Scalable Synthesis of Tert-Butyl (R)-4-Ethyl-1,2,3-Oxathiazolidine-3-Carboxylic Acid via Green Oxidation
The pharmaceutical industry continuously seeks robust and scalable pathways for complex heterocyclic intermediates, particularly those serving as chiral building blocks for advanced therapeutics. Patent CN116396241A introduces a groundbreaking preparation method for tert-butyl (R)-4-ethyl-2,2-dioxide-1,2,3-oxathiazolidine-3-carboxylic acid (CAS 1417287-40-1), a critical scaffold in modern drug discovery. This novel protocol departs from traditional reliance on precious metal catalysis, opting instead for a greener, three-step sequence initiated from the readily available chiral pool material, (R)-2-amino-1-butanol. By integrating a mild cyclization, an environmentally benign Oxone oxidation, and a standard Boc protection, this technology addresses the pressing demands for cost-effective and sustainable pharmaceutical intermediate manufacturing. For R&D directors and supply chain leaders, this represents a significant opportunity to secure a reliable source of high-purity materials while mitigating the risks associated with heavy metal contamination and volatile reagent handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonamide-based heterocycles like the target oxathiazolidine derivative has been plagued by significant technical and economic hurdles. Existing literature describes routes that rely heavily on N-Boc-(R)-2-amino-1-butanol as a starting point, necessitating early-stage protection that adds unnecessary steps and cost. More critically, the oxidation step in these conventional pathways often employs ruthenium trichloride (RuCl3) as a catalyst. As illustrated in the reaction scheme below, this heavy metal approach not only inflates the bill of materials due to the high price of ruthenium but also creates a substantial downstream burden for purification.
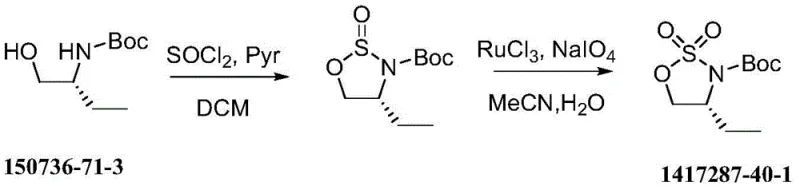
The removal of trace ruthenium to meet stringent pharmaceutical specifications typically requires specialized scavengers or repeated recrystallizations, which drastically reduce overall yield and extend production lead times. Furthermore, alternative one-step approaches utilizing sulfuryl chloride (SO2Cl2) present severe safety liabilities; this reagent is highly toxic, corrosive, and difficult to handle safely on a multi-kilogram scale, posing unacceptable risks for commercial API manufacturing facilities focused on operator safety and environmental compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN116396241A reinvents the synthetic logic by deferring the nitrogen protection until the final step and utilizing a metal-free oxidation strategy. This approach begins with the direct cyclization of the unprotected amino alcohol, leveraging the nucleophilicity of the amine and the hydroxyl group to form the cyclic sulfinyl intermediate efficiently. The subsequent oxidation utilizes Oxone (potassium persulfate), a cheap, stable, and non-toxic oxidant that converts the sulfinyl group to the desired sulfonyl dioxide without introducing heavy metal contaminants. This strategic shift not only simplifies the workup procedures but also aligns perfectly with the principles of green chemistry, offering a pathway that is inherently safer and more economically viable for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Oxone-Mediated Sulfoxide Oxidation
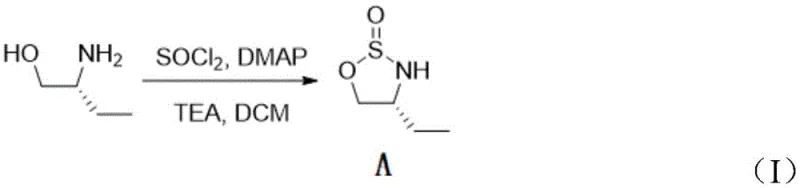
The core innovation of this process lies in the efficient transformation of the cyclic sulfinyl intermediate to the sulfone. The first step involves the reaction of (R)-2-amino-1-butanol with thionyl chloride in the presence of a base like triethylamine and a catalytic amount of DMAP. As shown in the reaction scheme, this proceeds through a chlorosulfite intermediate which undergoes intramolecular nucleophilic attack by the amine nitrogen, closing the five-membered ring to form the 1,2,3-oxathiazolidine 2-oxide structure. The use of DMAP is crucial here, as it accelerates the formation of the reactive sulfur species, allowing the reaction to proceed smoothly even at the low temperature of -20°C, thereby preserving the stereochemical integrity of the chiral center.
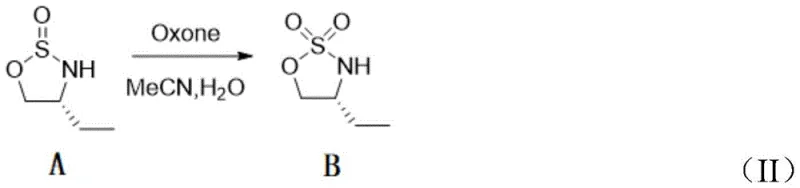
Following cyclization, the oxidation step utilizes the active oxygen species generated by Oxone in a biphasic acetonitrile-water system. The mechanism involves the transfer of an oxygen atom from the peroxymonosulfate anion to the sulfur atom of the sulfinyl ring. This reaction is remarkably clean, producing potassium sulfate and bisulfate as benign byproducts that are easily removed during the aqueous workup. Unlike transition metal catalysis, which can suffer from variable turnover numbers and catalyst deactivation, the stoichiometric Oxone oxidation provides consistent and high conversion rates. The final Boc protection step then caps the secondary sulfonamide nitrogen, yielding the stable, crystalline final product suitable for long-term storage and subsequent coupling reactions in drug synthesis.
How to Synthesize Tert-Butyl (R)-4-Ethyl-2,2-dioxide-1,2,3-oxathiazolidine-3-carboxylic acid Efficiently
The execution of this synthesis requires careful control of reaction parameters, particularly temperature during the initial cyclization and the stoichiometry of the oxidant. The process is designed to be telescoped where possible, although isolating the intermediates ensures the highest purity profile for the final API intermediate. The detailed standardized synthesis steps, including precise reagent equivalents, solvent volumes, and quenching protocols, are outlined in the structured guide below to facilitate immediate technology transfer to pilot plants.
- Cyclization of (R)-2-amino-1-butanol with thionyl chloride in DCM at -20°C to form the sulfinyl intermediate.
- Oxidation of the sulfinyl intermediate using Oxone (potassium persulfate) in acetonitrile/water at 80°C.
- Protection of the sulfonamide nitrogen with Boc-Cl in acetonitrile using TEA and DMAP at 60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. By eliminating the dependency on ruthenium catalysts, the supply chain becomes immune to the volatility of precious metal markets, ensuring more predictable pricing structures for long-term contracts. Furthermore, the replacement of hazardous sulfuryl chloride with safer reagents reduces the regulatory burden and insurance costs associated with handling extremely dangerous chemicals, directly contributing to cost reduction in API manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of expensive ruthenium catalysts and the associated metal scavenging resins results in a significantly lower cost of goods sold (COGS). Additionally, the use of Oxone, a commodity chemical, replaces costly specialized oxidants, while the high yields reported in each step minimize raw material waste and maximize throughput per batch.
- Enhanced Supply Chain Reliability: Starting from (R)-2-amino-1-butanol, a widely available chiral building block, ensures a robust upstream supply compared to protected amino alcohol derivatives which may have limited suppliers. The simplified purification process, which avoids complex chromatography in favor of crystallization and extraction, reduces the risk of production bottlenecks and ensures consistent on-time delivery for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The aqueous workup and the generation of non-toxic inorganic salts make waste treatment straightforward and compliant with increasingly strict environmental regulations. This 'green' profile facilitates easier permitting for scale-up activities, allowing manufacturers to rapidly expand capacity from pilot scale to multi-ton commercial production without significant infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational advantages for potential partners and licensees.
Q: Why is the Oxone oxidation method preferred over Ruthenium-catalyzed oxidation?
A: The Oxone method eliminates the need for expensive and toxic heavy metal catalysts like RuCl3, significantly reducing raw material costs and simplifying the removal of metal impurities to meet pharmaceutical standards.
Q: What are the safety advantages of this new preparation method?
A: Unlike conventional one-step methods using highly toxic sulfuryl chloride (SO2Cl2), this route uses thionyl chloride under controlled low temperatures and avoids large volumes of hazardous solvents, enhancing operational safety.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common reagents like Oxone and Boc-Cl, operates at moderate temperatures, and avoids complex chromatographic purifications in the final steps, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl (R)-4-Ethyl-2,2-dioxide-1,2,3-oxathiazolidine-3-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in CN116396241A can be seamlessly translated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of this critical intermediate meets the exacting standards required for global pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation drug candidates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your project's efficiency and profitability.
