Advanced Base-Catalyzed Synthesis of 1,3,4,5-Tetraaryl Pyrazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways for nitrogen-containing heterocycles, particularly pyrazoles, due to their ubiquitous presence in bioactive molecules. Patent CN108947906B, published in August 2021, introduces a groundbreaking methodology for the preparation of 1,3,4,5-tetraaryl substituted pyrazoles that addresses critical bottlenecks in existing manufacturing technologies. This innovation leverages a tandem dipolar cycloaddition and desulfonylation strategy, utilizing benzosultam and N-aryl arylmethyl hydrazine acyl chloride as key starting materials under mild basic catalysis. Unlike traditional methods that often suffer from harsh conditions or poor selectivity, this novel approach operates efficiently at temperatures ranging from 0°C to 40°C, ensuring high product yields and exceptional purity profiles. For global procurement teams and R&D directors, this patent represents a significant opportunity to optimize the supply chain for complex heterocyclic intermediates, offering a route that is not only chemically elegant but also commercially viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyaryl substituted pyrazoles has relied heavily on the condensation reaction between hydrazine compounds and 1,3-dicarbonyl compounds or α,β-unsaturated carbonyl compounds. As illustrated in the reaction scheme below, this classical approach frequently encounters severe limitations regarding substrate applicability and regioselectivity. The reaction often produces a complex mixture of N1 and N2 substituted isomers, necessitating difficult and costly separation processes that drastically reduce overall efficiency. Furthermore, the requirement for specific carbonyl precursors limits the structural diversity of the final products, restricting the ability of medicinal chemists to explore broad chemical space for drug discovery programs.
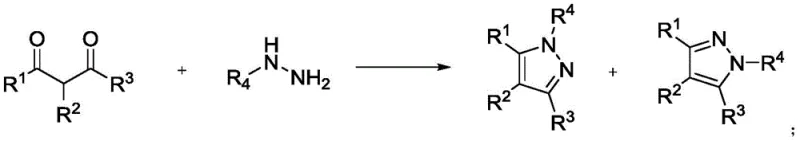
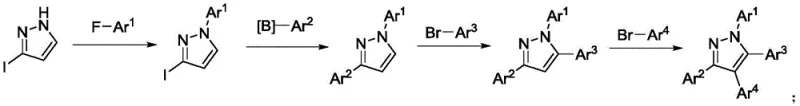
In addition to selectivity issues, more modern attempts to synthesize tetraaryl pyrazoles have involved multi-step sequences utilizing noble metal catalysts. For instance, previous literature describes a four-step reaction pathway starting from 3-iodo-1H-pyrazole, employing SNAr reactions, coupling reactions, and C-H arylation. While this method can achieve the desired substitution pattern, it relies heavily on expensive palladium catalysts, which negatively impacts the reaction economy and introduces the risk of heavy metal contamination in the final API. The lengthy synthetic route also results in cumulative yield losses and increased waste generation, making it less attractive for cost-sensitive commercial manufacturing environments where efficiency and sustainability are paramount.
The Novel Approach
The methodology disclosed in CN108947906B offers a transformative solution by streamlining the synthesis into a concise, one-pot transformation. The core of this innovation lies in the reaction between benzosultam (Formula I) and N-aryl arylmethyl hydrazine acyl chloride (Formula II) in the presence of a basic catalyst. This process triggers an addition reaction followed by a spontaneous loss of sulfur dioxide (SO2), directly yielding the 1,3,4,5-tetraaryl substituted pyrazole (Formula III). The reaction scheme demonstrates a remarkable simplicity, avoiding the need for transition metals and high-energy inputs. By operating under mild conditions with short reaction times, typically between 1.0 to 12.0 hours, this method significantly enhances throughput while minimizing energy consumption.
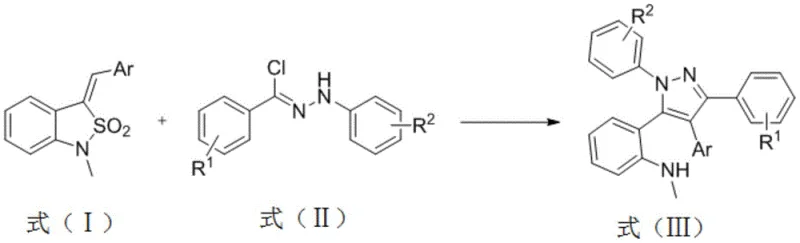
Moreover, the structural integrity of the product is exceptionally high, with the 5-position aromatic ring retaining a methylamino group that serves as a versatile handle for further derivatization. This feature is crucial for downstream applications, allowing chemists to easily modify the scaffold to create diverse libraries of pyrazole derivatives for biological testing. The high regioselectivity ensures that no other by-products are generated, simplifying the purification workflow to basic column chromatography or crystallization. For a reliable pharmaceutical intermediate supplier, this level of control translates directly into consistent quality and reduced batch-to-batch variability, which are critical metrics for regulatory compliance and customer satisfaction.
Mechanistic Insights into Base-Catalyzed Dipolar Cycloaddition
The mechanistic pathway of this reaction is a sophisticated example of 1,3-dipolar cycloaddition chemistry driven by the unique electronic properties of the benzosultam scaffold. Under the influence of a base such as triethylamine or diisopropylethylamine, the benzosultam undergoes deprotonation or activation to form a reactive dipole species. This dipole then engages in a concerted cycloaddition with the electrophilic carbon of the N-aryl arylmethyl hydrazine acyl chloride. The resulting cyclic intermediate is inherently unstable due to the presence of the sulfone moiety, which acts as a good leaving group. Consequently, the system undergoes a rapid extrusion of sulfur dioxide gas, aromatizing the five-membered ring to form the stable pyrazole core. This tandem sequence is thermodynamically favorable and kinetically fast, explaining the high yields observed even at ambient temperatures.
From an impurity control perspective, the mechanism inherently suppresses side reactions that plague other synthetic routes. Because the cyclization is driven by the specific geometry and electronic demand of the benzosultam dipole, there is minimal opportunity for alternative attack vectors that would lead to regioisomers. The absence of transition metals also eliminates the risk of metal-catalyzed homocoupling or decomposition of sensitive functional groups on the aryl rings. This clean reaction profile means that the crude product contains very few impurities, reducing the burden on downstream purification units. For R&D teams focused on process chemistry, understanding this mechanism allows for fine-tuning of reaction parameters, such as the choice of aprotic solvents like dichloromethane or toluene, to further optimize the reaction kinetics and solubility profiles of diverse substrates.
How to Synthesize 1,3,4,5-Tetraaryl Pyrazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and addition rates to maximize the benefits of the tandem cycloaddition-desulfonylation process. The patent outlines a straightforward protocol where benzosultam and the hydrazine acyl chloride are mixed in an aprotic solvent, followed by the controlled addition of the base. Detailed standardized synthesis steps see the guide below.
- Mix benzosultam and N-aryl arylmethyl hydrazine acyl chloride in an aprotic organic solvent such as dichloromethane or toluene at a molar ratio of 1: 1 to 1:1.5.
- Add a basic catalyst like triethylamine or diisopropylethylamine dropwise while maintaining the reaction temperature between 0°C and 40°C.
- Stir the mixture for 1.0 to 12.0 hours, then perform standard workup including phase separation, washing, and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and operational advantages over legacy methods. The elimination of palladium catalysts removes a significant cost driver from the bill of materials, as noble metals are subject to volatile market pricing and require specialized recovery processes. Furthermore, the mild reaction conditions reduce energy costs associated with heating and cooling, contributing to a lower overall cost of goods sold (COGS). The simplicity of the workup procedure, which avoids complex extraction or metal scavenging steps, also reduces labor time and solvent consumption, leading to substantial cost savings in manufacturing operations.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the replacement of expensive transition metal catalysts with inexpensive organic bases like triethylamine. This switch not only lowers direct material costs but also simplifies the purification process, as there is no need for expensive metal scavengers or rigorous testing for residual heavy metals. Additionally, the high atom economy of the reaction, characterized by the release of only gaseous SO2 as a byproduct, minimizes waste disposal fees. The ability to use common, low-cost solvents such as dichloromethane or toluene further enhances the financial viability of this process for large-scale production.
- Enhanced Supply Chain Reliability: The starting materials, benzosultam and N-aryl arylmethyl hydrazine acyl chloride, are chemically stable and can be sourced from a wide range of chemical suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or mixing efficiency, ensuring consistent output even in varied manufacturing environments. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing costly production delays caused by batch failures or quality deviations.
- Scalability and Environmental Compliance: The reaction generates minimal solid waste and the only significant byproduct is sulfur dioxide, which can be easily managed with standard scrubbing systems. The absence of heavy metals aligns with increasingly stringent environmental regulations regarding pharmaceutical waste, facilitating easier permitting and compliance auditing. The mild thermal profile (0°C to 40°C) makes the process inherently safer to scale up, reducing the risk of thermal runaways and allowing for the use of standard glass-lined or stainless steel reactors without specialized high-pressure equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: What are the primary advantages of this pyrazole synthesis method over palladium-catalyzed routes?
A: This method eliminates the need for expensive noble metal palladium catalysts, significantly reducing raw material costs and simplifying the purification process by avoiding heavy metal removal steps.
Q: How does this process improve regioselectivity compared to traditional condensation reactions?
A: By utilizing benzosultam as a specific dipole precursor in a tandem dipolar cycloaddition-desulfonylation sequence, the reaction achieves high regioselectivity, preventing the formation of N1/N2 substitution mixtures common in hydrazine-dicarbonyl condensations.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (0°C to 40°C) without heating, uses common aprotic solvents, and generates minimal waste, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4,5-Tetraaryl Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocycle synthesis in the development of next-generation therapeutics and agrochemicals. Our team of expert process chemists has extensively evaluated the methodology described in CN108947906B and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,3,4,5-tetraaryl pyrazole meets the exacting standards required for pharmaceutical grade materials.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your product development pipeline.
