Scalable Synthesis of Tetrahydropyrano Pyrazole Carboxylic Acid for Pharmaceutical Intermediates
Scalable Synthesis of Tetrahydropyrano Pyrazole Carboxylic Acid for Pharmaceutical Intermediates
The development of robust synthetic routes for heterocyclic scaffolds is critical for the uninterrupted supply of modern therapeutics, particularly kinase inhibitors and CFTR modulators. Patent CN108546266B introduces a transformative methodology for the production of 1,4,6,7-tetrahydropyrano[4,3-c]pyrazole-3-carboxylic acid, a pivotal building block in medicinal chemistry. This technical disclosure addresses the longstanding challenges of safety, yield, and scalability that have plagued previous generations of synthesis. By shifting away from hazardous diazo compounds and unstable acid chlorides, this new protocol leverages lithium bis(trimethylsilyl)amide (LiHMDS) mediated condensation to achieve a total yield of 65% with exceptional purity profiles reaching 99%. For procurement and R&D leaders, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials without the baggage of dangerous processing conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydropyrano-pyrazole core has been fraught with peril and inefficiency. One prominent legacy route relies on the reaction of tetrahydro-2H-pyran-3-ketone with ethyl diazoacetate under pyrrolidine catalysis. While literature suggests yields between 50-80% on a small laboratory scale, this method collapses catastrophically upon scale-up. As illustrated in the reaction scheme below, the use of ethyl diazoacetate introduces an unacceptable explosion hazard. Experimental data confirms that scaling this reaction from 10 grams to merely 50 grams results in violent flushing and complete material degradation, rendering it commercially non-viable for any serious manufacturing operation.
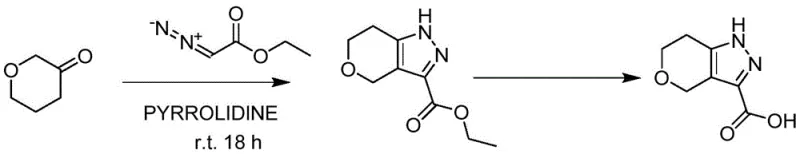
Furthermore, alternative pathways utilizing 3-bromotetrahydro-4H-pyran-4-one or oxalyl chloride monoethyl ester have failed to gain traction due to economic and technical barriers. The bromo-ketone route generates difficult-to-separate isomers during the cyclization step, drastically reducing the isolated yield. Similarly, the route employing oxalyl chloride monoethyl ester suffers from a dismal first-step yield of only 19%, compounded by the fact that the acid chloride reagent is hygroscopic and difficult to store. These factors collectively inflate the cost of goods sold (COGS) and introduce supply chain volatility, making these legacy methods unsuitable for the cost reduction in API manufacturing demanded by today's market.
The Novel Approach
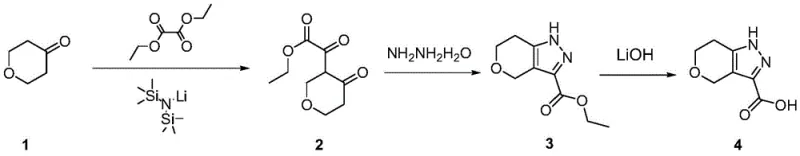
The inventive method disclosed in CN108546266B circumvents these pitfalls by employing a mild, three-step sequence starting from commodity chemicals. The process initiates with the condensation of tetrahydropyranone and diethyl oxalate, catalyzed by LiHMDS at cryogenic temperatures (-70 to -80°C), followed by a controlled warm-up. This is succeeded by a ring-closing reaction with hydrazine hydrate and a final hydrolysis step. Unlike the dangerous diazo route, this pathway utilizes stable, non-explosive reagents that are readily available in bulk quantities. The reaction conditions are sufficiently mild to prevent thermal runaway, and the workup procedures avoid complex chromatography, relying instead on simple extractions and pulping. This strategic shift not only enhances operator safety but also ensures consistent reproducibility from gram to kilogram scales, establishing a new standard for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into LiHMDS-Catalyzed Condensation and Cyclization
The success of this synthetic strategy hinges on the precise control of enolate chemistry using lithium bis(trimethylsilyl)amide (LiHMDS). In the first step, LiHMDS acts as a strong, non-nucleophilic base to deprotonate the alpha-position of tetrahydropyranone, generating a kinetic enolate. This enolate then performs a nucleophilic attack on diethyl oxalate. The low temperature requirement of -70 to -80°C is critical here; it suppresses side reactions such as self-condensation of the ketone or over-alkylation, ensuring high regioselectivity for the desired 2-oxo-2-(4-oxo-tetrahydro-2H-pyran-3-yl) ethyl acetate intermediate. Following the reaction, a careful quench with water and pH adjustment to 2-3 allows for the isolation of the crude keto-ester as a reddish-brown liquid, which is sufficiently pure for direct use in the subsequent step without further purification.
The subsequent cyclization mechanism involves the reaction of the 1,3-dicarbonyl system within the intermediate with hydrazine hydrate. In the presence of glacial acetic acid, hydrazine attacks the more electrophilic ketone carbonyl adjacent to the ester group, initiating a condensation cascade that forms the pyrazole ring. The reaction is conducted at a moderate 20-30°C overnight, allowing for complete conversion while minimizing the formation of regioisomers that plagued the bromo-ketone route. Finally, the saponification of the ethyl ester using lithium hydroxide at 40-60°C proceeds cleanly to yield the target carboxylic acid. The choice of LiOH over NaOH or KOH often facilitates easier filtration of the final lithium salt or the free acid upon acidification, contributing to the reported 99% UPLC purity without the need for recrystallization from toxic solvents.
How to Synthesize 1,4,6,7-Tetrahydropyrano[4,3-c]pyrazole-3-carboxylic Acid Efficiently
Implementing this synthesis requires strict adherence to temperature controls and stoichiometry to maximize the 65% overall yield. The process is designed for operational simplicity, avoiding the need for specialized pressure vessels or cryogenic equipment beyond standard low-temperature reactors. The following guide outlines the critical operational parameters derived from the patent examples, focusing on the transition from laboratory discovery to pilot-scale production. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- React tetrahydropyranone with diethyl oxalate using lithium bis(trimethylsilyl)amide (LiHMDS) at -70 to -80°C to form the keto-ester intermediate.
- Cyclize the intermediate with hydrazine hydrate in glacial acetic acid at 20-30°C to generate the ethyl ester pyrazole derivative.
- Hydrolyze the ethyl ester using lithium hydroxide in ethanol at 40-60°C, followed by acidification to isolate the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this novel synthetic route offers tangible benefits that extend far beyond simple yield metrics. By replacing hazardous and expensive starting materials with stable commodity chemicals, the process fundamentally alters the risk profile and cost structure of the supply chain. The elimination of ethyl diazoacetate removes the need for specialized explosion-proof facilities and rigorous safety monitoring associated with energetic materials, thereby lowering facility overheads. Furthermore, the stability of diethyl oxalate and tetrahydropyranone ensures consistent raw material availability, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production stoppages due to reagent degradation.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by both raw material costs and processing efficiency. By substituting the expensive 3-bromotetrahydro-4H-pyran-4-one and the unstable oxalyl chloride monoethyl ester with inexpensive tetrahydropyranone and diethyl oxalate, the Bill of Materials (BOM) cost is significantly reduced. Additionally, the avoidance of column chromatography in favor of simple pulping and filtration drastically cuts down on solvent consumption and silica gel waste, leading to substantial cost savings in waste disposal and solvent recovery operations.
- Enhanced Supply Chain Reliability: The robustness of the LiHMDS-mediated reaction ensures high reproducibility across different batch sizes, a critical factor for maintaining supply continuity. Unlike the legacy diazo route which failed at the 50g scale, this method has been demonstrated to perform consistently on multi-hundred gram scales (e.g., 500g of starting ketone), indicating a clear path to tonnage production. This scalability assures downstream partners that the supply of this critical kinase inhibitor intermediate will remain uninterrupted, supporting long-term project planning and inventory management.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns with green chemistry principles by minimizing the use of hazardous reagents and simplifying waste streams. The absence of heavy metal catalysts and explosive precursors simplifies the regulatory compliance burden for manufacturing sites. The straightforward workup procedures reduce the volume of organic waste generated per kilogram of product, facilitating easier adherence to increasingly stringent environmental regulations regarding solvent emissions and chemical waste disposal in pharmaceutical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 1,4,6,7-tetrahydropyrano[4,3-c]pyrazole-3-carboxylic acid. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, aiming to clarify the operational benefits and chemical rationale behind this improved methodology.
Q: Why is the conventional ethyl diazoacetate route considered unsafe for scale-up?
A: The conventional route utilizes ethyl diazoacetate, which is explosive and prone to thermal runaway. Patent data indicates that while feasible on a 10g scale, the reaction causes material flushing and complete degradation when scaled to 50g, posing severe safety hazards.
Q: What is the overall yield improvement of the new LiHMDS method compared to prior art?
A: The new method achieves a total yield of 65% with 99% purity. This is a substantial improvement over the oxalyl chloride monoethyl ester route, which suffered from a mere 19% yield in the first step due to reagent instability.
Q: Does this synthesis require complex purification like column chromatography?
A: No. A key advantage of this process is the elimination of column chromatography. The intermediates are purified via simple extraction and pulping with petroleum ether, making it highly suitable for industrial cost reduction and waste minimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,6,7-Tetrahydropyrano[4,3-c]pyrazole-3-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels described in CN108546266B can be reliably replicated in our facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1,4,6,7-tetrahydropyrano[4,3-c]pyrazole-3-carboxylic acid meets the exacting standards required for GMP pharmaceutical applications.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for your drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, quality, and economic efficiency.
