Advanced Industrial Synthesis of 3,3',6,6'-Tetramethoxy-2,2'-Binaphthyl for High-Performance OLED Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands increasingly sophisticated intermediates capable of delivering high fluorescence quantum yields and narrow emission spectra. Patent CN115521193A, published in late 2022, introduces a groundbreaking industrial preparation method for 3,3',6,6'-tetramethoxy-2,2'-binaphthyl, a critical precursor for dinaphtho-furan based blue light materials. This technical disclosure addresses long-standing inefficiencies in oxidative coupling reactions by replacing traditional iron-based catalysts with a specialized copper-lithium reagent system. For R&D directors and process chemists, this represents a significant leap forward in controlling regioselectivity and minimizing oligomerization side reactions. The patent outlines a robust pathway that not only enhances reaction conversion but also streamlines the downstream purification process, which is often the bottleneck in producing high-purity electronic chemicals. By shifting the mechanistic paradigm from iron-mediated to copper-lithium mediated coupling, the inventors have successfully tackled the issues of low yield and complex impurity profiles that have plagued previous synthetic attempts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO2018235953A1, typically rely on the use of n-butyllithium for lithiation followed by oxidative coupling using iron acetylacetonate (Fe(acac)3). While conceptually straightforward, this conventional approach suffers from severe practical drawbacks when scaled for industrial application. The primary issue lies in the poor selectivity of the iron catalyst, which frequently promotes the formation of undesirable trimer and tetramer by-products alongside the desired dimer. Furthermore, the solubility characteristics of iron salts in organic solvents complicate the workup procedure, often requiring extensive chromatography or multiple recrystallizations to achieve pharmaceutical or electronic grade purity. The generation of substantial solid waste containing heavy metals also poses significant environmental compliance challenges and increases the overall cost of goods sold. Consequently, the isolated yields in these legacy processes are often dismally low, sometimes ranging between 9% and 12% after rigorous purification, making them economically unviable for mass production.
The Novel Approach
In stark contrast, the methodology presented in CN115521193A employs a pre-formed copper-lithium reagent generated from 2,2,6,6-tetramethylpiperidinium lithium and cuprous cyanide. This specific reagent combination facilitates a much cleaner lithiation of the 2,7-dimethoxynaphthalene substrate. The subsequent oxidative coupling is driven by nitrobenzene rather than an iron salt, which fundamentally alters the reaction landscape to favor dimerization over oligomerization. 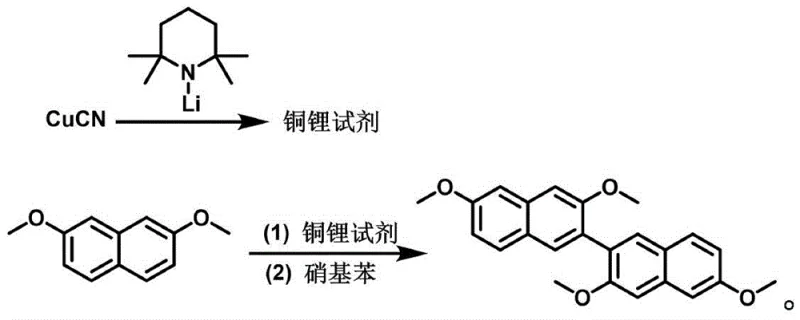 This strategic substitution results in a dramatic improvement in crude product quality, significantly reducing the burden on purification units. The process operates under controlled cryogenic conditions, initially forming the reagent at temperatures between -80°C and -70°C, before allowing the coupling to proceed upon warming. The outcome is a streamlined workflow that avoids the heavy metal sludge associated with iron catalysts, thereby offering a greener and more cost-effective solution for manufacturing high-value OLED intermediates.
This strategic substitution results in a dramatic improvement in crude product quality, significantly reducing the burden on purification units. The process operates under controlled cryogenic conditions, initially forming the reagent at temperatures between -80°C and -70°C, before allowing the coupling to proceed upon warming. The outcome is a streamlined workflow that avoids the heavy metal sludge associated with iron catalysts, thereby offering a greener and more cost-effective solution for manufacturing high-value OLED intermediates.
Mechanistic Insights into Copper-Lithium Mediated Oxidative Coupling
The core innovation of this patent lies in the precise generation and utilization of the copper-lithium species. The reaction begins with the deprotonation capabilities of the bulky base 2,2,6,6-tetramethylpiperidinium lithium (LiTMP), which selectively abstracts a proton from the 2,7-dimethoxynaphthalene substrate. However, unlike simple organolithium species which can be unstable or overly reactive leading to side reactions, the presence of cuprous cyanide (CuCN) stabilizes the intermediate through transmetallation. This forms a higher-order cuprate-like species that exhibits superior nucleophilicity and stability at low temperatures. The molar ratios specified in the patent, particularly the excess of LiTMP relative to the substrate (1:4 to 1:8), ensure complete conversion of the starting material while maintaining the integrity of the sensitive organometallic intermediate. This careful stoichiometric balance is crucial for preventing incomplete lithiation which could lead to recovered starting material or mixed coupling products.
Following the formation of the organocopper intermediate, the addition of nitrobenzene serves as the oxidant to trigger the homocoupling event. Nitrobenzene acts as a mild yet effective single-electron oxidant in this context, facilitating the radical-radical coupling or reductive elimination necessary to form the biaryl bond. This mechanism effectively suppresses the formation of higher-order oligomers (trimers and tetramers) that are prevalent in iron-catalyzed systems. The transition from -10°C during the lithiation phase to 0°C during the coupling phase allows for controlled kinetics, ensuring that the coupling occurs selectively at the desired positions on the naphthalene ring. The result is a product profile with significantly fewer impurities, allowing for high-purity isolation (>98% LC content) through simple recrystallization techniques rather than complex chromatographic separations.
How to Synthesize 3,3',6,6'-Tetramethoxy-2,2'-Binaphthyl Efficiently
The synthesis protocol described in the patent offers a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and reagent quality. The process initiates with the preparation of the copper-lithium reagent in tetrahydrofuran (THF), requiring strict adherence to cryogenic conditions to prevent reagent decomposition. Once the reagent is formed, the substrate is introduced, and the mixture is allowed to react for a defined period to ensure full lithiation. The subsequent addition of the coupling agent must be managed carefully to control the exotherm and reaction rate.
- Preparation of Copper-Lithium Reagent: React 2,2,6,6-tetramethylpiperidinium lithium with cuprous cyanide in THF at -80°C to -70°C.
- Lithiation Reaction: Add 2,7-dimethoxynaphthalene to the reagent mixture and react at -10°C for 3 hours.
- Oxidative Coupling & Workup: Add nitrobenzene at -80°C, warm to 0°C, then quench, concentrate, and recrystallize from toluene-ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route translates directly into tangible operational efficiencies and risk mitigation. The most immediate impact is observed in the drastic reduction of raw material waste. By achieving significantly higher yields compared to legacy iron-catalyzed methods, manufacturers can produce the same quantity of final product with substantially less input of expensive starting materials like 2,7-dimethoxynaphthalene and specialized bases. This efficiency gain lowers the variable cost per kilogram of the intermediate, providing a competitive edge in pricing negotiations with downstream OLED panel manufacturers. Furthermore, the elimination of iron-based catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which often require specialized resins or complex aqueous workups that delay batch release times.
- Cost Reduction in Manufacturing: The shift to a copper-lithium reagent system eliminates the reliance on iron acetylacetonate, which is known to generate difficult-to-remove solid waste. This change simplifies the downstream processing requirements, removing the need for extensive filtration of metal sludge and reducing the consumption of solvents required for purification. Consequently, the overall operational expenditure (OPEX) is lowered due to reduced utility usage and shorter cycle times per batch. The higher selectivity of the reaction also means that less material is lost to by-product formation, maximizing the return on investment for every kilogram of raw material purchased.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as cuprous cyanide and nitrobenzene, are commodity chemicals with stable global supply chains, unlike some specialized transition metal catalysts that may face sourcing bottlenecks. The robustness of the reaction conditions, specifically the tolerance for standard industrial solvents like THF and toluene, ensures that the process can be easily transferred between different manufacturing sites without significant re-validation. This flexibility enhances supply continuity, ensuring that critical OLED material intermediates can be delivered consistently to meet the tight production schedules of display manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the avoidance of heavy metal waste streams significantly reduces the regulatory burden associated with waste disposal. The process generates primarily organic waste that can be treated through standard incineration or recovery methods, rather than hazardous metal-contaminated sludge requiring specialized handling. This alignment with green chemistry principles not only lowers disposal costs but also future-proofs the manufacturing process against tightening environmental regulations. The simplified workup involving water washing and recrystallization is inherently scalable, allowing for seamless transition from pilot plant to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and process advantages. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: How does the copper-lithium method improve yield compared to iron-catalyzed routes?
A: The novel method utilizing a copper-lithium reagent and nitrobenzene oxidative coupling significantly suppresses the formation of trimer and tetramer by-products common in iron-catalyzed processes, thereby increasing isolated yields from approximately 10% to over 50%.
Q: What are the environmental benefits of this synthesis route?
A: By eliminating the use of iron acetylacetonate (Fe(acac)3), the process avoids the generation of large amounts of heavy metal-containing solid waste, simplifying wastewater treatment and reducing hazardous waste disposal costs.
Q: Is this process suitable for large-scale manufacturing of OLED intermediates?
A: Yes, the protocol utilizes standard industrial solvents like THF and toluene, and the simplified workup involving water washing and recrystallization makes it highly scalable for commercial production of electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',6,6'-Tetramethoxy-2,2'-Binaphthyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the performance of next-generation OLED displays. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115521193A are fully realized in a manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for electronic applications. Our commitment to quality assurance ensures that the impurity profiles, particularly regarding trimer and tetramer content, are kept well below acceptable thresholds.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this copper-lithium mediated process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a secure and efficient supply of this vital OLED material intermediate.
