Scalable Silver-Catalyzed Dearomatization for High-Purity Pharmaceutical Intermediates Manufacturing
Introduction to Advanced Dearomatization Technology
The landscape of fine chemical synthesis is constantly evolving, driven by the need for greener, more efficient, and highly selective methodologies. A significant breakthrough in this domain is documented in patent CN111393333B, which discloses a novel preparation method for 4-amino substituted cyclohexadienone derivatives. This technology addresses long-standing challenges in the functionalization of phenolic compounds, specifically achieving para-amination dearomatization with exceptional precision. Unlike traditional approaches that often rely on hazardous reagents or complex multi-step sequences, this invention utilizes a transition metal-catalyzed system operating in an aqueous phase. The core innovation lies in the use of silver oxide (Ag2O) as a catalyst to facilitate the reaction between p-cresol and various azodicarboxylates. This approach not only streamlines the synthetic route but also aligns with modern sustainability goals by employing water as the primary solvent. For R&D directors and process chemists, this represents a pivotal shift towards more robust and environmentally benign manufacturing protocols for complex cyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the dearomatization of phenols to generate cyclohexadienone scaffolds has been fraught with significant technical hurdles. Conventional strategies typically depend on high-valence iodine reagents or harsh transition metal-catalyzed alkylation and arylation conditions. These methods often suffer from poor chemo- and regioselectivity, primarily because the free phenolic hydroxyl group is both acidic and nucleophilic, leading to preferential O-H substitution rather than the desired C-H functionalization. Furthermore, in electrophilic reactions, the inherent nucleophilicity of both the ortho and para positions on the aromatic ring frequently results in a mixture of isomers, complicating downstream purification. Perhaps most critically, phenols are electron-rich aromatic hydrocarbons that are highly susceptible to oxidative decomposition under vigorous reaction conditions, leading to tar formation and reduced yields. These limitations have historically constrained the utility of phenol dearomatization in the commercial synthesis of high-value intermediates.
The Novel Approach
The methodology outlined in patent CN111393333B offers a transformative solution to these persistent issues by leveraging a mild, silver-catalyzed protocol in a green solvent system. By utilizing Ag2O in conjunction with azodicarboxylates in an aqueous environment, the reaction achieves high regioselectivity for the para-position, effectively bypassing the ortho-amination side reactions that plague other methods. The use of water as a solvent not only mitigates safety risks associated with volatile organic compounds but also simplifies the thermal management of the exothermic dearomatization process. The reaction proceeds smoothly under ice-bath conditions, demonstrating remarkable tolerance and stability. This novel approach ensures that the valuable phenolic starting materials are converted efficiently into the target 4-amino substituted cyclohexadienone derivatives with minimal byproduct formation. 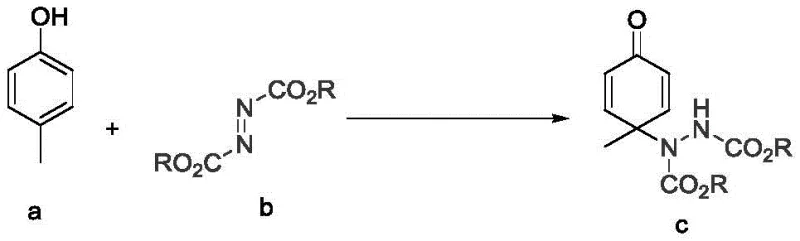
Mechanistic Insights into Ag2O-Catalyzed Para-Amination
The mechanistic pathway of this silver-catalyzed transformation is a testament to the power of transition metal mediation in controlling reactivity. The silver oxide catalyst likely acts as a Lewis acid or a soft metal center that activates the azodicarboxylate electrophile, facilitating its attack on the electron-rich phenolic ring. Crucially, the coordination environment provided by the silver species appears to direct the incoming amine moiety specifically to the para-position, overriding the natural electronic bias that might favor ortho-substitution in uncatalyzed scenarios. This level of control is essential for producing single-isomer intermediates required in pharmaceutical synthesis, where stereochemical and regiochemical purity are paramount. The mild oxidative potential of the system prevents the over-oxidation of the phenol substrate, preserving the integrity of the carbon skeleton while introducing the necessary nitrogen functionality. This delicate balance between activation and preservation is what distinguishes this catalytic cycle from harsher oxidative dearomatization methods.
From an impurity control perspective, the aqueous nature of the reaction plays a vital role in maintaining product quality. Water acts as a heat sink, preventing localized hot spots that could trigger thermal decomposition or polymerization of the reactive quinone-like intermediates. Additionally, the specific choice of azodicarboxylates—such as diisopropyl, di-tert-butyl, or dibenzyl azodicarboxylate—allows for the introduction of versatile protecting groups directly during the bond-forming event. As illustrated in the structural diversity of the products, the method accommodates different steric and electronic environments without compromising yield. 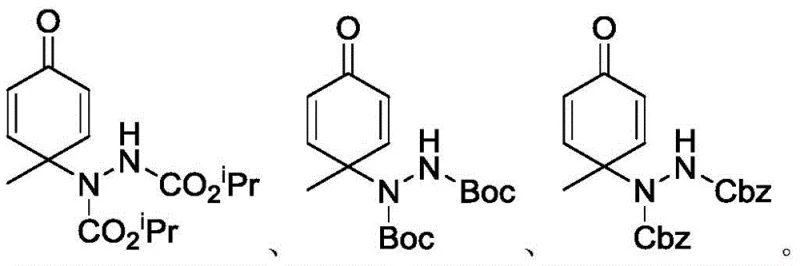
How to Synthesize 4-Amino Substituted Cyclohexadienone Derivatives Efficiently
The operational simplicity of this synthesis makes it highly attractive for process development teams looking to implement robust manufacturing routes. The procedure involves a straightforward charge of reagents into a reactor followed by a controlled stirring period under cooling. The workup relies on standard liquid-liquid extraction techniques, avoiding the need for specialized filtration or distillation equipment that might be sensitive to the reactive intermediates. The patent data indicates that the crude product can be effectively purified using standard column chromatography with ethyl acetate and petroleum ether mixtures, ensuring high purity levels suitable for downstream applications. For detailed operational parameters and stoichiometric ratios, please refer to the standardized synthesis guide below.
- Charge the reactor with silver oxide catalyst (Ag2O), p-cresol (Reactant I), azodicarboxylate (Reactant II), and water solvent.
- Maintain the reaction mixture in an ice bath with stirring for 0.5 to 10 hours to ensure controlled para-amination dearomatization.
- Extract the reaction liquid with ethyl acetate, concentrate under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this silver-catalyzed aqueous process presents compelling economic and logistical benefits. The shift away from expensive, hazardous organic solvents to water drastically reduces the cost of goods sold (COGS) related to solvent purchase, recovery, and waste disposal. Furthermore, the use of commodity chemicals like p-cresol and commercially available azodicarboxylates ensures a stable and resilient supply chain, minimizing the risk of raw material shortages. The mild reaction conditions also translate to lower energy consumption, as the process does not require high temperatures or pressures, thereby reducing the operational expenditure (OPEX) associated with utility usage in the manufacturing plant.
- Cost Reduction in Manufacturing: The elimination of costly hypervalent iodine reagents and the replacement of organic solvents with water lead to substantial cost savings. The catalyst, silver oxide, is used in relatively low loadings, and the simplicity of the workup reduces labor and processing time. By avoiding complex protection-deprotection sequences often required in alternative routes, the overall step count is minimized, which inherently drives down manufacturing costs and improves the overall yield of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The raw materials identified in this patent, specifically p-cresol and various azodicarboxylates, are bulk chemicals produced by multiple global suppliers. This commoditization ensures that the supply chain is not dependent on a single source or a niche vendor, thereby mitigating supply risk. The robustness of the reaction, which tolerates scaling without yield loss, means that production schedules can be met consistently, ensuring reliable delivery timelines for downstream customers who depend on these critical intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: The patent explicitly notes that the reaction yield remains unaffected even when the dosage is enlarged significantly, indicating excellent scalability from gram to ton scale. From an environmental compliance standpoint, the use of water as a solvent and the generation of minimal hazardous waste align perfectly with increasingly stringent global environmental regulations. This green chemistry profile facilitates easier permitting for new manufacturing lines and enhances the corporate sustainability metrics of companies adopting this technology, making it a future-proof choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dearomatization technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical aspects of the synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of using water as a solvent in this dearomatization reaction?
A: Using water as the reaction medium significantly reduces environmental impact and disposal costs compared to traditional organic solvents. It also simplifies the workup process, as the product can be easily extracted into an organic phase while inorganic salts and catalyst residues remain in the aqueous layer, enhancing overall process efficiency.
Q: How does the silver oxide catalyst improve regioselectivity compared to conventional methods?
A: Conventional electrophilic substitutions on phenols often suffer from mixed ortho/para selectivity or oxidative decomposition. The Ag2O-catalyzed system specifically promotes para-amination dearomatization, suppressing ortho-substitution and preventing the oxidative degradation of the electron-rich phenolic ring, resulting in a cleaner impurity profile.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates that scaling the reactant dosage by 100 times does not negatively affect the reaction yield. The mild conditions (ice bath, atmospheric pressure) and the use of inexpensive, commercially available raw materials like p-cresol make it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino Substituted Cyclohexadienone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of drug development and commercial manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-amino substituted cyclohexadienone derivatives meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for multinational corporations seeking reliable sources for complex fine chemical intermediates.
We invite you to collaborate with us to leverage this advanced silver-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this green synthesis route can optimize your budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
