Revolutionizing Brominated Fused-Ring Aromatic Synthesis for Commercial Scale-Up
Revolutionizing Brominated Fused-Ring Aromatic Synthesis for Commercial Scale-Up
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, safer, and more cost-effective synthetic methodologies. Patent CN107417543B, published in August 2020, presents a groundbreaking approach to synthesizing brominated fused-ring aromatic hydrocarbon compounds, a class of molecules that serves as critical building blocks in the pharmaceutical and agrochemical industries. This technology replaces hazardous liquid bromine and expensive N-bromosuccinimide (NBS) with a novel solid-state reagent system based on Zinc-Aluminum Layered Double Hydroxides (ZnAl-BrO3-LDHs). For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this innovation offers a pathway to high-purity products with drastically reduced environmental impact and operational risk. The method achieves excellent regioselectivity and yield under remarkably mild conditions, addressing long-standing pain points in the production of key precursors for drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bromination of fused-ring systems like naphthalene has relied heavily on elemental bromine (Br2) or N-bromosuccinimide (NBS), both of which present substantial challenges for modern industrial chemistry. The use of elemental bromine is inherently dangerous due to its high volatility, corrosivity, and toxicity; furthermore, the reaction generates stoichiometric amounts of hydrogen bromide (HBr) gas, leading to a theoretical atom utilization of only 50% and creating severe waste disposal issues. On the other hand, while NBS is safer to handle than liquid bromine, it is significantly more expensive and produces succinimide as a byproduct, which often requires complex and energy-intensive purification steps to remove from the final API intermediate. These conventional routes often demand harsh reaction conditions, specialized corrosion-resistant reactors, and rigorous safety protocols that inflate both capital expenditure and operational costs, making them less attractive for sustainable commercial scale-up of complex polymer additives or pharmaceutical precursors.
The Novel Approach
In stark contrast, the methodology disclosed in CN107417543B utilizes a solid ZnAl-BrO3-LDHs reagent in conjunction with inexpensive alkali metal bromides, such as potassium bromide, to achieve efficient bromination. This system operates in a biphasic mixture of water and organic solvents at ambient temperatures ranging from 20°C to 35°C, eliminating the need for extreme thermal inputs or cryogenic cooling. The solid nature of the brominating agent ensures precise control over the release of active bromine species, thereby minimizing side reactions and poly-bromination unless specifically desired. By shifting from hazardous liquids to stable solids, this approach not only enhances workplace safety but also simplifies logistics and storage, offering a robust solution for cost reduction in electronic chemical manufacturing and fine chemical production where purity and consistency are paramount.
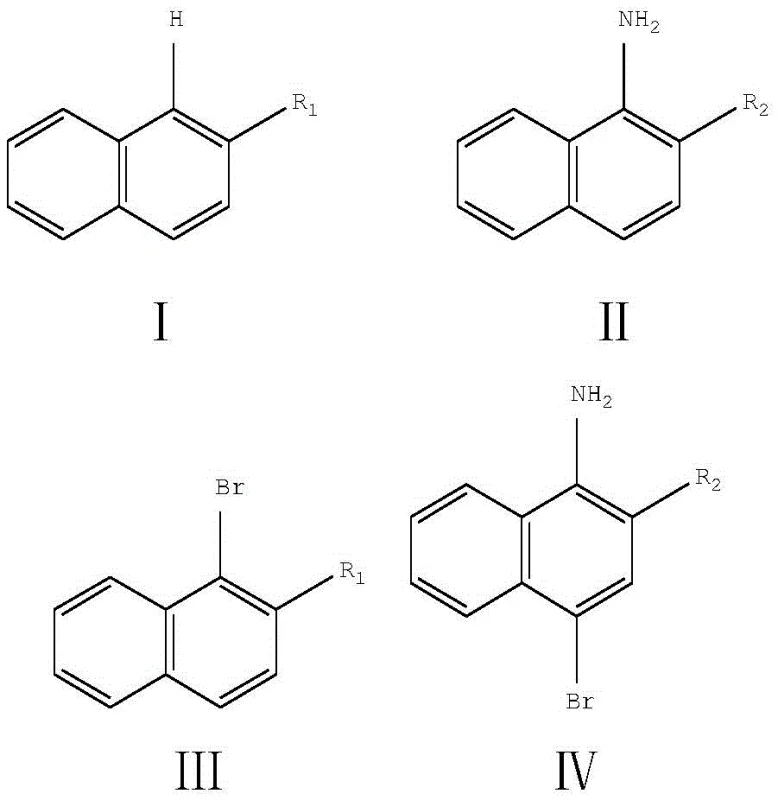
Mechanistic Insights into ZnAl-BrO3-LDHs Catalyzed Bromination
The core innovation of this technology lies in the unique structure and reactivity of the Zinc-Aluminum Layered Double Hydroxide (LDH) intercalated with bromate anions. In this catalytic system, the LDH acts not merely as a support but as a structured oxidant reservoir that facilitates the in situ generation of electrophilic bromine species. When the solid ZnAl-BrO3-LDHs is introduced to the reaction mixture containing the aromatic substrate and bromide ions (Br-), a redox reaction occurs within the interlayer space or at the surface of the material. This controlled generation of active bromine prevents the local concentration spikes typical of adding liquid bromine, which often lead to over-bromination and tar formation. The result is a highly selective electrophilic aromatic substitution that favors the alpha-position of the naphthalene ring, ensuring the production of the desired mono-brominated isomer with high fidelity.
Furthermore, the presence of acetic acid in the solvent system plays a crucial role in protonating intermediate species and maintaining the optimal pH for the redox cycle to proceed efficiently. The mild acidic environment helps to solubilize the inorganic salts while keeping the organic substrate in the organic phase, creating an effective interface for the reaction to occur. This mechanistic elegance translates directly to practical benefits: the reaction mixture can be easily worked up by simple washing with sodium sulfite to quench any residual oxidants, followed by standard extraction. The absence of heavy metal catalysts or complex ligands means that the final product is free from trace metal contaminants, a critical requirement for high-purity OLED material or pharmaceutical intermediate specifications where ppm-level impurities can disqualify a batch.
How to Synthesize Brominated Naphthalene Efficiently
The synthesis protocol outlined in the patent provides a straightforward, reproducible route for converting naphthalene and its derivatives into valuable brominated intermediates. The process begins by dissolving the substrate, such as naphthalene or 2-methylnaphthalene, along with an alkali metal bromide like potassium bromide, in a mixed solvent system typically comprising water, dichloromethane, and acetic acid. The key operational step involves the slow, batch-wise addition of the solid ZnAl-BrO3-LDHs reagent to the stirred mixture while maintaining the temperature between 20°C and 35°C. This controlled addition rate is vital for managing the exotherm and ensuring uniform reaction progress, which is monitored via TLC. Following the reaction period of 4 to 8 hours, the mixture undergoes a simple aqueous workup to remove inorganic salts, and the organic layer is concentrated to yield the crude product, which is then purified by column chromatography to obtain the target compound in high yield.
- Prepare a mixed solvent system of water, dichloromethane, and acetic acid, then dissolve the fused-ring aromatic substrate and alkali metal bromide.
- Slowly add the solid ZnAl-BrO3-LDHs reagent to the mixture at 20-35°C while stirring to initiate the controlled bromination reaction.
- Upon completion, wash the mixture with sodium sulfite, extract with dichloromethane, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this LDH-based bromination technology represents a strategic opportunity to optimize both cost structures and risk profiles. The shift from liquid bromine to solid reagents fundamentally alters the safety requirements of the manufacturing facility, potentially lowering insurance premiums and reducing the need for specialized containment infrastructure. Moreover, the use of commodity chemicals like potassium bromide and acetic acid ensures a stable and resilient supply chain, insulating production from the volatility associated with specialized reagents like NBS. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a smaller carbon footprint and aligning with increasingly stringent environmental regulations faced by global chemical manufacturers today.
- Cost Reduction in Manufacturing: The elimination of expensive N-bromosuccinimide and the replacement of hazardous elemental bromine with low-cost alkali metal bromides results in substantial raw material savings. Additionally, the high atom economy of this method means less waste is generated per kilogram of product, reducing the costs associated with waste treatment and disposal. The simplified workup procedure, which avoids complex distillation or recrystallization steps often needed to remove succinimide byproducts, further lowers labor and utility costs, driving down the overall cost of goods sold for these critical intermediates.
- Enhanced Supply Chain Reliability: Relying on solid, stable reagents like ZnAl-BrO3-LDHs and potassium bromide mitigates the risks associated with transporting and storing corrosive liquids. This stability ensures consistent quality across batches and reduces the likelihood of supply disruptions caused by regulatory restrictions on hazardous material transport. For a reliable agrochemical intermediate supplier, this means the ability to guarantee uninterrupted delivery schedules to downstream customers, fostering stronger long-term partnerships and trust in the supply network.
- Scalability and Environmental Compliance: The benign nature of the reagents and the aqueous-organic solvent system makes this process highly scalable from laboratory benchtop to multi-ton production without significant re-engineering. The absence of heavy metals and the generation of minimal hazardous waste simplify compliance with environmental protection laws, such as REACH in Europe or TSCA in the US. This ease of compliance accelerates the time-to-market for new drug candidates relying on these intermediates and ensures long-term viability of the manufacturing process in a regulated environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green bromination technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production workflows or for sourcing teams negotiating supply agreements for brominated aromatic compounds.
Q: What are the primary safety advantages of using ZnAl-BrO3-LDHs over elemental bromine?
A: Unlike elemental bromine, which is a corrosive liquid releasing toxic HBr gas and posing severe handling risks, ZnAl-BrO3-LDHs is a stable solid. This eliminates the need for specialized corrosion-resistant equipment and drastically reduces operator exposure to hazardous vapors during the bromination process.
Q: How does this method improve atom economy compared to traditional NBS bromination?
A: Traditional N-bromosuccinimide (NBS) methods generate stoichiometric amounts of succinimide waste, complicating purification. The LDH-based method utilizes alkali metal bromides efficiently with high atom utilization, minimizing organic waste streams and simplifying the downstream isolation of high-purity intermediates.
Q: Is this synthesis route scalable for industrial production of API intermediates?
A: Yes, the reaction operates at mild temperatures (20-35°C) without requiring cryogenic cooling or high-pressure vessels. The use of common solvents like dichloromethane and acetic acid, combined with a simple aqueous workup, makes the process highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brominated Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener synthetic routes is not just an environmental imperative but a commercial necessity for staying competitive in the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative ZnAl-BrO3-LDHs bromination method can be seamlessly transferred from the lab to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of brominated fused-ring aromatic hydrocarbon compounds we deliver meets the exacting standards required for pharmaceutical and electronic applications.
We invite forward-thinking partners to collaborate with us to leverage this advanced technology for their next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can drive value and efficiency in your supply chain.
