Advanced Synthetic Route for N-Acyl-Biphenylalanine: Enhancing Commercial Viability and Supply Stability
The pharmaceutical industry continuously seeks robust synthetic pathways for chiral amino acid derivatives, which serve as critical building blocks for next-generation therapeutics. Patent CN101684077B introduces a highly efficient methodology for the preparation of N-acyl-biphenylalanine, a key intermediate often utilized in the synthesis of novel drug candidates targeting various metabolic and oncological pathways. This technology represents a significant departure from legacy synthesis routes that have historically plagued R&D departments with prohibitive costs and supply chain bottlenecks. By leveraging a condensation-hydrolysis-hydrogenation sequence, the disclosed process transforms inexpensive commodity chemicals into high-value fine chemical intermediates with remarkable efficiency. For global procurement teams and supply chain directors, understanding the nuances of this patent is essential, as it offers a viable alternative to scarce chiral pool resources. The strategic implementation of this chemistry allows manufacturers to secure a reliable pharmaceutical intermediate supplier status by mitigating the risks associated with volatile raw material markets. Furthermore, the process design emphasizes operational simplicity, utilizing standard unit operations that are easily adaptable to existing manufacturing infrastructure, thereby ensuring continuity of supply for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenylalanine derivatives has been constrained by reliance on expensive and non-natural starting materials, creating substantial barriers to entry for cost-sensitive manufacturing projects. Prior art, such as methods described in the Journal of Medicinal Chemistry, predominantly utilized D-tyrosine as the foundational chiral source, a material that commands a premium price due to its limited natural abundance and complex extraction processes. Moreover, these traditional routes necessitated the use of trifluoromethanesulfonic anhydride for phenolic hydroxyl activation, a reagent that is not only costly but also poses significant handling hazards and waste disposal challenges in a commercial setting. The cumulative effect of these factors results in a manufacturing process with a bloated cost structure, making the final active pharmaceutical ingredient economically unviable for many therapeutic areas. Additionally, the multi-step nature of these older methodologies often led to compounded yield losses, further exacerbating the economic inefficiency and complicating the purification of the final product from persistent impurities. For a procurement manager, sourcing such specialized precursors often involves long lead times and single-source dependencies, introducing unacceptable risk into the supply chain.
The Novel Approach
In stark contrast, the methodology outlined in CN101684077B revolutionizes the production landscape by employing a convergent synthesis strategy centered on the Erlenmeyer-Plöchl azlactone synthesis. This innovative approach substitutes the costly chiral pool materials with biphenylcarboxaldehyde and N-acylglycine, both of which are widely available commodity chemicals with stable market pricing and robust supply chains. The elimination of exotic activating agents like trifluoromethanesulfonic anhydride in favor of simple acid anhydrides significantly lowers the barrier to entry for cost reduction in pharmaceutical intermediate manufacturing. The process flow is streamlined into three distinct, high-yielding stages that minimize unit operations and solvent exchanges, directly translating to reduced operational expenditures and a smaller environmental footprint. By shifting the complexity from raw material sourcing to process optimization, this method empowers manufacturers to achieve commercial scale-up of complex amino acid derivatives with greater predictability and control. This strategic pivot not only enhances profit margins but also ensures that the production of these vital intermediates remains resilient against market fluctuations in specialty chemical availability.
Mechanistic Insights into Azlactone Formation and Catalytic Hydrogenation
The core of this synthetic innovation lies in the initial condensation reaction, where biphenylcarboxaldehyde reacts with N-acylglycine in the presence of an acid anhydride and a base to form an oxazolone intermediate, commonly known as an azlactone. This transformation proceeds through the formation of an enolate from the N-acylglycine, which subsequently attacks the carbonyl carbon of the aldehyde, followed by cyclization and dehydration driven by the anhydride. The choice of base, ranging from tertiary amines like triethylamine to inorganic salts like sodium acetate, plays a critical role in modulating the reaction kinetics and ensuring high conversion rates without promoting side reactions. The resulting azlactone serves as a versatile synthon, stabilizing the reactive intermediate and allowing for easy isolation and purification before proceeding to the next stage. This mechanistic pathway is particularly advantageous because it avoids the formation of difficult-to-remove byproducts often associated with direct peptide coupling methods, thereby simplifying the downstream purification burden.
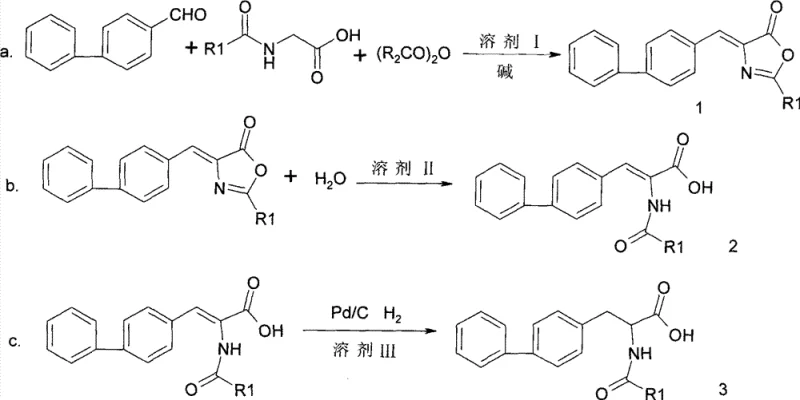
Following the formation of the azlactone, the process advances through a hydrolysis step to generate the dehydro-amino acid derivative, which is then subjected to catalytic hydrogenation to yield the final saturated N-acyl-biphenylalanine. The hydrolysis step is carefully controlled using water or aqueous-organic solvent mixtures under reflux conditions to open the oxazolone ring without degrading the sensitive olefinic bond. The final hydrogenation step utilizes palladium on carbon (Pd/C) as a heterogeneous catalyst under acidic conditions, typically maintained by the addition of glacial acetic acid. This acidic environment is crucial for protonating the substrate and facilitating the reduction of the carbon-carbon double bond while preserving the stereochemical integrity required for subsequent resolution steps. The ability to tune hydrogen pressure between 0.2MPa and 10.0MPa provides operators with precise control over reaction rates and selectivity, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications. This comprehensive mechanistic understanding allows process chemists to optimize reaction parameters for maximum efficiency and minimal impurity generation.
How to Synthesize N-Acyl-Biphenylalanine Efficiently
Implementing this synthesis route requires a systematic approach to reaction engineering, focusing on the precise control of stoichiometry, temperature, and pressure across the three sequential stages. The initial condensation demands rigorous exclusion of moisture to prevent premature hydrolysis of the anhydride, while the subsequent hydrolysis step requires careful management of solvent ratios to maximize precipitation of the intermediate. The final hydrogenation step necessitates specialized high-pressure equipment and strict safety protocols to handle hydrogen gas, but the robustness of the Pd/C catalyst system allows for multiple cycles or continuous processing configurations. Detailed standard operating procedures for each stage are critical to reproducing the high yields reported in the patent examples, which consistently exceed 80% across all steps. For technical teams looking to adopt this technology, the following guide outlines the fundamental operational framework derived from the patent disclosure.
- Condense biphenylcarboxaldehyde with N-acylglycine and acid anhydride in the presence of a base under reflux to form the oxazolone intermediate.
- Hydrolyze the oxazolone intermediate in water or organic solvent mixtures under reflux to obtain the dehydro-amino acid derivative.
- Perform catalytic hydrogenation using Pd/C under acidic conditions and controlled hydrogen pressure to yield the final N-acyl-biphenylalanine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for organizations aiming to optimize their cost structures and secure their supply lines against external disruptions. The shift away from proprietary or scarce chiral starting materials towards bulk commodity chemicals fundamentally alters the cost dynamics of the manufacturing process, enabling significant savings that can be passed down the value chain. This strategic alignment with readily available feedstocks ensures that production schedules are not held hostage by the lead times of niche suppliers, thereby enhancing overall supply chain reliability and responsiveness to market demand. Furthermore, the simplified workflow reduces the number of isolation and purification steps, which directly correlates to lower utility consumption, reduced solvent waste, and decreased labor hours per kilogram of product. These operational efficiencies compound to create a highly competitive manufacturing profile that is attractive for long-term partnerships in the generic and specialty pharmaceutical sectors.
- Cost Reduction in Manufacturing: The elimination of expensive reagents such as trifluoromethanesulfonic anhydride and D-tyrosine removes a major cost driver from the bill of materials, allowing for a drastic reduction in the overall cost of goods sold. By utilizing acetic anhydride and simple bases like sodium acetate, the process leverages low-cost inputs that are produced at massive global scales, ensuring price stability and availability. Additionally, the high yields achieved in each step minimize the loss of valuable intermediates, further enhancing the economic viability of the process by maximizing the output from every batch. This cost efficiency is critical for maintaining competitiveness in markets where price pressure is intense and margin preservation is paramount for business sustainability.
- Enhanced Supply Chain Reliability: Sourcing biphenylcarboxaldehyde and N-acylglycine is significantly less risky than procuring specialized chiral amino acids, as these materials are produced by multiple vendors worldwide, reducing the threat of single-point failures. The robustness of the chemical transformations means that minor variations in raw material quality can often be accommodated without compromising the final product specification, adding a layer of resilience to the supply chain. This flexibility allows procurement managers to negotiate better terms with suppliers and maintain leaner inventory levels without fear of production stoppages. Consequently, the ability to rapidly scale production in response to surges in demand is greatly improved, ensuring that downstream customers receive their orders on time and in full.
- Scalability and Environmental Compliance: The process conditions, primarily involving reflux temperatures and standard pressures, are easily transferable from pilot plants to multi-ton reactors without requiring exotic equipment or extreme operating parameters. The use of common solvents like ethanol, acetone, and ethyl acetate simplifies solvent recovery and recycling systems, contributing to a greener manufacturing profile that aligns with modern environmental regulations. Reduced waste generation from fewer synthetic steps and the avoidance of hazardous fluorinated reagents lower the costs associated with waste treatment and disposal. This environmental stewardship not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for sustainability-conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acyl-biphenylalanine synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent, providing a factual basis for decision-making. Understanding these details is crucial for technical directors evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers highlight the balance between chemical efficiency and operational practicality that defines this innovative approach.
Q: What are the primary cost advantages of this synthesis route compared to traditional methods?
A: Unlike conventional methods relying on expensive non-natural D-tyrosine and trifluoromethanesulfonic anhydride, this process utilizes readily available biphenylcarboxaldehyde and N-acylglycine, drastically reducing raw material costs and simplifying procurement logistics.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is designed for large-scale production, utilizing standard reflux conditions and common solvents like acetic anhydride and ethanol, which facilitates easy scale-up from laboratory to commercial tonnage without complex equipment requirements.
Q: How does the hydrogenation step ensure high purity of the final product?
A: The final step employs Pd/C catalysis under controlled acidic conditions and specific hydrogen pressures (0.2MPa to 10.0MPa), which effectively reduces the double bond while minimizing side reactions, resulting in high yields ranging from 80% to over 94% in demonstrated examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl-Biphenylalanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the future of pharmaceutical development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into reliable industrial realities. We are committed to delivering high-purity N-acyl-biphenylalanine intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of this synthesis, including high-pressure hydrogenation and precise temperature control, guaranteeing consistent quality batch after batch.
We invite you to collaborate with us to leverage this advanced technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization. Let us be your partner in navigating the complexities of fine chemical synthesis and achieving your supply chain goals.
