Advanced Synthetic Route for Dihydrosafrole: Enhancing Purity and Scalability for Global Supply Chains
The global demand for high-purity agrochemical and fragrance intermediates necessitates robust synthetic methodologies that balance efficiency with environmental compliance. Patent CN102070596A introduces a refined preparation method for dihydrosafrole, a critical precursor for the insecticide synergist piperonyl butoxide, addressing longstanding limitations in traditional synthesis routes. This technology leverages a two-step sequence involving Friedel-Crafts acylation followed by a modified Wolff-Kishner-Huang Minglong reduction, achieving a total yield of approximately 84% under mild conditions. For R&D directors and procurement specialists, this patent represents a pivotal shift away from hazardous reagents like mercury amalgams towards safer, scalable catalytic systems using zinc chloride or ferric chloride. The implementation of this process not only enhances the purity profile of the final product but also aligns with modern green chemistry principles by operating at atmospheric pressure and utilizing readily available petrochemical feedstocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of dihydrosafrole has been plagued by significant technical and environmental hurdles that compromise supply chain reliability and operational safety. Traditional methods often relied on the direct reduction of safrole extracted from sassafras roots, a practice that is increasingly unsustainable due to vegetation destruction and regulatory restrictions on natural resource exploitation. Alternative synthetic routes, such as those disclosed in earlier patents, frequently employed Clemmensen reduction utilizing zinc amalgam, which introduces severe toxicity concerns and complex waste disposal challenges for manufacturing facilities. Furthermore, catalytic hydrogenation pathways, while effective, demand high-pressure equipment and rigorous safety protocols, escalating capital expenditure and limiting the flexibility of production sites. These conventional approaches often suffer from moderate yields, typically hovering around 77% or lower, and involve corrosive catalysts like perchloric acid that pose significant risks to equipment integrity and personnel safety.
The Novel Approach
The methodology outlined in CN102070596A offers a transformative solution by replacing hazardous reduction steps with a sophisticated Wolff-Kishner-Huang Minglong protocol enhanced by polyethylene glycol-400. This novel approach initiates with the Friedel-Crafts acylation of 1,3-benzodioxole using propionyl chloride, catalyzed by safer Lewis acids such as zinc chloride or ferric chloride in chlorinated solvents. The subsequent reduction step eliminates the need for high-pressure hydrogenation or toxic mercury, instead utilizing hydrazine hydrate and potassium hydroxide in aromatic solvents like toluene or xylene. 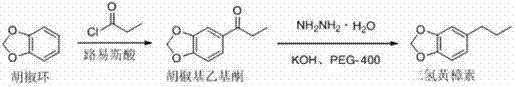 This strategic modification allows the reaction to proceed under atmospheric pressure with exceptional control over impurity formation. The integration of PEG-400 serves as a crucial phase transfer facilitator, ensuring efficient contact between the organic hydrazone intermediate and the inorganic base, thereby driving the decomposition reaction to completion with yields exceeding 90% in the second step. This comprehensive redesign of the synthetic pathway directly addresses the pain points of cost reduction in fine chemical intermediates manufacturing by simplifying equipment requirements and minimizing hazardous waste generation.
This strategic modification allows the reaction to proceed under atmospheric pressure with exceptional control over impurity formation. The integration of PEG-400 serves as a crucial phase transfer facilitator, ensuring efficient contact between the organic hydrazone intermediate and the inorganic base, thereby driving the decomposition reaction to completion with yields exceeding 90% in the second step. This comprehensive redesign of the synthetic pathway directly addresses the pain points of cost reduction in fine chemical intermediates manufacturing by simplifying equipment requirements and minimizing hazardous waste generation.
Mechanistic Insights into ZnCl2-Catalyzed Acylation and PEG-Assisted Reduction
The first stage of this synthesis relies on the precise electrophilic aromatic substitution of 1,3-benzodioxole, where the methylenedioxy group directs the incoming acyl group to the para-position relative to the oxygen atoms. The use of zinc chloride or ferric chloride as Lewis acid catalysts provides a balanced activation of the propionyl chloride, generating the acylium ion necessary for the attack on the electron-rich aromatic ring without inducing excessive polymerization or side reactions common with stronger catalysts like aluminum chloride. Maintaining the reaction temperature between 0°C and 5°C is critical for kinetic control, preventing polyacylation and ensuring the formation of piperonyl ethyl ketone with high regioselectivity. The stoichiometry is carefully managed with a slight excess of propionyl chloride (1.1 to 1.3 molar equivalents) to drive the conversion of the starting material to completion, as monitored by gas chromatography, ensuring minimal residual starting material carries over to the next stage.
In the second stage, the mechanism shifts to the formation and subsequent decomposition of a hydrazone intermediate, a process significantly accelerated by the presence of polyethylene glycol-400. The initial condensation of piperonyl ethyl ketone with hydrazine hydrate forms the corresponding hydrazone, releasing water which is removed via azeotropic distillation with the solvent to shift the equilibrium forward. Upon addition of potassium hydroxide and PEG-400, the system undergoes a base-catalyzed decomposition where the hydrazone loses nitrogen gas to form the final alkane product, dihydrosafrole. The PEG-400 molecules likely coordinate with potassium ions, increasing the solubility and nucleophilicity of the hydroxide ions in the organic phase, which lowers the activation energy for the proton abstraction steps required for nitrogen extrusion. This mechanistic nuance explains the observed high yields and purity levels, as the enhanced reaction kinetics reduce the residence time at elevated temperatures, thereby minimizing thermal degradation and the formation of tarry byproducts that often complicate downstream purification.
How to Synthesize Dihydrosafrole Efficiently
The operational protocol derived from this patent provides a clear roadmap for scaling this chemistry from laboratory benchtop to industrial reactor vessels. The process begins with the controlled addition of propionyl chloride to a cooled mixture of 1,3-benzodioxole and the Lewis acid catalyst in dichloroethane, requiring precise thermal management to maintain the exothermic acylation within the 0-5°C window. Following the isolation of the intermediate ketone, the workflow transitions to the reduction phase where the ketone is refluxed with hydrazine hydrate in toluene, followed by the critical addition of the base and phase transfer catalyst for the decomposition step. Detailed standardized synthesis steps see the guide below.
- Perform Friedel-Crafts acylation of 1,3-benzodioxole with propionyl chloride using ZnCl2 or FeCl3 catalyst at 0-5°C to obtain piperonyl ethyl ketone.
- Conduct Wolff-Kishner-Huang Minglong reduction by reacting the ketone with hydrazine hydrate in toluene, followed by decomposition with KOH and PEG-400.
- Isolate the final dihydrosafrole product through phase separation, washing, and solvent removal to achieve yields exceeding 84%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits that extend beyond simple yield metrics. By transitioning from plant-extracted safrole to fully synthetic petrochemical derivatives, manufacturers can decouple their production schedules from agricultural volatility, ensuring a consistent and reliable agrochemical intermediate supplier status regardless of seasonal harvest fluctuations. The elimination of high-pressure hydrogenation equipment reduces the barrier to entry for contract manufacturing organizations, allowing for more flexible capacity allocation and faster response times to market demand surges. Furthermore, the avoidance of mercury-based reagents drastically simplifies environmental compliance and waste treatment protocols, leading to substantial cost savings in effluent management and regulatory reporting.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous catalysts with commodity chemicals like zinc chloride and ferric chloride significantly lowers the raw material cost base for each production batch. Additionally, the ability to conduct the reduction step at atmospheric pressure eliminates the need for specialized high-pressure autoclaves, reducing both capital investment and maintenance overheads associated with pressure vessel certification. The high atom economy of the acylation step, combined with the efficient recovery of solvents like toluene and dichloroethane, further optimizes the overall cost structure, making the final dihydrosafrole product highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing 1,3-benzodioxole and propionyl chloride from established petrochemical supply chains ensures a steady flow of raw materials that is immune to the geopolitical and climatic risks associated with botanical sourcing. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without significant yield loss, contributes to higher batch success rates and predictable delivery timelines. This reliability is crucial for downstream customers producing piperonyl butoxide, as it minimizes the risk of production stoppages due to intermediate shortages, thereby strengthening the entire value chain resilience.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex fragrance intermediates, utilizing standard glass-lined or stainless steel reactors commonly found in fine chemical plants. The absence of heavy metal contaminants in the final product streamlines the purification process, often requiring only simple washing and distillation rather than complex chromatographic separations. From an environmental perspective, the shift away from zinc amalgam and perchloric acid aligns with increasingly stringent global regulations on hazardous substances, future-proofing the manufacturing site against potential regulatory bans and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dihydrosafrole synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process capabilities and product specifications. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production portfolios.
Q: Why is the Wolff-Kishner-Huang Minglong reduction preferred over Clemmensen reduction for dihydrosafrole?
A: The Wolff-Kishner-Huang Minglong reduction avoids the use of highly toxic zinc amalgam and strong acids required in Clemmensen reduction, significantly improving environmental compliance and operator safety while maintaining high yields under atmospheric pressure.
Q: What role does PEG-400 play in the reduction step of this synthesis?
A: Polyethylene glycol-400 acts as a phase transfer catalyst and stabilizer during the hydrazone decomposition, facilitating the reaction between the organic substrate and the inorganic base (KOH), which enhances reaction efficiency and simplifies post-reaction workup.
Q: How does this synthetic route impact the supply stability of piperonyl butoxide precursors?
A: By utilizing petrochemical-derived starting materials like 1,3-benzodioxole and propionyl chloride instead of plant-extracted safrole, this method eliminates dependency on agricultural harvests, ensuring consistent year-round production capacity and stable pricing for agrochemical manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrosafrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic technologies to meet the evolving needs of the global agrochemical and fragrance industries. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering high-purity dihydrosafrole that meets stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical instrumentation to verify every batch against exacting standards. Our facility is equipped to handle the specific solvent systems and thermal profiles required by this optimized route, guaranteeing consistent quality and supply continuity for our partners.
We invite prospective clients to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits of switching to this mercury-free, atmospheric pressure process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency and product competitiveness in the global marketplace.
