Advanced Synthesis of Chiral Alpha-Tribromomethyl Amines for Pharmaceutical Intermediates
Introduction to Advanced Chiral Amine Synthesis
The landscape of modern pharmaceutical development increasingly demands access to complex chiral building blocks that can serve as robust scaffolds for bioactive molecules. Patent CN102627590A introduces a significant breakthrough in this domain by disclosing a novel preparation method for chiral α-(tribromomethyl)amine compounds. These specific structural units are notoriously difficult to synthesize with high optical purity using traditional methods, yet they hold immense potential in the fields of asymmetric synthesis and medicinal chemistry. The invention addresses a critical gap in the availability of enantiomerically pure tribromomethylated amines, which are valuable intermediates for constructing nitrogen-containing heterocycles and other pharmacologically active structures. By leveraging a unique combination of sulfinylimines and bromoform under controlled basic conditions, this technology offers a pathway to high-value intermediates that were previously inaccessible or required prohibitively expensive resolution techniques.
For research and development teams focused on novel drug discovery, the ability to introduce a tribromomethyl group with defined stereochemistry opens new avenues for structure-activity relationship (SAR) studies. The patent outlines a general reaction scheme that is both versatile and robust, accommodating a wide range of substituents on the aromatic or aliphatic portions of the molecule. This flexibility is crucial for medicinal chemists who need to rapidly iterate on lead compounds. Furthermore, the methodology described ensures that the resulting products possess high optical purity, a non-negotiable requirement for regulatory approval in the pharmaceutical industry. As a reliable pharmaceutical intermediate supplier, understanding and implementing such advanced synthetic routes is essential for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of α-(tribromomethyl)amine compounds was fraught with significant technical challenges and limitations that hindered their widespread adoption in industrial settings. Existing methodologies primarily relied on the use of tribromoethyliminum precursors or tribromoacetic acid as reaction reagents, which often necessitated harsh reaction conditions that could compromise the integrity of sensitive functional groups. More critically, these conventional routes typically yielded achiral or racemic mixtures, failing to meet the stringent stereochemical requirements of modern life science applications. The lack of stereocontrol meant that extensive and costly downstream purification processes, such as chiral resolution or preparative HPLC, were required to isolate the desired enantiomer, drastically reducing overall process efficiency. Additionally, the stability of the tribromomethyl group under the aggressive conditions often employed in older methods was a persistent concern, leading to decomposition and lower yields.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent utilizes a mild and highly efficient strategy centered on the nucleophilic addition of a tribromomethyl anion to a chiral sulfinylimine. This method operates under remarkably gentle conditions, with reaction temperatures ranging from -60°C to 0°C, which effectively preserves the stability of the sensitive C-Br bonds while ensuring high selectivity. The use of commercially available and economically viable raw materials, such as bromoform and various sulfinylimines, significantly lowers the barrier to entry for production. The reaction proceeds with excellent diastereoselectivity, often achieving dr values as high as 99:1, which simplifies the purification process to mere recrystallization in many cases. This represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, as it eliminates the need for expensive chiral catalysts or complex separation technologies.

The versatility of this new route is further demonstrated by its compatibility with a diverse array of substrates, including those with electron-withdrawing or electron-donating groups on the aromatic ring. Whether the target molecule requires a simple phenyl group or a more complex heteroaromatic system like furyl or pyridyl, the protocol remains effective. This broad substrate scope makes it an invaluable tool for the commercial scale-up of complex pharmaceutical intermediates. By shifting the synthetic burden to the initial construction of the chiral center via the sulfinyl auxiliary, the process ensures that the final product is obtained with the necessary optical purity directly from the reaction vessel. This level of control is what distinguishes a laboratory curiosity from a viable industrial process capable of supporting the supply chains of major multinational corporations.
Mechanistic Insights into Asymmetric Nucleophilic Addition
The core of this technological advancement lies in the precise mechanistic pathway of the asymmetric nucleophilic addition. The reaction initiates with the deprotonation of bromoform by a strong base, such as sodium bis(trimethylsilyl)amide or potassium tert-butoxide, to generate the reactive tribromomethyl carbanion species. This nucleophile then attacks the electrophilic carbon of the C=N double bond in the sulfinylimine substrate. The chirality of the sulfinyl group, typically a tert-butyl sulfinyl moiety, acts as a powerful stereochemical director, shielding one face of the imine and forcing the nucleophilic attack to occur from the less hindered side. This steric guidance is the fundamental reason for the high diastereomeric ratios observed across the various examples provided in the patent data. The transition state is tightly organized, minimizing entropy loss and ensuring that the energy barrier for the formation of the major diastereomer is significantly lower than that of the minor one.
Beyond stereocontrol, the mechanism also inherently supports impurity control, which is a critical parameter for GMP manufacturing. The mild basic conditions and low temperatures prevent common side reactions such as the elimination of HBr from the tribromomethyl group, which would otherwise lead to the formation of unstable dibromo-imine byproducts. Furthermore, the choice of solvent, such as THF or ether, plays a crucial role in solvating the cation of the base and stabilizing the carbanion, thereby enhancing the reaction rate without compromising selectivity. The subsequent hydrolysis or transformation of the sulfinyl group can be performed under separate, optimized conditions, allowing for the modular synthesis of various amine derivatives. This mechanistic robustness ensures that the process is not only scientifically elegant but also practically reliable for producing high-purity pharmaceutical intermediates that meet strict regulatory specifications.
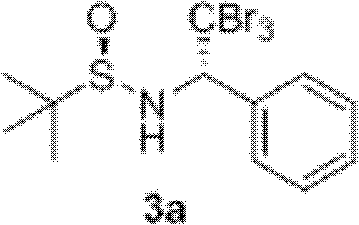
The utility of these chiral amines extends beyond their immediate isolation, as they serve as potent precursors for further chemical transformations. For instance, the patent demonstrates the conversion of the synthesized α-(tribromomethyl)amine into 2,2-dibromo ethylenimine derivatives through a base-mediated elimination reaction. This downstream capability highlights the dual functionality of the intermediate: it is both a stable, isolable product and a reactive synthon for constructing even more complex nitrogenous heterocycles. Such versatility is highly prized in process chemistry, where a single intermediate can feed into multiple synthetic pathways for different drug candidates. Understanding these mechanistic nuances allows process engineers to design flexible manufacturing lines that can adapt to changing project requirements without the need for entirely new infrastructure. It underscores the strategic value of adopting this technology for long-term R&D pipelines.
How to Synthesize Chiral Alpha-Tribromomethyl Amines Efficiently
Implementing this synthesis in a practical setting requires careful attention to the specific operational parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction system under an inert atmosphere, typically using dry THF or ether as the solvent to prevent premature quenching of the reactive base. The stoichiometry is critical, with a molar ratio of base to sulfinylimine to bromoform generally maintained around 1:1:1 to 1:1.5 to drive the reaction to completion while minimizing waste. Temperature control is paramount; the addition of reagents must be performed slowly at temperatures between -60°C and 0°C to manage the exotherm and maintain high stereoselectivity. Following the reaction period of 30 minutes to 3 hours, the mixture is quenched with water and extracted with organic solvents like ethyl acetate. The crude product is then purified, often simply by recrystallization from a mixture of ethyl acetate and petroleum ether, yielding the final high-purity compound.
- Prepare the reaction system by dissolving the sulfinylimine substrate in an anhydrous organic solvent such as THF or ether under inert atmosphere.
- Add a strong base like sodium bis(trimethylsilyl)amide and bromoform to the mixture while maintaining the temperature between -60°C and 0°C.
- Stir the reaction for 30 minutes to 3 hours, then quench with water and extract the product using ethyl acetate followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical industry. The shift away from precious metal catalysts and complex chiral auxiliaries that require stoichiometric amounts of expensive reagents translates into a leaner, more cost-effective supply chain. The reliance on commodity chemicals like bromoform and standard bases ensures that raw material sourcing is stable and not subject to the volatility often seen with specialized organometallic reagents. Moreover, the simplicity of the workup procedure, which avoids column chromatography in favor of crystallization, significantly reduces solvent consumption and processing time. These factors combine to create a manufacturing process that is not only economically superior but also more environmentally sustainable, aligning with the increasing corporate mandates for green chemistry practices.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available bromoform drastically lower the direct material costs associated with production. By achieving high diastereoselectivity directly in the reaction, the need for costly chiral separation steps is removed, which represents a significant portion of the total manufacturing cost for chiral intermediates. The ability to purify the product via recrystallization rather than preparative chromatography further reduces solvent usage and waste disposal costs. This streamlined approach allows for a much higher margin profile, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The use of robust, commercially available starting materials mitigates the risk of supply disruptions that can occur with proprietary or niche reagents. The reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment beyond standard low-temperature reactors, meaning the process can be easily transferred between different manufacturing sites. This flexibility ensures continuity of supply even in the face of regional logistical challenges. Additionally, the high yield and purity reduce the likelihood of batch failures, providing a more predictable and reliable delivery schedule for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without significant re-optimization. The avoidance of heavy metals simplifies the environmental compliance landscape, as there is no need for rigorous testing and removal of trace metal impurities to meet ICH Q3D guidelines. The reduced solvent intensity and the potential for solvent recovery in the crystallization steps contribute to a lower environmental footprint. This makes the technology particularly attractive for companies aiming to reduce their carbon emissions and adhere to stricter environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific technical details and beneficial effects described in the patent documentation. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this route into their existing portfolios. The answers provide clarity on the scope, limitations, and strategic benefits of the method.
Q: What are the key advantages of using sulfinylimines for this synthesis?
A: Using sulfinylimines allows for excellent stereocontrol during the nucleophilic addition, resulting in high diastereomeric ratios (dr up to 99:1) without the need for chiral chromatography.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes inexpensive raw materials like bromoform and avoids expensive transition metal catalysts, making it highly cost-effective and scalable for industrial production.
Q: What downstream applications do these compounds serve?
A: These chiral amines serve as versatile building blocks for synthesizing bioactive molecules, including precursors for 2,2-dibromo ethylenimines used in further asymmetric transformations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Tribromomethyl Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN102627590A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from clinical trials to full-scale market supply. Our facilities are equipped with state-of-the-art low-temperature reactors and rigorous QC labs capable of verifying the stringent purity specifications required for pharmaceutical applications. We are committed to delivering chiral alpha-tribromomethyl amines that meet the highest standards of quality and consistency, enabling our partners to accelerate their drug development timelines with confidence.
We invite you to engage with our technical team to explore how this technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel synthesis route. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this approach for your target molecules. Contact our technical procurement team today to discuss your requirements and secure a reliable supply of these critical building blocks for your next generation of therapeutics.
