Revolutionizing Montelukast Intermediate Production with Advanced Asymmetric Hydrogenation Technology
Revolutionizing Montelukast Intermediate Production with Advanced Asymmetric Hydrogenation Technology
The pharmaceutical industry constantly seeks more efficient, sustainable, and cost-effective pathways for synthesizing complex active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in patent CN103420908A, which discloses a novel preparation method for a critical chiral intermediate used in the synthesis of Montelukast Sodium, a widely prescribed leukotriene receptor antagonist for asthma management. This technology represents a paradigm shift from traditional stoichiometric reductions to a highly efficient catalytic asymmetric hydrogenation process. By leveraging a specialized iridium-based chiral catalyst system, this method addresses long-standing challenges regarding catalyst loading, optical purity, and overall yield. For R&D directors and procurement strategists, understanding this technological leap is essential for optimizing supply chains and reducing the cost of goods sold (COGS) for respiratory therapeutics. The patent demonstrates that through precise ligand design and reaction engineering, it is possible to overcome the inherent coordination issues posed by the substrate's heterocyclic nitrogen and olefinic bonds, achieving yields exceeding 90% and optical purities greater than 98%.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key chiral alcohol intermediate for Montelukast has relied heavily on stoichiometric chiral reducing agents, most notably derivatives of boron such as (-)-DIP-Cl or complexes derived from (-)-α-pinene. These conventional approaches suffer from significant intrinsic drawbacks that hinder large-scale manufacturing efficiency. Firstly, the substrate molecule contains both a carbon-carbon double bond and a quinoline nitrogen atom, which act as potent ligands that coordinate strongly with the chiral catalyst. This coordination effectively poisons the catalyst, necessitating the use of massive excesses—often up to 1.8 equivalents relative to the substrate—to drive the reaction to completion. This stoichiometric requirement not only inflates raw material costs exponentially but also generates substantial quantities of boron-containing waste, complicating downstream purification and environmental compliance. Furthermore, reagents like (-)-DIP-Cl are notoriously moisture-sensitive and corrosive, requiring stringent anhydrous conditions and specialized handling equipment, which increases operational complexity and safety risks. Reported yields for these older methods often struggle to exceed 68-87%, with optical purity frequently requiring additional recrystallization steps to meet pharmaceutical standards.
The Novel Approach
In stark contrast, the methodology outlined in CN103420908A introduces a robust catalytic asymmetric hydrogenation strategy that fundamentally alters the economic and technical landscape of this synthesis. The core innovation lies in the utilization of a cationic iridium complex bearing a chiral P,N-ligand (specifically a DTB-type ligand structure). This catalyst system exhibits remarkable tolerance to the coordinating functional groups present in the substrate, allowing the reaction to proceed with catalytic rather than stoichiometric quantities of the chiral source. The patent highlights mole ratios of catalyst to substrate as low as 1:50,000, a staggering improvement over the 1:1.8 ratio of prior art. The process involves reacting the ketone precursor (Formula II) under hydrogen pressure (0.2 to 10 MPa) in the presence of a mild base and the iridium catalyst. This approach not only simplifies the reaction setup by using common solvents like ethanol or methanol but also delivers superior performance metrics. As demonstrated in the experimental data, this novel route consistently achieves yields above 90% and optical purities surpassing 98% ee, often reaching near-perfect stereocontrol without the need for extensive downstream purification.
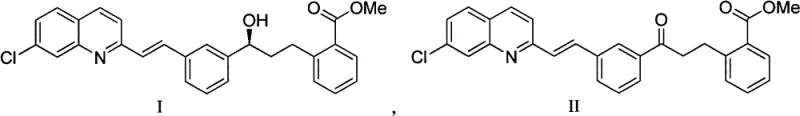
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this transformation hinges on the unique electronic and steric properties of the iridium-DTB catalyst system. Unlike boron-based reagents that rely on a four-centered transition state susceptible to interference, the iridium catalyst operates through a coordination-insertion mechanism that is kinetically favored even in the presence of potential catalyst poisons. The bulky di-tert-butylphosphino (DTB) group on the ligand creates a highly congested chiral pocket around the metal center. This steric bulk effectively shields the active site, preventing the quinoline nitrogen and the vinyl double bond of the substrate from binding in a manner that would deactivate the catalyst. Instead, the ketone carbonyl is selectively coordinated and reduced. The mechanism likely involves the oxidative addition of hydrogen to the Ir(I) center to form an Ir(III) dihydride species, followed by the coordination of the ketone substrate. Subsequent migratory insertion of the hydride into the carbonyl bond, directed by the chiral environment of the ligand, establishes the stereocenter with high fidelity. Finally, reductive elimination releases the chiral alcohol product and regenerates the active catalyst species, allowing the cycle to turnover thousands of times.
Impurity control is another critical aspect where this mechanistic understanding translates to practical value. In conventional boron reductions, side reactions often lead to the formation of over-reduced byproducts or regioisomers (such as Formula III mentioned in the background art) due to the lack of chemoselectivity. The iridium catalyst described here demonstrates exceptional chemoselectivity for the ketone functionality over the conjugated alkene and the heterocyclic ring. This selectivity minimizes the formation of structural impurities, resulting in a cleaner crude reaction profile. The high enantioselectivity (>98% ee) observed suggests that the energy difference between the diastereomeric transition states leading to the (R) and (S) alcohols is substantial, driven by the rigid conformation of the P,N-ligand framework. This inherent selectivity reduces the burden on crystallization processes, ensuring that the final API meets stringent regulatory specifications for chiral purity with minimal material loss.
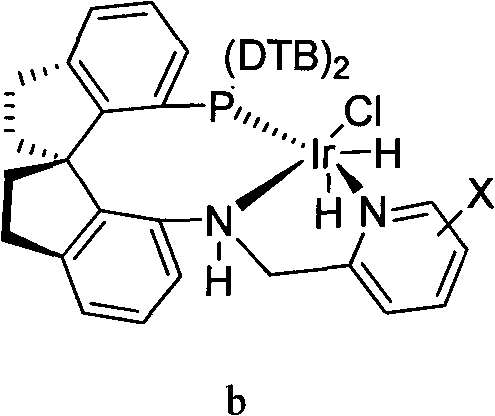
How to Synthesize (S)-Methyl 2-(3-(2-(7-chloroquinolin-2-yl)vinyl)phenyl)-3-hydroxypropyl benzoate Efficiently
The synthesis of this high-value chiral intermediate is streamlined into a manageable unit operation suitable for modern multipurpose reactors. The process begins with the careful charging of the ketone substrate, the iridium catalyst, and an alkali base into a pressure vessel under an inert atmosphere. The choice of base, such as potassium tert-butoxide or sodium ethylate, plays a vital role in activating the catalyst precursor and maintaining the reaction pH. Following the addition of the solvent system, typically an alcohol like ethanol or methanol, the reactor is sealed and pressurized with hydrogen. The reaction is then heated to moderate temperatures, generally around 50°C, and stirred until hydrogen uptake ceases, indicating complete conversion. Workup involves standard aqueous extraction and drying, yielding the crude chiral alcohol which can be further purified by recrystallization if necessary to boost optical purity to >99% ee.
- Prepare the reaction vessel under nitrogen protection and charge with the ketone substrate (Formula II), a specialized iridium chiral catalyst (Formula a or b), and a suitable alkali base such as potassium tert-butoxide.
- Add an alcoholic solvent system, typically ethanol or methanol, potentially mixed with polar aprotic solvents like DMF to enhance solubility and reaction kinetics.
- Pressurize the autoclave with hydrogen gas to 0.2-10 MPa and maintain the temperature between 0-80°C until conversion is complete, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenation technology offers transformative benefits that extend far beyond simple yield improvements. The shift from stoichiometric chiral auxiliaries to a catalytic system fundamentally reshapes the cost structure of the manufacturing process. By eliminating the need for expensive, moisture-sensitive boron reagents that were previously required in nearly double the molar amount of the substrate, the direct material costs are drastically reduced. Furthermore, the simplified waste stream, devoid of heavy boron loads, lowers the costs associated with hazardous waste disposal and environmental remediation. This process intensification allows manufacturers to produce high-purity pharmaceutical intermediates with a significantly smaller physical footprint and reduced utility consumption, aligning perfectly with green chemistry initiatives and corporate sustainability goals.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic reduction in catalyst loading. Moving from a stoichiometric requirement (1.8 equivalents) to a catalytic one (parts per million levels) decouples production costs from the price volatility of chiral starting materials. Additionally, the use of commodity chemicals like hydrogen gas and ethanol as reagents and solvents replaces specialized, high-cost reagents. This transition ensures a more predictable and stable cost of goods, protecting margins against supply shocks in the fine chemical sector. The high yield (>90%) also means less raw material is wasted per kilogram of product, further enhancing overall process economics.
- Enhanced Supply Chain Reliability: Relying on stoichiometric chiral boranes often creates supply chain bottlenecks, as these reagents have limited shelf lives and require cold chain logistics due to their sensitivity. In contrast, the iridium catalyst system described is more robust, and the primary reductant (hydrogen) is universally available at industrial scales. This reliability reduces the risk of production delays caused by reagent degradation or supply shortages. Moreover, the simplified reaction conditions reduce the dependency on highly specialized operators, making it easier to transfer technology between manufacturing sites or contract manufacturing organizations (CMOs) without compromising quality.
- Scalability and Environmental Compliance: Scaling up reactions involving pyrophoric or corrosive boron reagents presents significant engineering challenges and safety hazards. The hydrogenation approach described here utilizes standard high-pressure reactor technology that is commonplace in the fine chemical industry, facilitating seamless scale-up from pilot plant to commercial tonnage. From an environmental perspective, the atom economy of hydrogenation is superior, generating water as the primary byproduct rather than stoichiometric amounts of boron waste. This cleaner profile simplifies regulatory filings and ensures long-term compliance with increasingly strict environmental regulations regarding heavy metal and chemical waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims within patent CN103420908A, providing a factual basis for evaluating the process viability. Understanding these details is crucial for technical teams assessing the feasibility of integrating this route into existing manufacturing workflows.
Q: How does this iridium-catalyzed method improve upon traditional boron-based reductions?
A: Traditional methods using stoichiometric chiral boranes like (-)-DIP-Cl require excessive amounts (1.8 equivalents) due to catalyst poisoning by the substrate's nitrogen and double bonds. This new iridium method operates at catalytic levels (ratios as low as 1:50,000), drastically reducing material costs and waste while achieving superior optical purity (>98% ee).
Q: What are the critical reaction conditions for maintaining high enantioselectivity?
A: Maintaining strict control over hydrogen pressure (typically 1.4-3.2 MPa in examples) and temperature (around 50°C) is crucial. The choice of base, such as potassium tert-butoxide or sodium ethylate, and the specific substitution on the DTB ligand (e.g., 3-methyl vs 3-methoxy) significantly influences the reaction rate and final ee value.
Q: Is this process scalable for industrial API production?
A: Yes, the process is highly scalable. It utilizes standard hydrogenation equipment (autoclaves) and common solvents like ethanol and methanol. The elimination of moisture-sensitive and corrosive boron reagents simplifies the safety profile and waste treatment, making it ideal for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Intermediate Supplier
The technological advancements detailed in CN103420908A underscore the immense potential of modern catalytic methods to redefine the production of complex pharmaceutical building blocks. At NINGBO INNO PHARMCHEM, we recognize that translating such patent innovations into commercial reality requires not just chemical expertise, but robust engineering capabilities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, ensuring that every batch of Montelukast intermediate meets the highest global standards for chirality and chemical purity.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and sustainable synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can secure your supply chain and optimize your production costs for this critical respiratory medicine intermediate.
