Scalable Synthesis of (2S,4S)-4-Fluoropyrrolidine-2-Carboxylic Acid via Safe DeoxoFluor Fluorination
Introduction to Advanced Fluorinated Intermediate Synthesis
The pharmaceutical industry continuously demands high-purity chiral building blocks that can be manufactured safely and efficiently. Patent CN116396202A introduces a robust methodology for the preparation of (2S,4S)-4-fluoropyrrolidine-2-carboxylic acid (CAS: 2438-57-5), a critical scaffold in the development of protease inhibitors and peptidomimetics. This technical disclosure represents a significant paradigm shift from traditional fluorination protocols, addressing long-standing safety concerns associated with reactive sulfur-fluorine reagents. By leveraging a novel sequence involving DeoxoFluor-mediated substitution, the process achieves high stereochemical fidelity while operating under markedly milder conditions. For R&D directors and process chemists, this route offers a compelling alternative to legacy methods, promising enhanced operational safety and reduced environmental footprint without compromising the stringent purity specifications required for GMP production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-fluoropyrrolidine derivatives has relied heavily on diethylaminosulfur trifluoride (DAST) as the primary fluorinating agent. As illustrated in the prior art reaction scheme, this conventional approach necessitates extremely harsh cryogenic conditions, typically requiring reaction temperatures as low as -78°C to control the exotherm and prevent reagent decomposition. 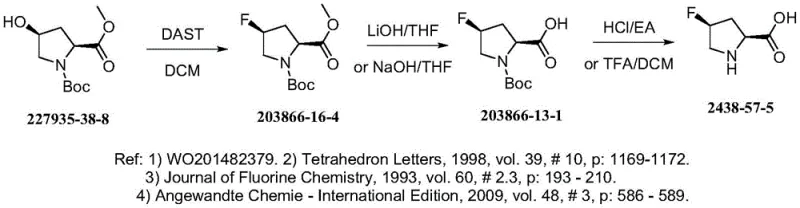 The inherent instability of DAST poses severe safety risks, particularly during the post-reaction workup and heating phases where rapid degradation can lead to dangerous pressure build-ups or explosions. Furthermore, the aggressive nature of DAST often results in complex impurity profiles, including elimination byproducts and rearrangement species, which complicate downstream purification and significantly lower the overall yield. These factors collectively create substantial bottlenecks for procurement and supply chain teams, as the specialized equipment needed for cryogenic processing drives up capital expenditure and limits the feasible batch sizes for commercial manufacturing.
The inherent instability of DAST poses severe safety risks, particularly during the post-reaction workup and heating phases where rapid degradation can lead to dangerous pressure build-ups or explosions. Furthermore, the aggressive nature of DAST often results in complex impurity profiles, including elimination byproducts and rearrangement species, which complicate downstream purification and significantly lower the overall yield. These factors collectively create substantial bottlenecks for procurement and supply chain teams, as the specialized equipment needed for cryogenic processing drives up capital expenditure and limits the feasible batch sizes for commercial manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN116396202A utilizes bis(2-methoxyethyl)aminosulfur trifluoride, commercially known as DeoxoFluor, to effect the critical hydroxyl-to-fluorine transformation. This modern reagent exhibits superior thermal stability, allowing the reaction to proceed efficiently at temperatures ranging from 0°C to ambient conditions, thereby eliminating the need for energy-intensive cryogenic cooling. The new route initiates with the esterification of (2S,4R)-N-Boc-4-hydroxy-proline, followed by the key fluorination step which proceeds with excellent conversion rates. By avoiding the volatility and instability associated with DAST, this novel approach drastically simplifies the engineering controls required for production. The transition to DeoxoFluor not only mitigates safety hazards but also enhances the reproducibility of the reaction, ensuring consistent quality across different production batches and facilitating a smoother technology transfer from laboratory to pilot plant scales.
Mechanistic Insights into DeoxoFluor-Mediated Stereoinversion
The core of this synthetic strategy lies in the precise stereochemical control exerted during the fluorination step. The reaction mechanism involves the activation of the secondary hydroxyl group at the C4 position of the pyrrolidine ring by the sulfur center of the DeoxoFluor reagent. 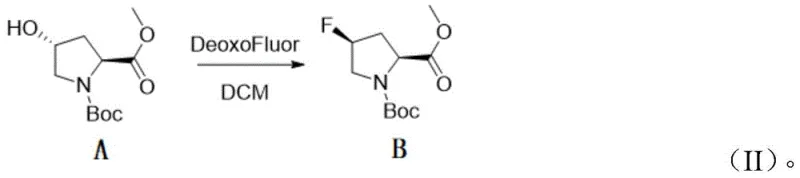 This activation generates a highly reactive sulfonium intermediate, which is subsequently displaced by a fluoride ion in a classic bimolecular nucleophilic substitution (SN2) fashion. Crucially, this backside attack results in the complete inversion of configuration at the chiral center, converting the (4R)-hydroxy precursor into the desired (4S)-fluoro product with high fidelity. The use of dichloromethane (DCM) as the solvent provides an optimal medium for solubilizing both the organic substrate and the fluorinating agent while maintaining the necessary reaction kinetics. From an impurity control perspective, the mildness of DeoxoFluor minimizes competing elimination reactions that typically lead to dehydro-proline byproducts, thereby simplifying the purification workflow and enhancing the overall mass balance of the process.
This activation generates a highly reactive sulfonium intermediate, which is subsequently displaced by a fluoride ion in a classic bimolecular nucleophilic substitution (SN2) fashion. Crucially, this backside attack results in the complete inversion of configuration at the chiral center, converting the (4R)-hydroxy precursor into the desired (4S)-fluoro product with high fidelity. The use of dichloromethane (DCM) as the solvent provides an optimal medium for solubilizing both the organic substrate and the fluorinating agent while maintaining the necessary reaction kinetics. From an impurity control perspective, the mildness of DeoxoFluor minimizes competing elimination reactions that typically lead to dehydro-proline byproducts, thereby simplifying the purification workflow and enhancing the overall mass balance of the process.
Furthermore, the subsequent deprotection and hydrolysis steps are designed to preserve the integrity of the newly formed carbon-fluorine bond. The removal of the tert-butoxycarbonyl (Boc) group using trifluoroacetic acid (TFA) is conducted under controlled conditions to prevent acid-catalyzed defluorination, a common pitfall in fluorinated amino acid synthesis. The final saponification using lithium hydroxide in a tetrahydrofuran-water system ensures gentle cleavage of the methyl ester without racemization at the alpha-carbon. This meticulous attention to reaction conditions throughout the four-step sequence underscores the robustness of the design, ensuring that the final active pharmaceutical ingredient (API) intermediate meets the rigorous enantiomeric excess and chemical purity standards demanded by regulatory bodies.
How to Synthesize (2S,4S)-4-Fluoropyrrolidine-2-Carboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing this valuable intermediate with high efficiency. The process begins with the esterification of the starting material, followed by the pivotal fluorination, deprotection, and final hydrolysis. Each step has been optimized to maximize yield and minimize waste, utilizing readily available reagents and standard unit operations.
- Perform methyl esterification of (2S,4R)-N-Boc-4-hydroxy-proline using methanol and p-toluenesulfonic acid at reflux.
- Conduct stereoselective fluorination using DeoxoFluor in DCM at 0°C to room temperature to invert stereochemistry.
- Remove the Boc protecting group with TFA, followed by ester hydrolysis using LiOH to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DeoxoFluor-based route offers transformative economic and logistical benefits. The elimination of cryogenic requirements translates directly into substantial cost reduction in API manufacturing, as facilities no longer need to invest in or maintain specialized low-temperature reactors and cooling infrastructure. The improved safety profile of DeoxoFluor reduces insurance premiums and operational downtime associated with hazard management, while the higher thermal stability allows for larger batch sizes that improve throughput. Additionally, the simplified workup procedures reduce solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The shift from DAST to DeoxoFluor removes the necessity for ultra-low temperature cooling (-78°C), which is energy-intensive and requires expensive equipment. By operating at 0°C to room temperature, the process significantly lowers utility costs and capital investment. Furthermore, the higher selectivity of the new method reduces the formation of difficult-to-remove impurities, lowering the cost of goods sold (COGS) associated with chromatographic purification and yield losses.
- Enhanced Supply Chain Reliability: The starting material, (2S,4R)-N-Boc-4-hydroxy-proline, is a commercially abundant chiral pool derivative, ensuring a stable and secure supply base. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or strict environmental constraints. This reliability allows for more accurate demand forecasting and inventory planning, reducing the risk of stockouts for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex fluorinated intermediates. The absence of thermally unstable reagents minimizes the risk of runaway reactions, a critical factor when scaling from kilograms to tons. Moreover, the reduced generation of hazardous byproducts simplifies waste treatment protocols, facilitating compliance with global environmental standards and accelerating the regulatory approval timeline for new drug applications utilizing this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (2S,4S)-4-fluoropyrrolidine-2-carboxylic acid. These insights are derived directly from the experimental data and process descriptions within the patent literature, providing a factual basis for decision-making.
Q: Why is DeoxoFluor preferred over DAST for this synthesis?
A: DeoxoFluor offers superior thermal stability and safety compared to DAST, eliminating the need for hazardous cryogenic conditions (-78°C) and reducing the risk of explosive decomposition during scale-up.
Q: What is the stereochemical outcome of the fluorination step?
A: The reaction proceeds via an SN2-type mechanism, resulting in the inversion of configuration from the (4R)-hydroxy precursor to the desired (4S)-fluoro product with high stereoselectivity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process operates at mild temperatures (0°C to ambient) and avoids unstable reagents, making it highly amenable to commercial scale-up with simplified engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,4S)-4-Fluoropyrrolidine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your supply chain efficiency.
