Advanced Synthesis of 4-Tetrazolyl-Benzoxazine Derivatives for Pharmaceutical Applications
Advanced Synthesis of 4-Tetrazolyl-Benzoxazine Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer enhanced biological activity and improved pharmacokinetic profiles. Patent CN111138419B introduces a groundbreaking methodology for the synthesis of 4-tetrazolyl-substituted-benzoxazine derivatives, a class of compounds with significant potential in drug discovery. These molecules combine the pharmacophore of the benzoxazine ring, known for its presence in various bioactive natural products and synthetic drugs, with the metabolic stability and hydrogen-bonding capabilities of the tetrazole moiety. The disclosed technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, efficient one-pot process. By leveraging a tandem Passerini-type multicomponent reaction followed by a palladium-catalyzed cyclization, this method addresses critical pain points in medicinal chemistry, such as step economy and functional group tolerance. For R&D teams exploring new chemical space, access to such versatile intermediates is invaluable.
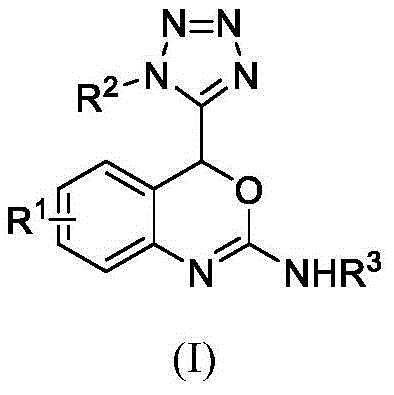
As a reliable pharmaceutical intermediate supplier, understanding the structural versatility of these compounds is key. The general structure, as depicted in the patent, allows for extensive diversification at the R1, R2, and R3 positions. This modularity enables the rapid generation of compound libraries for structure-activity relationship (SAR) studies. The ability to introduce diverse substituents such as halogens, alkyl groups, and aryl moieties without compromising the integrity of the sensitive tetrazole or oxazine rings underscores the robustness of this synthetic strategy. Consequently, this technology not only facilitates the discovery of new therapeutic agents but also provides a scalable pathway for the cost reduction in API manufacturing where such complex heterocycles are required.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzoxazine core has relied on methodologies that pose significant challenges for both laboratory synthesis and industrial production. Representative conventional methods often involve the cyclization of o-aminobenzyl alcohol with aldehydes under the influence of strong oxidants such as sodium hypochlorite (NaClO). The use of such aggressive oxidizing agents necessitates stringent safety protocols and generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs. Furthermore, alternative routes utilizing 2-aminobenzonitrile and ketones typically require high-temperature reflux conditions in polar aprotic solvents like DMF, catalyzed by Lewis acids such as zinc chloride. These harsh thermal conditions can lead to the decomposition of sensitive functional groups, limiting the substrate scope and resulting in lower overall yields. The combination of high energy consumption, hazardous reagents, and poor atom economy renders these traditional approaches suboptimal for the modern demands of green chemistry and sustainable manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN111138419B offers a sophisticated solution that circumvents these limitations through a clever design of reaction cascades. The novel approach utilizes a one-pot, two-step sequence that begins with the formation of a key azide intermediate via a multicomponent reaction at room temperature. This mild initial step ensures that sensitive functionalities remain intact. Subsequently, the reaction mixture is subjected to a palladium-catalyzed transformation at a moderate temperature of 60°C to effect cyclization. This transition metal-catalyzed step is highly efficient, enabling the simultaneous formation of the tetrazole and benzoxazine rings with impressive regioselectivity. The entire process eliminates the need for strong oxidants and extreme temperatures, thereby enhancing the safety profile and operational simplicity. For procurement managers, this translates to a more reliable supply chain with reduced risk of batch failures due to harsh reaction conditions.
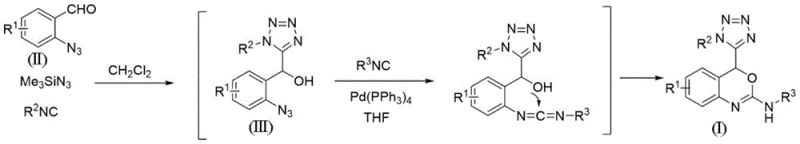
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The success of this synthesis hinges on the intricate interplay between the Passerini-type multicomponent reaction and the subsequent palladium-catalyzed cascade. In the first stage, o-azidobenzaldehyde reacts with trimethylsilyl azide and an isocyanide (R2NC) in dichloromethane. This step effectively constructs the alpha-hydroxy tetrazole motif, generating the critical intermediate (Formula III) which possesses both an ortho-azido group on the aromatic ring and a hydroxyl group on the side chain. The formation of the tetrazole ring in this manner is highly advantageous as it avoids the use of explosive hydrazoic acid, utilizing the safer trimethylsilyl azide instead. The resulting intermediate is chemically poised for the subsequent cyclization, with the azide and hydroxyl groups positioned perfectly for intramolecular interaction.
The second stage involves the addition of a second isocyanide (R3NC) and a catalytic amount of tetrakis(triphenylphosphine)palladium(0) in tetrahydrofuran. The palladium catalyst likely facilitates the activation of the ortho-azide group, potentially generating a reactive nitrene or imino-palladium species. This activated species then undergoes insertion of the second isocyanide, followed by nucleophilic attack by the proximal hydroxyl group to close the oxazine ring. This mechanistic pathway is exceptionally clean, minimizing the formation of by-products that typically plague complex heterocycle synthesis. From an impurity control perspective, the specificity of the palladium catalyst ensures that the reaction proceeds predominantly towards the desired 4-tetrazolyl-benzoxazine scaffold. This high level of selectivity simplifies downstream purification, allowing for the isolation of high-purity pharmaceutical intermediates with minimal effort, which is a critical factor for regulatory compliance in drug substance manufacturing.
How to Synthesize 4-Tetrazolyl-Benzoxazine Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible for both process research and production environments. The protocol begins by combining the aldehyde, silyl azide, and the first isocyanide in a simple solvent system, allowing the reaction to proceed to completion as monitored by thin-layer chromatography. Without the need for isolating the intermediate, the solvent is swapped or adjusted, and the catalytic system is introduced. This telescoping of steps significantly reduces material loss and processing time. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to guide technical teams in replicating these results effectively.
- Dissolve o-azidobenzaldehyde, trimethylsilyl azide, and isonitrile I in dichloromethane at room temperature to form the azide intermediate.
- Without isolation, switch solvent to THF, add Pd(PPh3)4 and isonitrile II, and heat to 60°C to induce cyclization.
- Remove solvent under reduced pressure and purify the crude residue via column chromatography to obtain the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this synthetic route offers compelling economic and logistical benefits. The shift from hazardous, high-energy processes to a mild, catalytic one fundamentally alters the cost structure of producing these valuable intermediates. By eliminating the need for expensive and dangerous oxidizing agents, the process reduces the burden on safety infrastructure and waste management systems. Furthermore, the use of commercially available starting materials such as o-azidobenzaldehyde and various isocyanides ensures that the supply chain remains robust and less susceptible to raw material shortages. The simplicity of the workup, primarily involving solvent removal and standard chromatography, means that production cycles can be shortened, enhancing overall throughput.
- Cost Reduction in Manufacturing: The elimination of strong oxidants and high-temperature reflux conditions leads to significant savings in energy consumption and specialized equipment maintenance. Additionally, the high yields reported in the patent, reaching up to 84% for certain derivatives, maximize the utilization of raw materials, directly lowering the cost of goods sold. The avoidance of complex protection-deprotection sequences further streamlines the process, reducing labor hours and solvent usage, which are major cost drivers in fine chemical synthesis.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like trimethylsilyl azide and common isocyanides mitigates the risks associated with sourcing unstable or highly regulated precursors. The mild reaction conditions also reduce the likelihood of runaway reactions or thermal incidents, ensuring consistent batch-to-batch quality and uninterrupted production schedules. This reliability is crucial for maintaining the continuity of supply for downstream drug development projects that depend on these intermediates.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes the number of unit operations, making the transition from gram-scale laboratory synthesis to kilogram or ton-scale production more straightforward. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, facilitating easier permitting and operation of manufacturing facilities. This green chemistry approach not only protects the environment but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel benzoxazine derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees.
Q: What are the advantages of this synthesis method over conventional benzoxazine production?
A: Unlike conventional methods requiring strong oxidants like NaClO or high-temperature reflux with ZnCl2, this patented route operates under mild conditions (room temperature to 60°C) and avoids harsh reagents, significantly improving safety and scalability.
Q: What is the typical yield range for these derivatives?
A: The patent data demonstrates robust yields ranging from 59% to 84% across various substituents, with specific examples like the tert-butyl derivative achieving an 84% isolated yield, indicating high efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the one-pot two-step design minimizes unit operations and solvent exchanges. The use of readily available starting materials and standard purification techniques like column chromatography supports feasible commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Tetrazolyl-Benzoxazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN111138419B for advancing pharmaceutical pipelines. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next-generation therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the delivery of life-saving medicines to patients worldwide.
