Optimizing Meglumine Clofenazate Production: A High-Purity Scalable Route for Global API Supply Chains
Introduction to Advanced Meglumine Clofenazate Manufacturing
The global demand for transthyretin stabilizers has surged following regulatory approvals for treating ATTR-CM and ATTR-PN, placing immense pressure on supply chains to deliver high-quality active pharmaceutical ingredients reliably. Patent CN113735792A introduces a transformative preparation method for meglumine clofenazate and its critical intermediates, addressing long-standing inefficiencies in the production of this life-saving cardiovascular medication. This proprietary technology shifts the paradigm from hazardous, low-yield legacy processes to a streamlined, environmentally friendly synthesis that ensures consistent supply continuity. By leveraging mild reaction conditions and avoiding toxic reagents, this innovation not only enhances operator safety but also drastically simplifies downstream processing, resulting in intermediates with exceptional purity profiles suitable for direct pharmaceutical formulation.
The strategic value of this patent lies in its ability to overcome the specific bottlenecks that have historically plagued the commercial manufacturing of tafamidis derivatives. Traditional routes often suffered from cumulative yield losses and difficult-to-remove impurities, necessitating expensive chromatographic purifications that are impractical at scale. In contrast, the disclosed methodology employs a robust four-step sequence that maximizes atom economy and minimizes waste generation. For procurement leaders and supply chain directors, this represents a critical opportunity to secure a more resilient source of high-purity pharmaceutical intermediates, reducing the risk of production delays caused by complex purification failures or regulatory scrutiny over solvent residues.
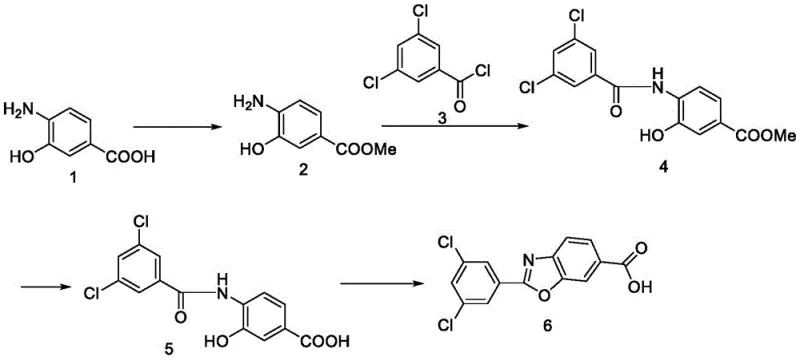
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for the key benzoxazole intermediate have been fraught with significant operational and safety hazards that render them suboptimal for modern GMP manufacturing environments. As illustrated in earlier literature, such as the route reported in patent CN100448852C, the reliance on pyridine, xylene, and trimethylsilyldiazomethane introduces severe toxicity risks and complicates waste disposal protocols. Furthermore, these legacy methods typically achieve a dismal total yield of less than 18%, forcing manufacturers to process vast quantities of raw materials to obtain minimal product output. The accumulation of byproducts in these older pathways often results in crude products with poor purity, necessitating rigorous and costly purification steps that erode profit margins and extend lead times.
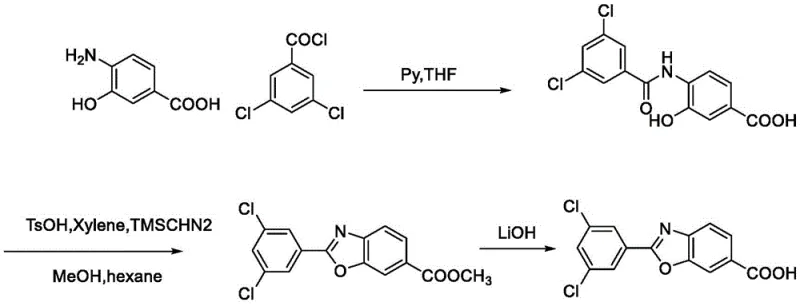
The Novel Approach
The innovative strategy detailed in the present patent fundamentally reengineers the synthetic pathway to prioritize safety, efficiency, and scalability without compromising chemical integrity. By substituting hazardous reagents with benign alternatives like methanesulfonic acid and utilizing common solvents such as tetrahydrofuran and isopropanol, the new process aligns perfectly with green chemistry principles. This approach facilitates a dramatic improvement in total yield and product quality, achieving pharmaceutical-grade purity directly from crystallization. The elimination of transition metal catalysts and toxic diazo compounds means that the burden on environmental health and safety departments is significantly reduced, allowing for smoother regulatory filings and faster time-to-market for the final drug product.
Mechanistic Insights into Methanesulfonic Acid Catalyzed Cyclization
The cornerstone of this improved synthesis is the intramolecular condensation reaction that constructs the benzoxazole core, a step that historically presented the greatest challenge regarding yield and impurity control. In the novel protocol, the precursor carboxylic acid undergoes cyclization in the presence of methanesulfonic acid acting as a potent organic acid catalyst within a mixed solvent system of THF and toluene. This specific catalytic environment promotes the dehydration and ring-closure mechanism efficiently at elevated temperatures around 108°C, driving the equilibrium towards the desired heterocyclic product while minimizing side reactions. The use of a water separator to continuously remove generated water further pushes the reaction to completion, ensuring that the conversion of the linear amide precursor into the cyclic benzoxazole structure is nearly quantitative.
From an impurity control perspective, the choice of methanesulfonic acid over harsher inorganic acids or Lewis acids prevents the degradation of the sensitive dichlorophenyl moiety and the glucosamine-compatible carboxylic acid group. The reaction conditions are tuned to avoid the formation of polymeric byproducts or over-chlorinated species that often plague Friedel-Crafts type cyclizations. Consequently, the crude product emerging from this step exhibits an HPLC purity exceeding 99%, which simplifies the subsequent salt formation step. This high level of mechanistic control ensures that the final API meets the stringent impurity thresholds required by global pharmacopoeias, reducing the need for repetitive recrystallization cycles that lower overall throughput.
How to Synthesize 2-(3,5-dichlorophenyl)-1,3-benzoxazole-6-carboxylic Acid Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature profiles across four distinct chemical transformations to maximize yield and purity. The process begins with the esterification of the starting amino-hydroxy benzoic acid, followed by selective acylation, hydrolysis, and finally the critical cyclization step. Each stage has been optimized to utilize straightforward workup procedures such as filtration and pulping, avoiding complex extractions or column chromatography. For detailed operational parameters including specific molar ratios, temperature ramps, and isolation techniques, please refer to the standardized synthesis guide provided below.
- Perform condensation of compound 1 with thionyl chloride and methanol to obtain the methyl ester intermediate (compound 2).
- React compound 2 with 3,5-dichlorobenzoyl chloride in the presence of triethylamine to form the amide precursor (compound 4).
- Hydrolyze the ester group of compound 4 using lithium hydroxide to generate the free acid (compound 5), followed by acidification.
- Execute intramolecular cyclization of compound 5 using methanesulfonic acid catalyst in refluxing toluene/THF to yield the final benzoxazole acid (compound 6).
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced manufacturing route offers substantial strategic benefits for organizations seeking to optimize their supply chain resilience and cost structures in the competitive cardiovascular therapeutic market. The shift away from exotic and hazardous reagents towards commodity chemicals significantly lowers the barrier to entry for production and reduces the volatility associated with sourcing specialized raw materials. Moreover, the simplified post-treatment workflow, which relies heavily on crystallization and filtration rather than energy-intensive distillation or chromatography, translates directly into reduced utility consumption and shorter batch cycle times. These operational efficiencies allow manufacturers to respond more agilely to fluctuations in market demand while maintaining robust inventory levels of critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents such as trimethylsilyldiazomethane and pyridine removes the need for specialized containment systems and costly hazardous waste disposal services. By utilizing readily available solvents like THF and isopropanol, the direct material costs are significantly lowered, and the overall process mass intensity is improved. The high yield achieved at each step minimizes the loss of valuable starting materials, ensuring that the cost per kilogram of the final API is optimized for commercial viability without sacrificing quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials mitigates the risk of supply disruptions that often occur with custom-synthesized specialty reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and ensuring that downstream formulation partners receive their shipments on time, thereby strengthening the overall integrity of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in multipurpose chemical plants, such as stirred tank reactors and filter dryers. The absence of heavy metal catalysts simplifies the regulatory clearance process and reduces the environmental footprint of the manufacturing site. This alignment with sustainability goals not only future-proofs the production facility against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the global healthcare ecosystem.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion. The answers provided reflect the specific advantages of this route over legacy methods, focusing on purity, safety, and operational simplicity.
Q: How does the new synthesis route improve upon conventional methods for Tafamidis intermediates?
A: The novel route eliminates highly toxic solvents like pyridine, xylene, and trimethylsilyldiazomethane used in prior art, replacing them with safer alternatives like THF and methanesulfonic acid while significantly boosting total yield and product purity to pharmaceutical standards.
Q: What are the critical quality attributes achieved in the final Meglumine Clofenazate API?
A: The process delivers an API with HPLC purity exceeding 99.8%, with maximum single impurity levels controlled below 0.08%, meeting stringent raw material drug specifications without requiring complex purification steps.
Q: Is this manufacturing process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions, commercially available reagents, and simple post-treatment procedures such as filtration and pulping, making it highly adaptable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meglumine Clofenazate Supplier
As the demand for ATTR-CM treatments continues to grow, securing a dependable source of high-quality intermediates is paramount for pharmaceutical developers and generic manufacturers alike. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent results. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of meglumine clofenazate intermediate meets the exacting standards required for clinical and commercial use. We combine deep technical expertise with a commitment to operational excellence to support your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless path from development to commercial success.
