Advanced Manufacturing of Chiral Bisphosphine Ligands via Safe Grignard Coupling Routes
Advanced Manufacturing of Chiral Bisphosphine Ligands via Safe Grignard Coupling Routes
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for safer, more scalable synthetic routes for high-value chiral ligands. Patent CN114835749B introduces a groundbreaking methodology for the preparation of bisphosphine ligands, specifically targeting widely used structures such as DuPhos and BPE derivatives. This innovation addresses critical bottlenecks in the supply chain of chiral catalysts by replacing hazardous phosphorus-hydrogen intermediates with robust Grignard-based coupling strategies. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this represents a pivotal shift towards more reliable bisphosphine ligand supplier capabilities. The process leverages readily available dicarbonyl precursors and standard organometallic reagents to construct the complex P-C-P backbone under mild conditions, ensuring that the production of these essential catalytic components can be scaled from laboratory grams to multi-ton commercial quantities without compromising safety or purity standards.
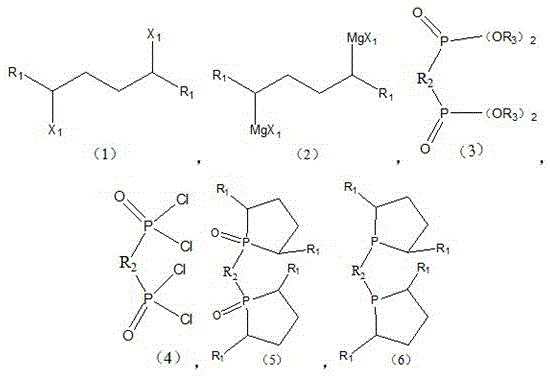
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral diphosphine ligands has been plagued by significant operational hazards and process complexities that hinder large-scale adoption. Traditional routes, as illustrated in the background art of the patent, frequently rely on the generation of phosphorus-hydrogen (P-H) intermediates. These species are notoriously unstable, exhibiting extreme sensitivity to atmospheric moisture and oxygen, which necessitates rigorous anhydrous conditions and specialized handling equipment. Furthermore, P-H compounds often possess potent, unpleasant odors and present high safety risks due to their potential pyrophoric nature, making them unsuitable for standard industrial reactor setups. The requirement to synthesize these intermediates adds multiple steps to the workflow, increasing the overall cost reduction in chiral catalyst manufacturing challenges. Additionally, the harsh reaction conditions often required to couple these fragile intermediates with cyclic backbones can lead to racemization or decomposition, resulting in lower yields and difficult purification processes that strain supply chain continuity.
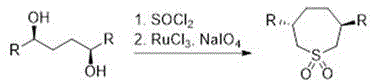
The Novel Approach
In stark contrast, the methodology disclosed in CN114835749B circumvents these dangers by employing a Grignard coupling strategy that is both chemically elegant and industrially robust. Instead of relying on volatile P-H species, the process utilizes stable organomagnesium reagents derived from simple dihaloalkanes. This fundamental shift allows the construction of the phosphine framework through nucleophilic substitution on phosphorus chlorides, a reaction type that is well-understood and easily controlled in large-scale reactors. The new approach operates under mild process conditions, typically avoiding extreme temperatures or pressures that degrade equipment or compromise product quality. By eliminating the need for hazardous P-H intermediates, the process not only enhances worker safety but also simplifies the waste stream management, contributing to a greener manufacturing profile. This streamlined pathway facilitates the commercial scale-up of complex phosphine ligands, ensuring a consistent supply of high-purity materials for asymmetric hydrogenation applications in drug synthesis.
Mechanistic Insights into Grignard-Mediated Phosphine Construction
The core of this innovative synthesis lies in the precise orchestration of organometallic transformations, beginning with the formation of the Grignard reagent (Compound 2). This step involves reacting a dihaloalkane precursor (Compound 1) with magnesium metal in an etheral solvent like tetrahydrofuran at moderate temperatures between 30°C and 40°C. The use of iodine as a catalyst ensures rapid initiation of the magnesium surface, promoting high conversion rates often exceeding 90%. Following this, the phosphorus component is activated by reacting a phosphonate or phosphine oxide precursor (Compound 3) with phosphorus pentachloride at elevated temperatures of 100°C to 120°C. This substitution generates the highly electrophilic dichlorophosphine species (Compound 4), which serves as the coupling partner. The subsequent reaction between the nucleophilic Grignard reagent and the electrophilic phosphorus chloride is the critical bond-forming event. Conducted at low temperatures ranging from -78°C to 25°C, this step minimizes side reactions such as Wurtz coupling or over-alkylation, ensuring the structural integrity of the cyclic phosphine oxide backbone.
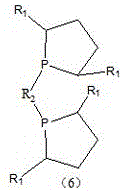
Impurity control is rigorously managed through a series of purification and resolution steps designed to meet the stringent purity specifications required for pharmaceutical catalysis. After the coupling reaction, the resulting phosphine oxide racemate (Compound 5) undergoes recrystallization in organic alcohols to remove inorganic salts and unreacted starting materials. To achieve the necessary optical purity, the racemate is subjected to optical resolution using chiral resolving agents such as L-(-)-dibenzoyltartaric acid. This process separates the enantiomers based on the solubility differences of their diastereomeric salts, allowing for the isolation of the desired R-configuration with enantiomeric excess values often surpassing 99%. The final reduction of the phosphine oxide to the active phosphine ligand (Compound 6) is achieved using a silane-based reducing system, such as polymethoxysilane with titanium tetraisopropoxide. This mild reduction protocol avoids the use of aggressive reagents like trichlorosilane alone, further enhancing the safety profile and simplifying the workup procedure to yield the final high-purity bisphosphine ligand.
How to Synthesize Bisphosphine Ligands Efficiently
The synthesis of these high-value chiral ligands requires precise control over reaction parameters to maximize yield and enantioselectivity. The patented process outlines a clear sequence starting from commodity chemicals, moving through Grignard formation, phosphorus activation, coupling, resolution, and final reduction. Each stage is optimized for scalability, utilizing solvents and reagents that are compatible with standard stainless steel reactor infrastructure. The detailed standardized synthesis steps below provide a roadmap for replicating this efficient production method in a GMP-compliant environment.
- Perform a Grignard reaction on compound (1) with magnesium metal at 30-40°C to generate the organomagnesium intermediate compound (2).
- React compound (3) with phosphorus pentachloride at 100-120°C to synthesize the reactive phosphorus chloride species, compound (4).
- Execute a second substitution reaction between compound (2) and compound (4) at temperatures ranging from -78°C to 25°C to form the oxide precursor compound (5).
- Conduct a reduction reaction on compound (5) using a silane reducing agent system to yield the final diphosphine ligand of formula (6).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous P-H intermediates removes a major bottleneck in the supply chain, as these materials often require specialized vendors and incur high logistics costs due to their dangerous goods classification. By shifting to stable Grignard reagents and phosphorus chlorides, manufacturers can source raw materials from a broader base of commodity chemical suppliers, significantly enhancing supply chain reliability and reducing the risk of production stoppages. Furthermore, the simplified workup procedures, which avoid complex distillation of sensitive intermediates, lead to drastically simplified processing times and lower energy consumption. This efficiency translates directly into cost optimization, allowing for more competitive pricing models for downstream users in the API and fine chemical sectors without sacrificing the quality of the final catalytic product.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the replacement of expensive and difficult-to-handle P-H precursors with inexpensive, bulk-available starting materials such as dicarbonyl compounds and phosphorus pentachloride. The avoidance of specialized containment systems required for pyrophoric substances reduces capital expenditure on reactor modifications and safety infrastructure. Additionally, the high yields reported in the patent examples, often exceeding 80-90% for key steps, minimize material waste and improve the overall atom economy of the synthesis. This efficiency ensures that the cost per kilogram of the final ligand is significantly lowered, providing a compelling value proposition for large-volume consumers of chiral catalysts.
- Enhanced Supply Chain Reliability: Supply continuity is critically improved by the robustness of the reaction conditions, which tolerate minor variations in temperature and mixing better than the fragile traditional routes. The use of common solvents like tetrahydrofuran and ethanol, along with standard reagents like magnesium and sodium borohydride, ensures that raw material availability is not a constraint even during global supply disruptions. The process design inherently supports continuous manufacturing or large-batch processing, enabling producers to maintain substantial inventory levels of key intermediates. This stability allows supply chain planners to forecast lead times with greater accuracy, reducing the need for emergency air freight and ensuring that production schedules for downstream asymmetric hydrogenation processes remain uninterrupted.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with modern green chemistry principles by minimizing the generation of toxic waste streams. The absence of malodorous phosphines and the use of aqueous workups for salt removal simplify wastewater treatment protocols, reducing the burden on environmental compliance teams. The mild reaction temperatures and pressures facilitate easier scale-up from pilot plants to multi-ton commercial production facilities without the need for exotic high-pressure equipment. This scalability ensures that the technology can meet the growing global demand for chiral intermediates in the pharmaceutical and agrochemical industries while maintaining a low environmental footprint and adhering to strict international safety regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bisphosphine ligand synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the operational realities of this advanced manufacturing route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing production lines.
Q: Why is this new process safer than traditional bisphosphine ligand synthesis?
A: Traditional methods often require the preparation and handling of phosphorus-hydrogen (P-H) intermediates, which are highly sensitive to moisture, possess unpleasant odors, and carry significant safety risks including pyrophoricity. This patented process eliminates the need for P-H intermediates entirely by utilizing stable Grignard reagents and phosphorus chlorides, significantly enhancing operational safety.
Q: What are the typical reaction conditions for the key coupling step?
A: The critical coupling step involves reacting the Grignard reagent (compound 2) with the phosphorus chloride derivative (compound 4). This second substitution reaction is conducted under mild conditions, typically ranging from -78°C to 25°C, which allows for precise control over stereochemistry and minimizes side reactions compared to harsher traditional protocols.
Q: How is optical purity achieved in the final ligand product?
A: Optical purity is secured through a resolution process applied to the racemate of compound (5). By using resolving agents such as L-(-)-dibenzoyltartaric acid, the specific R-configuration is isolated with high enantiomeric excess (ee values >99% reported in examples) before the final reduction step ensures the integrity of the chiral phosphine structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the synthesis of next-generation therapeutics and agrochemicals. Our technical team has extensively analyzed the pathways described in CN114835749B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are committed to delivering high-purity bisphosphine ligands that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch performs consistently in your asymmetric hydrogenation reactions. Our facility is equipped to handle the specific solvent systems and temperature controls necessary for the Grignard and resolution steps, guaranteeing a supply of catalyst components that drive efficiency in your own manufacturing processes.
We invite you to collaborate with us to optimize your catalytic processes and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can secure your supply chain and enhance your competitive edge in the global market.
