Advanced Synthesis of Pentacyclic Sesquiterpene Hydroquinone Intermediates for Pharma R&D
Introduction to Advanced Sesquiterpene Hydroquinone Synthesis
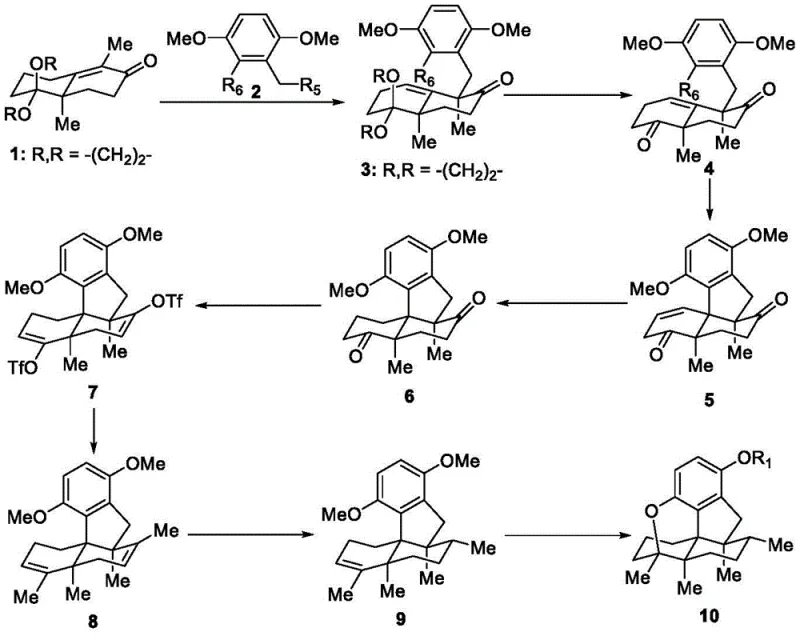
The field of natural product synthesis continues to push the boundaries of organic chemistry, particularly when addressing complex scaffolds like sesquiterpene hydroquinones. These mixed biogenic heteroterpenoids, characterized by a sesquiterpene fragment linked to a hydroquinone moiety, exhibit a remarkable spectrum of biological activities including antibacterial, antifungal, anti-HIV, and antitumor properties. However, their dense chiral centers and crowded polycyclic skeletons have historically made their acquisition difficult. Patent CN115197188A introduces a groundbreaking preparation method for a class of sesquiterpene hydroquinone compounds featuring a pentacyclic skeleton, as shown in Formula (I). This innovation represents a significant leap forward for reliable pharmaceutical intermediate suppliers seeking to provide high-quality materials for drug discovery programs. By leveraging a derivative of the Wieland-Miescher ketone as a strategic starting point, the inventors have established a route that balances structural complexity with operational simplicity.
The significance of this patent lies not just in the molecules produced, but in the strategic disconnection of the complex pentacyclic core. Traditional approaches to such densely functionalized natural products often rely on lengthy linear sequences or low-yielding biomimetic cyclizations that struggle with stereocontrol. In contrast, the methodology described in CN115197188A offers a convergent strategy that builds complexity early in the sequence. This allows for the efficient generation of diverse analogs by varying the substituents on the aromatic ring or the terpene fragment. For R&D teams evaluating structure-activity relationships (SAR), having access to a flexible synthetic platform is invaluable. The ability to rapidly access these pentacyclic scaffolds ensures that biological evaluation is not bottlenecked by material scarcity, thereby accelerating the identification of new lead compounds for cancer therapy and other indications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of polycyclic sesquiterpene hydroquinones has been plagued by significant supply chain vulnerabilities inherent to natural product isolation. Extraction from natural sources is notoriously inefficient, often requiring tons of biomass to yield grams of pure compound, leading to exorbitant costs and inconsistent quality. Furthermore, natural extracts frequently contain a cocktail of structurally similar analogs that are exceedingly difficult to separate, complicating the generation of pure standards required for rigorous toxicological and pharmacological profiling. From a synthetic chemistry perspective, prior art methods for constructing these fused ring systems often necessitated harsh reaction conditions, such as strong Lewis acids at elevated temperatures, which could degrade sensitive functional groups or lead to racemization of chiral centers. These conventional routes typically lacked modularity, meaning that synthesizing a single analog required a completely redesigned pathway, rendering the cost reduction in pharmaceutical intermediate manufacturing nearly impossible for diverse libraries.
The Novel Approach
The methodology disclosed in the patent data fundamentally shifts the paradigm by utilizing a robust, step-wise construction of the pentacyclic core from a chiral pool precursor. The process initiates with the alkylation of a Wieland-Miescher ketone derivative, a commercially accessible and well-understood building block that already possesses the necessary stereochemical information. This strategic choice eliminates the need for difficult asymmetric inductions later in the synthesis. As illustrated in the comprehensive reaction scheme, the route proceeds through a series of high-fidelity transformations including palladium-catalyzed cyclizations and selective hydrogenations. This approach ensures that the commercial scale-up of complex pharmaceutical intermediates is feasible because each step relies on established unit operations familiar to process chemists. The modularity of the aromatic substitution allows for the rapid generation of a library of derivatives without altering the core terpene synthesis, providing a versatile platform for medicinal chemistry optimization.
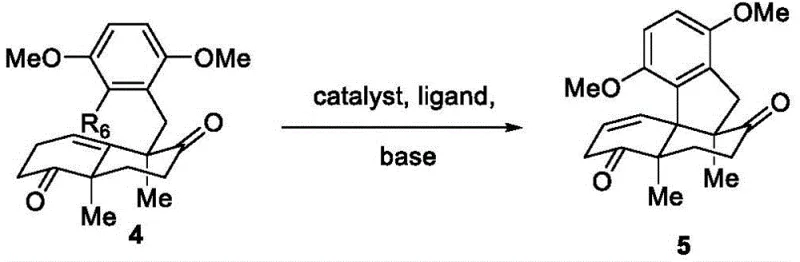
Mechanistic Insights into Palladium-Catalyzed Cyclization
A critical juncture in this synthetic sequence is the formation of the tetracyclic enone intermediate via an intramolecular palladium-catalyzed coupling. This transformation, converting the diketone precursor into the fused ring system, is the engine of complexity generation in this route. The reaction employs a palladium catalyst system, such as Pd2(dba)3 paired with bulky phosphine ligands like SPhos or XantPhos, in a polar aprotic solvent like DMF at elevated temperatures (around 120°C). Mechanistically, this likely proceeds through an oxidative addition of the aryl halide moiety to the Pd(0) center, followed by coordination and insertion into the enolate generated from the adjacent ketone. The steric bulk of the ligand is crucial here; it facilitates the reductive elimination step to close the strained ring while preventing the formation of palladium black or inactive clusters. This careful tuning of the catalytic cycle ensures that the reaction proceeds with high regioselectivity, minimizing the formation of regioisomeric byproducts that would be difficult to remove downstream.
Following the construction of the carbon skeleton, the final closure of the pentacyclic ether ring represents another sophisticated mechanistic feat. This step typically involves the activation of a phenolic or benzylic position using a Lewis acid or Brønsted acid, such as boron tribromide (BBr3) or p-toluenesulfonic acid. The mechanism involves the generation of a reactive carbocation or activated complex that undergoes an intramolecular nucleophilic attack by a proximal hydroxyl group. The stereochemical outcome of this cyclization is dictated by the rigid conformation of the pre-existing tetracyclic framework, which directs the incoming nucleophile to a specific face, thereby setting the final stereocenters with high fidelity. Impurity control in this stage is managed by strict temperature regulation and stoichiometry of the acid reagent; excessive acidity or heat could lead to elimination side reactions or rearrangement of the sensitive terpene skeleton. By optimizing these parameters, the process achieves a clean conversion to the target pentacyclic ether, ensuring the final API intermediate meets stringent purity specifications required for biological testing.
How to Synthesize Pentacyclic Sesquiterpene Hydroquinone Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation, particularly during the metal-catalyzed steps. The process begins with the careful preparation of the alkylated ketone intermediate, where moisture control is essential to prevent hydrolysis of the enolate. Subsequent steps involve standard workup procedures like aqueous quenching and organic extraction, but the purification of the polycyclic intermediates often benefits from gradient column chromatography to separate closely related diastereomers. The patent details specific conditions for each transformation, providing a robust blueprint for laboratory execution. For process chemists looking to implement this route, understanding the kinetics of the cyclization steps is key to reducing lead time for high-purity pharmaceutical intermediates. Detailed standardized synthetic steps for this pathway are provided in the guide below.
- Alkylation of Wieland-Miescher ketone derivative with a substituted benzyl bromide using a strong base like t-BuOK to form the initial carbon-carbon bond.
- Intramolecular palladium-catalyzed cyclization to construct the crowded polycyclic core skeleton under heating in polar aprotic solvents.
- Final acid-mediated cyclization or deprotection using reagents like boron tribromide to close the ether ring and finalize the pentacyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers distinct advantages over traditional sourcing models. The reliance on the Wieland-Miescher ketone derivative as a starting material anchors the supply chain in stable, commodity chemical markets, reducing the risk of raw material shortages that often plague exotic natural product synthesis. Furthermore, the synthetic pathway avoids the use of extremely hazardous reagents or cryogenic conditions in the majority of steps, which simplifies the engineering requirements for manufacturing facilities. This operational simplicity translates directly into lower capital expenditure for equipment and reduced operational costs for energy and safety compliance. By establishing a domestic or regional synthesis capability for these complex intermediates, organizations can mitigate geopolitical supply risks and ensure a continuous flow of materials for their R&D pipelines.
- Cost Reduction in Manufacturing: The synthetic strategy significantly lowers the cost baseline by utilizing a convergent approach that maximizes atom economy in the key ring-forming steps. Unlike total synthesis routes that might require 20+ steps with cumulative yield losses, this streamlined pathway reduces the number of unit operations, thereby cutting down on solvent consumption, labor hours, and waste disposal costs. The use of recyclable palladium catalysts in the cyclization step further enhances the economic viability, as precious metal recovery protocols can be integrated into the process workflow to reclaim value from the spent catalyst streams.
- Enhanced Supply Chain Reliability: By shifting from extraction-dependent sourcing to a fully synthetic route, supply continuity is decoupled from agricultural variables such as harvest seasons, weather patterns, or geographical instability in sourcing regions. The synthetic intermediates are stable solids that can be stockpiled effectively, allowing for just-in-time manufacturing strategies. Additionally, the robustness of the chemical steps means that technology transfer between different manufacturing sites is straightforward, enabling a diversified supply base that protects against single-point failures in the production network.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like THF, DMF, and ethyl acetate which are standard in large-scale pharmaceutical manufacturing and have well-established recovery and recycling infrastructures. The avoidance of heavy metal contaminants in the final product, achieved through efficient purification and catalyst scavenging, ensures compliance with strict ICH guidelines for elemental impurities. This environmental profile simplifies the regulatory filing process and reduces the burden of waste treatment, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pentacyclic sesquiterpene hydroquinone compounds. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is critical for making informed decisions about integrating these intermediates into your drug discovery workflows.
Q: What are the key challenges in synthesizing polycyclic sesquiterpene hydroquinones?
A: The primary challenges involve managing dense chiral centers, relieving significant molecular ring strain in the crowded polycyclic skeleton, and achieving high regioselectivity during ring-closing steps without forming complex impurity profiles.
Q: How does this novel route improve upon traditional extraction methods?
A: Unlike natural extraction which suffers from low yields and difficult purification due to structural analogs, this chemical synthesis provides a consistent, scalable supply of specific isomers with defined stereochemistry using readily available starting materials.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the route utilizes robust reactions such as hydrogenation and standard palladium couplings in common industrial solvents like THF and DMF, avoiding extreme cryogenic conditions in most steps, which facilitates commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sesquiterpene Hydroquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating the drug discovery timeline. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop synthesis to industrial manufacturing is seamless. We are committed to delivering stringent purity specifications for every batch, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require milligram quantities for initial screening or metric tons for clinical supply, our flexible manufacturing capabilities are designed to meet your evolving needs with speed and precision.
We invite you to collaborate with us to unlock the full potential of this novel synthetic technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments for your next project. Let us be your strategic partner in navigating the complexities of fine chemical synthesis and securing a reliable supply of advanced pharmaceutical intermediates.
