Scalable Synthesis of Vilazodone Intermediate Using Stable Aluminum Chloride Catalysts
Scalable Synthesis of Vilazodone Intermediate Using Stable Aluminum Chloride Catalysts
The global pharmaceutical landscape is continuously evolving to address critical mental health challenges, with depression remaining a leading cause of disability worldwide according to World Health Organization statistics. In this context, Vilazodone (Formula A), a dual-acting serotonin reuptake inhibitor and 5-HT1A receptor partial agonist, has emerged as a vital therapeutic agent approved by the FDA for treating major depressive disorder. 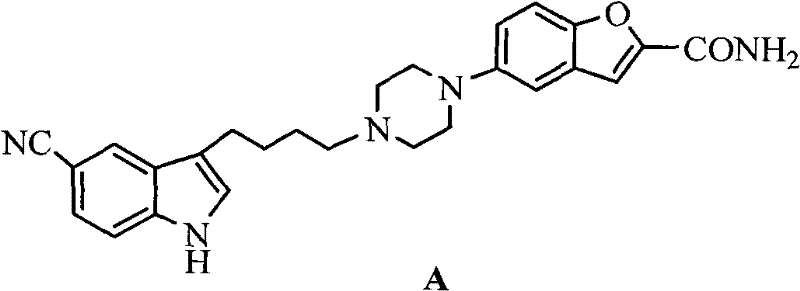 The commercial success of Vilazodone hinges on the efficient and cost-effective production of its key building blocks, specifically the intermediate 3-(4-chlorobutyl)-1H-5-cyanoindole. Patent CN102690224A introduces a groundbreaking preparation method that addresses longstanding bottlenecks in the supply chain of this critical pharmaceutical intermediate. By shifting away from hazardous and expensive catalysts towards stable, commodity-grade reagents, this technology offers a robust pathway for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The commercial success of Vilazodone hinges on the efficient and cost-effective production of its key building blocks, specifically the intermediate 3-(4-chlorobutyl)-1H-5-cyanoindole. Patent CN102690224A introduces a groundbreaking preparation method that addresses longstanding bottlenecks in the supply chain of this critical pharmaceutical intermediate. By shifting away from hazardous and expensive catalysts towards stable, commodity-grade reagents, this technology offers a robust pathway for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-(4-chlorobutyl)-1H-5-cyanoindole has been plagued by significant safety and efficiency hurdles inherent to traditional Friedel-Crafts acylation and reduction protocols. Prior art, such as the methods disclosed in patent DE433254 and literature from the Journal of Medicinal Chemistry, relied heavily on isobutyl aluminum chloride (i-BuAlCl2) as the primary Lewis acid catalyst. 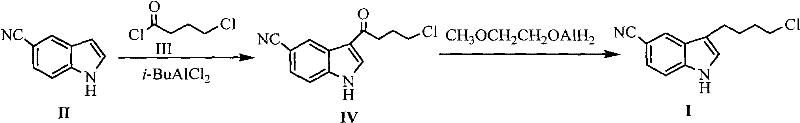 This reagent is chemically unstable and pyrophoric, meaning it ignites spontaneously upon contact with air, necessitating complex pipeline transportation systems and specialized reaction equipment to prevent catastrophic safety incidents. Furthermore, the reduction step in these conventional routes often utilized methoxyethoxy aluminum dihydride, which resulted in disappointingly low yields, reported as low as 26%, and required tedious column chromatography for purification. These factors collectively rendered the traditional processes economically unviable for large-scale industrial application, creating a fragile supply chain for high-purity pharmaceutical intermediates.
This reagent is chemically unstable and pyrophoric, meaning it ignites spontaneously upon contact with air, necessitating complex pipeline transportation systems and specialized reaction equipment to prevent catastrophic safety incidents. Furthermore, the reduction step in these conventional routes often utilized methoxyethoxy aluminum dihydride, which resulted in disappointingly low yields, reported as low as 26%, and required tedious column chromatography for purification. These factors collectively rendered the traditional processes economically unviable for large-scale industrial application, creating a fragile supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative methodology presented in patent CN102690224A fundamentally reengineers the synthetic pathway to eliminate these critical vulnerabilities. Instead of relying on hazardous organoaluminum compounds, the new process employs anhydrous aluminum trichloride (AlCl3) for the initial acylation step, followed by a reduction system utilizing a mixture of sodium borohydride and aluminum trichloride. 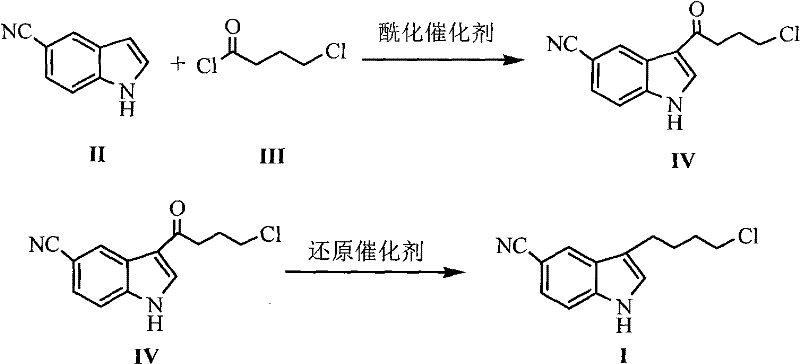 This strategic substitution transforms the reaction profile from a high-risk operation into a manageable, standard organic synthesis procedure. The novel approach not only mitigates the safety risks associated with pyrophoric materials but also dramatically improves the reaction efficiency, achieving yields up to 89% in optimized examples. By enabling purification through simple recrystallization rather than column chromatography, this method significantly reduces solvent consumption and processing time, offering a clear advantage for cost reduction in API manufacturing.
This strategic substitution transforms the reaction profile from a high-risk operation into a manageable, standard organic synthesis procedure. The novel approach not only mitigates the safety risks associated with pyrophoric materials but also dramatically improves the reaction efficiency, achieving yields up to 89% in optimized examples. By enabling purification through simple recrystallization rather than column chromatography, this method significantly reduces solvent consumption and processing time, offering a clear advantage for cost reduction in API manufacturing.
Mechanistic Insights into AlCl3-Catalyzed Acylation and Borohydride Reduction
The core of this technological advancement lies in the precise manipulation of Lewis acid catalysis and hydride reduction mechanisms. In the first step, anhydrous aluminum trichloride acts as a potent Lewis acid to activate the 4-chlorobutyryl chloride, facilitating a highly regioselective Friedel-Crafts acylation at the 3-position of the 5-cyanoindole ring. The reaction is carefully controlled at temperatures between -5°C and 5°C to prevent side reactions and ensure the formation of the 3-(4-chlorobutyryl)-1H-5-cyanoindole ketone intermediate with high fidelity. This step is crucial as it establishes the carbon skeleton required for the final antidepressant structure without compromising the sensitive cyano group on the indole ring.
The subsequent reduction step represents a masterclass in chemoselectivity and process safety. By generating a reactive hydride species in situ through the interaction of sodium borohydride and aluminum trichloride, the system effectively reduces the ketone carbonyl to a methylene group. This reduction is conducted at low temperatures, specifically between -10°C and -15°C, to maintain control over the exothermic nature of the reaction and prevent the reduction of other functional groups. The mechanism ensures that the chloro-butyl side chain remains intact, avoiding dehalogenation which is a common impurity in such reductions. This precise control over the impurity profile is essential for meeting the stringent purity specifications required for clinical-grade active pharmaceutical ingredients.
How to Synthesize 3-(4-Chlorobutyl)-1H-5-cyanoindole Efficiently
The implementation of this synthesis route requires strict adherence to the optimized parameters defined in the patent to maximize yield and safety. The process begins with the preparation of the acylation mixture under inert atmosphere, followed by the controlled addition of reagents to manage heat evolution. Detailed operational procedures regarding stoichiometry, solvent selection (preferably dichloromethane or dichloroethane), and quenching protocols are critical for reproducibility. For R&D teams looking to adopt this technology, the following standardized synthesis steps outline the critical path from raw materials to the purified intermediate, ensuring a smooth transition from laboratory scale to pilot production.
- Perform Friedel-Crafts acylation of 5-cyanoindole with 4-chlorobutyryl chloride using anhydrous AlCl3 in dichloromethane at 0°C to form the ketone intermediate.
- Reduce the ketone intermediate using a mixture of sodium borohydride and anhydrous AlCl3 in dichloromethane at -10°C to -15°C.
- Quench the reaction with ice water, extract with ethyl acetate, and purify the crude solid via recrystallization from isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from pyrophoric catalysts to stable aluminum salts represents a transformative opportunity to de-risk the supply of critical antidepressant intermediates. The elimination of isobutyl aluminum chloride removes the need for specialized storage facilities and hazardous material transport logistics, which are often bottlenecks in global chemical supply chains. This simplification directly translates to enhanced supply chain reliability, as the raw materials required—anhydrous AlCl3 and sodium borohydride—are commodity chemicals available from multiple global sources, reducing dependency on single-source suppliers of exotic reagents.
- Cost Reduction in Manufacturing: The replacement of expensive, difficult-to-handle organoaluminum catalysts with inexpensive, stable inorganic salts drives substantial cost savings in raw material procurement. Additionally, the ability to purify the final product via recrystallization rather than column chromatography significantly lowers solvent costs and waste disposal fees. This process intensification reduces the overall manufacturing footprint and energy consumption, contributing to a more sustainable and economically competitive production model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing reagents that do not require special pipeline transport or inert atmosphere storage beyond standard nitrogen blanketing, the logistical complexity of the supply chain is drastically reduced. This stability ensures consistent production schedules and minimizes the risk of shutdowns due to reagent availability issues or safety incidents. The robustness of the chemistry allows for flexible manufacturing planning, ensuring that downstream API production is not interrupted by upstream intermediate shortages.
- Scalability and Environmental Compliance: The simplified workup procedure, which avoids complex chromatographic separations, makes this route inherently scalable from kilogram to multi-ton quantities. The reduction in solvent usage and the avoidance of hazardous pyrophoric waste streams simplify environmental compliance and wastewater treatment processes. This alignment with green chemistry principles facilitates easier regulatory approval and supports the long-term sustainability goals of modern pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN102690224A, offering clarity on why this method is becoming the preferred choice for industrial production. Understanding these nuances is vital for technical teams evaluating process transfers and for commercial teams negotiating supply agreements for vilazodone precursors.
Q: Why is the new AlCl3/NaBH4 method superior to the conventional i-BuAlCl2 route?
A: The conventional method relies on isobutyl aluminum chloride, a pyrophoric Lewis acid that requires specialized pipeline transport and poses significant safety risks. The new method utilizes standard anhydrous aluminum chloride and sodium borohydride, which are stable, inexpensive, and easy to handle, drastically improving operational safety and reducing raw material costs.
Q: What yield improvements does patent CN102690224A offer over prior art?
A: Prior art methods, such as those described in Journal of Medicinal Chemistry (2004), reported yields as low as 26% and required complex column chromatography for purification. The patented process achieves yields up to 89% through optimized reaction conditions and allows for simple recrystallization, significantly enhancing overall process efficiency.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the route is specifically designed for industrial scalability. By eliminating the need for hazardous pyrophoric reagents and replacing column chromatography with recrystallization, the process simplifies equipment requirements and waste treatment, making it highly viable for commercial-scale production of antidepressant intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Chlorobutyl)-1H-5-cyanoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of life-saving medications like Vilazodone. Our technical team has extensively analyzed the advantages of the AlCl3-mediated reduction pathway and possesses the expertise to implement this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of intermediates that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, guaranteeing the quality required for subsequent API synthesis steps.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their antidepressant portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient and safe chemical foundations available in the market.
