raw material consumption and simplified downstream processing. For a reliable pharmaceutical intermediate supplier, adopting such a route means offering a product with a superior impurity profile and a significantly lower carbon footprint.
Mechanistic Insights into Fe/Pd-Catalyzed Cyclization and Thermal Elimination
The core of this synthetic breakthrough lies in the sophisticated manipulation of functional groups through catalytic hydrogenation and thermal elimination. In the first stage, the carboxylic acid is activated using thionyl chloride (SOCl2) to form the reactive acyl chloride in situ. This intermediate is immediately trapped by benzenesulfonyl hydrazide in the presence of triethylamine to form Intermediate (1). This step is crucial as it installs the sulfonyl hydrazide group, which acts as a leaving group precursor for the final aldehyde formation. The reaction conditions are meticulously controlled, with the acylation occurring under reflux and the coupling performed at low temperatures (0°C) to prevent side reactions, ensuring high fidelity in the formation of the hydrazide bond. The avoidance of water introduction during this phase is paramount to prevent hydrolysis of the sensitive acyl chloride, demonstrating the process's robustness against moisture sensitivity.
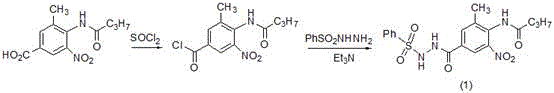
The second mechanistic pillar involves the simultaneous reduction of the nitro group and cyclization of the benzimidazole ring. Intermediate (1) is subjected to hydrogenation in a mixed solvent system of ethanol and glacial acetic acid. The catalyst system comprises iron powder and palladium chloride (PdCl2). While iron alone can reduce nitro groups, the addition of palladium creates a synergistic catalytic effect that operates under mild hydrogen pressure (0.15-0.2 MPa). This catalytic reduction converts the nitro group to an amine, which then spontaneously condenses with the adjacent amide carbonyl to close the benzimidazole ring, yielding Intermediate (2). The use of acetic acid serves a dual purpose: it acts as a solvent and provides the acidic medium necessary to facilitate the cyclization dehydration. This one-pot transformation is a masterclass in atom economy, combining two distinct chemical events into a single operational unit, thereby drastically cutting down on processing time and solvent waste compared to stepwise reductions.
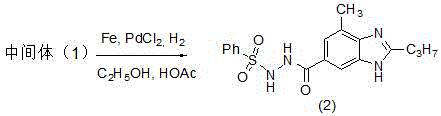
The final transformation is a thermal elimination reaction that unveils the aldehyde functionality. Intermediate (2) is heated in ethylene glycol to high temperatures (160-165°C) in the presence of a solid base such as sodium carbonate or potassium carbonate. Crucially, the patent specifies the addition of solid support materials like industrial zeolite, soft glass powder, or ceramsite. These solids likely serve to disperse the reactants and prevent localized overheating or bumping during the vigorous gas evolution that accompanies the elimination of the benzenesulfinic acid moiety. The mechanism involves the base-mediated deprotonation followed by the extrusion of sulfur dioxide and the benzenesulfinate anion, leaving behind the formyl group at the 5-position of the benzimidazole ring. This step effectively converts the hydrazide surrogate into the desired aldehyde with high precision. The ability to recover sodium benzene sulfinate from the aqueous phase adds an additional layer of economic value, turning a potential waste stream into a saleable byproduct.

How to Synthesize 7-methyl-2-n-propyl-3H-benzimidazole-5-aldehyde Efficiently
Executing this synthesis requires precise adherence to the patented parameters to maximize yield and purity. The process is divided into three distinct operational stages, each optimized for scale-up. The initial acylation-hydrazide coupling sets the foundation for the molecule's architecture, requiring strict anhydrous conditions to maintain the integrity of the acyl chloride. The subsequent catalytic reduction demands careful control of hydrogen pressure and catalyst loading to ensure complete nitro reduction without over-reduction of other sensitive functionalities. Finally, the thermal elimination step requires robust heating equipment capable of sustaining temperatures up to 165°C safely while managing the evolution of gases. Detailed standard operating procedures for these steps are critical for technology transfer and successful commercial implementation.
- React 3-methyl-4-n-butyramido-5-nitrobenzoic acid with thionyl chloride followed by benzenesulfonyl hydrazide to form Intermediate (1).
- Perform catalytic hydrogenation using Fe/PdCl2 in ethanol/acetic acid to reduce the nitro group and cyclize into Intermediate (2).
- Heat Intermediate (2) in ethylene glycol with solid base and support materials at 160-165°C to effect elimination and form the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic simplification of the supply chain. By utilizing 3-methyl-4-n-butyramido-5-nitrobenzoic acid as a starting material, manufacturers bypass the need for multiple upstream precursors like o-benzylamine and the associated reagents for chloromethylation and oxidation. This consolidation of raw materials reduces the number of vendors required and minimizes the risk of supply disruptions caused by bottlenecks in earlier synthesis steps. Furthermore, the high yields reported in the patent examples—consistently exceeding 90% for the first two steps and reaching nearly 80% for the final step—imply a significant reduction in the cost of goods sold (COGS). Higher yields mean less raw material is needed per kilogram of final product, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The economic implications of switching to this catalytic route are profound. Traditional methods relying on stoichiometric iron powder generate tons of hazardous sludge, the disposal of which incurs substantial environmental fees and logistical costs. By replacing this with a catalytic Fe/Pd system, the volume of solid waste is minimized, leading to substantial cost savings in waste management. Additionally, the one-pot nature of the first two steps reduces solvent consumption and energy usage associated with isolation and purification between steps. The recovery of sodium benzene sulfinate as a byproduct further offsets production costs, creating a circular economy within the manufacturing process. These factors combine to offer a highly competitive pricing structure for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals. Reagents such as thionyl chloride, ethanol, acetic acid, and sodium carbonate are globally available bulk chemicals with stable pricing and abundant supply. Unlike specialized reagents that might be sourced from single suppliers, these commodities ensure that production can continue uninterrupted even during market fluctuations. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high pressures, further ensures that the process can be replicated across different manufacturing sites without significant capital expenditure. This flexibility allows for diversified sourcing strategies, reducing the risk of single-point failures in the supply network.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden pitfalls, but this route is designed with scalability in mind. The exothermic nature of the reactions is manageable, and the use of solid supports in the high-temperature elimination step mitigates safety risks associated with thermal runaways. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding heavy metal waste and solvent emissions. The elimination of iron sludge and the reduction in solvent intensity make this process inherently greener. For multinational corporations with strict ESG (Environmental, Social, and Governance) mandates, sourcing intermediates produced via this method supports their sustainability goals. The ability to produce commercial quantities from 100 kgs to 100 MT annually with consistent quality ensures that the supply chain can grow in tandem with market demand for Telmisartan.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding the practical application of patent CN113045501B. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making. Whether you are concerned about catalyst recovery, solvent choices, or byproduct management, the following insights clarify the operational realities of this advanced manufacturing route.
Q: How does this new route improve upon conventional Telmisartan intermediate synthesis?
A: Conventional methods often involve lengthy sequences starting from o-benzylamine with poor yields and significant iron sludge waste. This patent introduces a streamlined 3-step route using cheaper raw materials, one-pot reactions, and catalytic reduction, drastically reducing environmental impact and improving overall yield.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization occurs in Step 2 under nitrogen protection using a catalytic system of Iron powder and Palladium Chloride (PdCl2) in an ethanol and glacial acetic acid solvent mixture. The reaction requires hydrogen pressure of 0.15-0.2 MPa at room temperature, followed by heating to reflux to complete the ring closure.
Q: Is the process scalable for commercial API manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like ethylene glycol and dichloromethane, avoids exotic reagents, and features high-yield one-pot transformations. The recovery of sodium benzene sulfinate as a byproduct further enhances the economic viability and waste management profile for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-methyl-2-n-propyl-3H-benzimidazole-5-aldehyde Supplier
The technical elegance of patent CN113045501B represents the future of efficient API intermediate manufacturing, and NINGBO INNO PHARMCHEM is positioned at the forefront of this evolution. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this synthesis, including high-pressure hydrogenation units and high-temperature reactors necessary for the thermal elimination step. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of 7-methyl-2-n-propyl-3H-benzimidazole-5-aldehyde meets the exacting standards required for antihypertensive drug formulation. Our commitment to quality assurance means that we not only follow the patent but optimize it further to minimize impurities and maximize throughput.
We invite global partners to collaborate with us to leverage this cost-effective technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this novel synthesis can integrate into your existing supply chain. Let us help you secure a stable, high-quality supply of this critical Telmisartan intermediate while driving down costs and enhancing your sustainability profile.
