Scalable Production of Dehydroisoandrosterone: A Novel Chemical Route for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of critical steroid intermediates. Patent CN102603841B introduces a transformative preparation method for dehydroisoandrosterone (DHEA), a pivotal precursor in the synthesis of steroid hormones and contraceptives. This technology addresses long-standing inefficiencies in traditional manufacturing by leveraging a streamlined four-step sequence involving esterification, ketalation, reduction, and hydrolysis. Unlike conventional routes that rely on the extraction and oxidative cracking of diosgenin—a process notorious for generating difficult-to-treat wastewater and environmental hazards—this innovative approach utilizes raw materials derived from microbial fermentation of sterols. This shift not only aligns with modern green chemistry principles but also secures a more stable and cost-effective supply chain foundation. By optimizing reaction conditions such as temperature control, solvent ratios, and drying techniques, the method achieves a refined yield of up to 80.79%, significantly outperforming older synthetic strategies that often struggle with yields near 70%. For global procurement and R&D teams, this patent represents a viable pathway to cost reduction in steroid manufacturing while ensuring the high purity required for downstream pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dehydroisoandrosterone has been plagued by significant technical and environmental bottlenecks. Traditional methods typically start with diosgenin, requiring a series of harsh chemical transformations including protection, oxidative cracking, elimination, oximation, Beckmann rearrangement, and hydrolysis. These multi-step processes are inherently cumbersome, leading to cumulative yield losses and generating substantial volumes of hazardous waste that are difficult and expensive to treat. Furthermore, alternative synthetic routes involving oxime formation and Beckmann rearrangement, such as those disclosed in earlier patents, exhibit high sensitivity to temperature fluctuations and require precise control over solvent ratios, often resulting in inconsistent product quality. The reliance on large amounts of solvents like acetic anhydride and acetone in these legacy processes increases both the operational risk and the environmental footprint. Additionally, the purification steps in these conventional methods are often complex, involving multiple crystallizations and washes that further erode overall process efficiency. For supply chain managers, these factors translate into longer lead times, higher production costs, and increased regulatory scrutiny regarding waste disposal and environmental compliance.
The Novel Approach
In stark contrast, the methodology described in CN102603841B offers a streamlined and chemically elegant solution that bypasses the pitfalls of traditional synthesis. The core innovation lies in a carefully orchestrated sequence that converts a readily available starting material (Compound 2) into the target dehydroisoandrosterone (Compound 1) through four distinct, high-yielding transformations. This route eliminates the need for dangerous oxidative cracking and complex rearrangement reactions, replacing them with more predictable and controllable unit operations like esterification and ketal protection. The use of microbial fermentation-derived sterols as the ultimate source of raw materials ensures a renewable and economically advantageous feedstock, decoupling production from the volatility of plant-extracted diosgenin markets. Moreover, the process incorporates specific optimization strategies, such as the use of vacuum drying at controlled temperatures, which dramatically enhances product purity compared to atmospheric drying methods. This technological leap facilitates the commercial scale-up of complex steroid intermediates by simplifying post-treatment procedures and reducing the burden on waste management systems. The result is a manufacturing process that is not only safer and cleaner but also capable of delivering superior economic value through improved yields and operational simplicity.
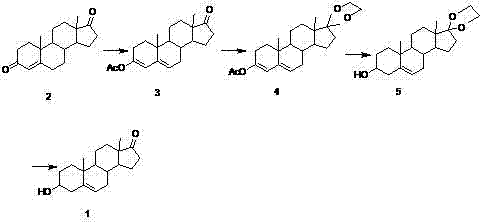
Mechanistic Insights into the Four-Step Synthetic Sequence
The success of this preparation method hinges on the precise execution of four key chemical transformations, each designed to maximize selectivity and minimize byproduct formation. The process initiates with an esterification reaction where Compound 2 reacts with acetic anhydride in the presence of a tosic acid catalyst. This step is critical for activating the molecule for subsequent transformations, and strict temperature control between 20°C and 25°C is mandated to prevent the formation of polarized impurities that could compromise downstream reactions. Following esterification, the synthesis proceeds to a ketalation reaction, where the C17 ketone group is protected using ethylene glycol and a water-removal agent such as triethyl orthoformate. This protection strategy is essential for ensuring chemoselectivity during the subsequent reduction step, preventing unwanted reduction of the C17 position. The rigorous control of moisture content, kept below 0.1% of the total reactive material weight, is a vital parameter here, as excess water can hydrolyze the ketal prematurely or inhibit the reaction equilibrium. The third stage involves a selective reduction using borohydride salts, likely targeting the C3 position to establish the necessary hydroxyl functionality while preserving the protected C17 ketal. Finally, the sequence concludes with an acid-catalyzed hydrolysis that simultaneously removes the acetyl and ketal protecting groups, unveiling the final dehydroisoandrosterone structure. This logical progression of functional group manipulation demonstrates a sophisticated understanding of steroid chemistry, allowing for the efficient construction of the target molecule with minimal side reactions.
Beyond the reaction mechanics, the patent highlights the profound impact of physical processing parameters on the final quality of the active pharmaceutical ingredient. A detailed analysis of drying conditions reveals that vacuum drying at 40°C yields a product with a mass fraction purity of 96.68%, whereas atmospheric drying at the same temperature results in a purity of only 72.71%. This stark difference underscores the sensitivity of the intermediate compounds to thermal degradation or oxidation when exposed to air at elevated temperatures. Similarly, the optimization of solvent quantities during the esterification phase plays a crucial role; increasing the volume of acetic anhydride from 15 ml to 35 ml per gram of substrate improves the content from 92.11% to 97.18%. These findings provide R&D directors with actionable data points for process validation, emphasizing that high-purity dehydroisoandrosterone is not just a function of chemical reagents but also of meticulous engineering controls. By adhering to these specific parameters, manufacturers can consistently achieve the stringent purity specifications required for hormonal therapies, thereby reducing the risk of batch failures and ensuring regulatory compliance.
How to Synthesize Dehydroisoandrosterone Efficiently
The synthesis of dehydroisoandrosterone via this patented route requires careful attention to reaction stoichiometry, temperature profiles, and workup procedures to ensure optimal yield and purity. The process is designed to be robust enough for industrial application while maintaining the precision necessary for pharmaceutical grade output. Operators must strictly follow the specified molar ratios for reagents such as acetic anhydride, ethylene glycol, and borohydride salts, as deviations can lead to incomplete conversions or difficult-to-remove impurities. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process engineers looking to implement this technology.
- Perform esterification of the starting dione compound with acetic anhydride and tosic acid catalyst under nitrogen protection at 20-25°C to form the enol acetate intermediate.
- Execute ketalization using ethylene glycol and a water-removal agent like triethyl orthoformate with tosic acid catalysis to protect the C17 ketone group.
- Conduct selective reduction of the intermediate using borohydride salts (e.g., sodium borohydride) in absolute ethanol to generate the alcohol precursor.
- Finalize the synthesis through acid-catalyzed hydrolysis in a lower boiling water-soluble solvent like acetone to remove protecting groups and yield dehydroisoandrosterone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The shift towards raw materials sourced from microbial fermentation represents a significant departure from the traditional reliance on agricultural extracts like diosgenin, which are subject to seasonal variability and geopolitical supply risks. By securing a supply chain based on fermentation-derived sterols, manufacturers can achieve greater consistency in raw material availability and pricing, effectively insulating their operations from the volatility of the botanical market. Furthermore, the simplified post-treatment processes described in the patent, such as direct filtration and straightforward washing protocols, reduce the consumption of auxiliary materials and labor hours. This operational efficiency translates directly into cost reduction in steroid manufacturing, allowing companies to offer more competitive pricing to their downstream clients without sacrificing margin. The ability to produce high-quality intermediates with fewer processing steps also means faster turnaround times, enabling suppliers to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of hazardous oxidative cracking and complex rearrangement steps significantly lowers the operational expenditure associated with safety measures and specialized equipment. By avoiding the use of expensive and dangerous reagents required for Beckmann rearrangements, the process reduces the overall cost of goods sold. Additionally, the high yield of up to 80.79% after refining minimizes raw material waste, ensuring that a greater proportion of input costs are converted into saleable product. The use of common, inexpensive solvents like acetic anhydride and acetone, coupled with optimized recovery protocols, further enhances the economic viability of the process. These factors collectively drive down the unit cost of production, providing a strong competitive edge in the global marketplace for steroid intermediates.
- Enhanced Supply Chain Reliability: Sourcing raw materials from microbial fermentation provides a stable and scalable foundation for production, independent of agricultural harvest cycles. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the just-in-time delivery expectations of major pharmaceutical customers. The robustness of the chemical route, characterized by easy-to-control reaction conditions and tolerance to minor variations, reduces the likelihood of batch failures that can disrupt supply timelines. Moreover, the simplified purification steps decrease the time required for quality control testing and release, accelerating the flow of goods from the production line to the warehouse. This enhanced reliability strengthens the partnership between suppliers and buyers, fostering long-term contracts and strategic alliances based on trust and consistent performance.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that can be easily translated from laboratory benchtops to multi-ton industrial reactors. The avoidance of heavy metal catalysts and toxic reagents simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The reduction in wastewater toxicity and volume lowers the cost of environmental management and mitigates the risk of regulatory fines or shutdowns. Furthermore, the energy-efficient nature of the process, particularly the use of moderate temperatures and vacuum drying, aligns with corporate sustainability goals and carbon reduction initiatives. This environmental stewardship not only protects the company's reputation but also appeals to eco-conscious consumers and investors who prioritize green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial implications of this dehydroisoandrosterone synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating the technology for potential adoption or procurement. Understanding these nuances is essential for making informed decisions about integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis route over traditional diosgenin methods?
A: This novel route avoids the lengthy steps, difficult wastewater treatment, and environmental pollution associated with traditional diosgenin oxidation and cracking. It utilizes microbial fermentation-derived raw materials which are cheaper and more environmentally friendly.
Q: How does the new method improve yield compared to oxime-based synthesis?
A: Traditional oxime and Beckmann rearrangement methods often suffer from yields around 70% and sensitive temperature controls. This patented method achieves a refined yield of up to 80.79% through optimized reaction conditions and simplified post-treatment processes.
Q: What specific process controls are critical for maintaining high purity?
A: Critical controls include maintaining esterification temperatures between 20-25°C to prevent polarization impurities, ensuring moisture content is below 0.1% during ketalization, and utilizing vacuum drying at 40°C instead of atmospheric drying to achieve purity levels exceeding 96%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroisoandrosterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in CN102603841B can be seamlessly transitioned from pilot scale to full industrial output. We are committed to delivering stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex steroid intermediates allows us to offer tailored solutions that meet the specific needs of R&D directors and procurement managers alike, bridging the gap between laboratory discovery and commercial reality.
We invite you to explore how our expertise can optimize your supply chain and reduce your overall manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic potential of switching to this novel synthesis route for your specific application. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make a confident sourcing decision. Let us help you secure a reliable supply of high-quality dehydroisoandrosterone that drives your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
