Revolutionizing Carbonate Hydroboration: A Novel Lithium Catalyst for Efficient Boronate Ester Synthesis
Revolutionizing Carbonate Hydroboration: A Novel Lithium Catalyst for Efficient Boronate Ester Synthesis
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, safer, and cost-effective catalytic processes. A significant breakthrough in this domain is documented in patent CN111763226A, which introduces a novel hydroboration reaction method specifically designed for carbonates. This technology leverages a unique deprotonated phenyl-bridged β-ketimine lithium compound to catalyze the reduction of carbonates using borane, yielding valuable boronate esters. Unlike traditional methods that often rely on expensive transition metals or harsh conditions, this innovation utilizes a structurally distinct lithium cluster that offers exceptional stability and activity. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this represents a pivotal shift towards more sustainable and scalable manufacturing of key intermediates. The ability to operate under mild conditions with low catalyst loading addresses critical pain points in process chemistry, paving the way for streamlined production workflows.
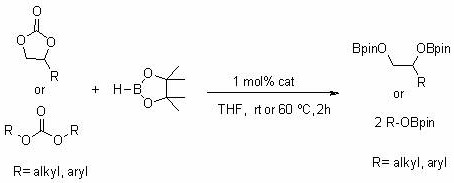
The core of this technological advancement lies in the transformation of relatively inert carbonate substrates into highly versatile boronate esters, which are indispensable building blocks in modern organic synthesis, particularly for Suzuki-Miyaura cross-coupling reactions. The patent details a robust protocol where carbonates such as ethylene carbonate, propylene carbonate, and dimethyl carbonate are effectively reduced. This process not only expands the toolbox available to synthetic chemists but also offers a reliable pathway for the commercial scale-up of complex fine chemical intermediates. By utilizing a catalyst that is easier to handle and store than conventional pyrophoric organolithium reagents, the method significantly reduces operational risks and infrastructure requirements, making it an attractive option for both laboratory-scale discovery and industrial-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroboration of esters and carbonates has been a challenging endeavor due to the inherent stability of the carbonyl group and steric hindrance associated with these substrates. Prior art has explored various catalytic systems, including main group metal magnesium complexes, transition metal manganese pincer complexes, and rare earth metal compounds. While some of these systems, such as manganese-based catalysts reported by Walter's group or magnesium catalysts by Rueping's group, have shown promise, they often come with significant drawbacks. Transition metal catalysts can be prohibitively expensive, suffer from metal contamination issues that require rigorous purification steps, and may exhibit limited substrate scope. Furthermore, many existing protocols require elevated temperatures or extended reaction times to achieve acceptable conversion rates. The reliance on sensitive reagents that demand stringent storage conditions, such as strict moisture exclusion and specialized ventilation, adds layers of complexity and cost to the manufacturing process. These factors collectively hinder the widespread adoption of carbonate hydroboration in cost-sensitive industries.
The Novel Approach
The methodology presented in patent CN111763226A offers a compelling alternative by introducing a specialized lithium-based catalytic system that overcomes many of these limitations. The key differentiator is the use of a tetrameric lithium cluster supported by a β-ketimine ligand, which provides a unique electronic and steric environment conducive to activating the carbonate bond. This novel approach allows the reaction to proceed efficiently at mild temperatures ranging from room temperature to 60°C, drastically reducing energy consumption compared to high-temperature alternatives. Moreover, the catalyst loading is remarkably low, typically around 1 mol%, yet it achieves yields exceeding 90% for a wide range of substrates. This high turnover efficiency translates directly into cost reduction in fine chemical intermediates manufacturing by minimizing the amount of catalyst required per batch. The structural integrity of the catalyst also ensures consistent performance, reducing the variability often seen with less stable catalytic species.
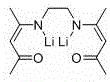
Mechanistic Insights into Lithium-Catalyzed Carbonate Hydroboration
Understanding the mechanistic underpinnings of this reaction is crucial for appreciating its superiority over conventional systems. The active catalyst, identified as [Lph’Li4(THF)4]2, features a tetrameric lithium core coordinated by deprotonated β-ketimine ligands. This multinuclear structure is believed to facilitate a cooperative activation mechanism where multiple lithium centers work in concert to polarize the carbonyl oxygen of the carbonate substrate. The β-ketimine ligand plays a pivotal role by modulating the electron density at the metal center through its conjugated system, thereby enhancing the nucleophilicity of the hydride source. The presence of THF molecules in the coordination sphere further stabilizes the complex, preventing aggregation into inactive species while maintaining sufficient openness for substrate binding. This delicate balance of stability and reactivity is what enables the catalyst to perform effectively under such mild conditions.
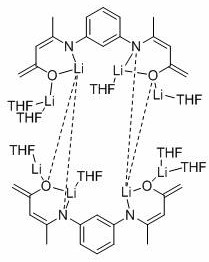
Furthermore, the mechanism likely involves a concerted insertion of the carbonate into the lithium-hydride bond, followed by transmetallation with the borane reagent. The unique geometry of the tetrameric cluster, as depicted in the structural analysis, prevents the formation of unreactive byproducts that often plague monomeric lithium systems. Comparative studies within the patent highlight that altering the ligand structure or the nuclearity of the lithium cluster can lead to a dramatic drop in activity, as evidenced by control experiments where alternative lithium structures yielded only 15% conversion under identical conditions. This underscores the importance of the specific [Lph’Li4(THF)4]2 architecture in achieving high catalytic turnover. For R&D teams, this implies that precise control over catalyst synthesis and structural integrity is paramount to replicating the high yields reported. The robustness of this mechanism against varying electronic properties of the carbonate substrates explains the broad applicability of the method across different alkyl and aryl carbonates.
How to Synthesize Boronate Esters Efficiently
The synthesis of boronate esters using this patented method is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins with the preparation of the catalyst itself, which involves the deprotonation of a readily available β-ketimine ligand using a small molecule organolithium reagent. Once the catalyst is prepared, it can be stored conveniently without the extreme precautions needed for simple organolithiums. The actual hydroboration reaction is performed by mixing the carbonate substrate, pinacolborane, and the catalyst in a suitable ether solvent like THF. The reaction proceeds rapidly under a nitrogen atmosphere, requiring minimal thermal input. Detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined in the comprehensive guide below to ensure reproducibility and optimal yield for your specific application.
- Prepare the catalyst by reacting m-phenylene-bridged beta-ketimine ligand with n-butyllithium in THF/hexane to form the tetrameric lithium complex.
- In an inert atmosphere, mix the carbonate substrate (e.g., ethylene carbonate) and pinacolborane with 1 mol% of the lithium catalyst in THF solvent.
- Stir the reaction mixture at mild temperatures (25-60°C) for approximately 2 hours, then terminate by exposure to air and purify via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage lies in the simplification of the supply chain for catalyst materials. Since the novel lithium catalyst can be synthesized from commercially available precursors and stored under ambient conditions in standard glassware, the logistical burden of sourcing and storing hazardous pyrophoric reagents is significantly alleviated. This ease of handling translates into reduced safety training costs and lower insurance premiums associated with hazardous material storage. Additionally, the high activity of the catalyst means that less material is needed to produce the same amount of product, optimizing inventory management and reducing the frequency of catalyst replenishment orders. The mild reaction conditions also imply that existing reactor infrastructure can be utilized without the need for expensive upgrades to high-pressure or high-temperature systems, facilitating a smoother transition from lab to plant.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive transition metals and the reduction of catalyst loading to just 1 mol%. By avoiding the use of precious metals like palladium or complex manganese pincer ligands, the raw material costs are substantially lowered. Furthermore, the high yields (>90%) minimize waste generation and the need for extensive purification steps, which are often the most costly part of fine chemical production. The ability to terminate the reaction simply by exposure to air also streamlines the quenching process, saving time and resources compared to complex workup procedures required for sensitive metal catalysts. These factors combine to create a leaner, more cost-effective manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of earth-abundant lithium and readily available organic ligands, reducing dependence on geopolitically sensitive transition metal supply chains. The stability of the catalyst allows for bulk preparation and long-term storage, ensuring that production schedules are not disrupted by catalyst degradation or delivery delays. This reliability is crucial for maintaining continuous production lines in the pharmaceutical and agrochemical industries, where downtime can be extremely costly. The robustness of the method across various substrates also means that a single catalyst system can be used for multiple products, simplifying procurement strategies and reducing the complexity of the chemical inventory.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the mild operating conditions and the absence of toxic heavy metals. The use of lithium, while requiring care, is generally more environmentally benign than many transition metal alternatives, simplifying waste disposal and regulatory compliance. The high atom economy of the hydroboration reaction, coupled with the ability to recover and recycle solvents like THF, aligns with green chemistry principles. This makes the process attractive for companies aiming to reduce their environmental footprint and meet increasingly stringent sustainability goals. The simplified purification process, often involving just filtration and vacuum distillation, further reduces the generation of hazardous waste streams.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and practical implementation of this carbonate hydroboration technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this process into your existing manufacturing workflows.
Q: What are the storage requirements for the novel lithium catalyst compared to traditional organolithium reagents?
A: Unlike traditional organolithium reagents like n-butyllithium which require strict ventilation and waterproofing, the deprotonated phenyl-bridged ketimine lithium compound disclosed in this patent can be stored simply in glass bottles within conventional reagent cabinets, significantly enhancing operational safety and ease of handling.
Q: How does the catalytic activity of this lithium complex compare to existing manganese or magnesium systems?
A: The novel lithium complex demonstrates superior activity, achieving yields over 90% with only 1 mol% loading at mild temperatures (25-60°C). In contrast, comparative examples with other lithium structures showed significantly lower yields (e.g., 15%), highlighting the unique efficiency of the tetrameric cluster structure.
Q: What types of carbonate substrates are compatible with this hydroboration method?
A: The method exhibits a broad substrate scope, successfully converting various carbonates including ethylene carbonate, propylene carbonate, dimethyl carbonate, dibenzyl carbonate, and 1,3-dioxan-2-one into their corresponding boronate esters with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronate Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN111763226A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methods into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity boronate esters that meet the stringent purity specifications required by the global pharmaceutical industry. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing consistency and reliability for our clients.
We invite you to explore how this novel hydroboration method can optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and conduct comprehensive route feasibility assessments to demonstrate the viability of this technology for your projects. Let us partner with you to drive innovation and efficiency in your chemical manufacturing operations.
