Advanced Synthesis of Dihydropyrazole Sulfonamide Derivatives for Oncology Applications
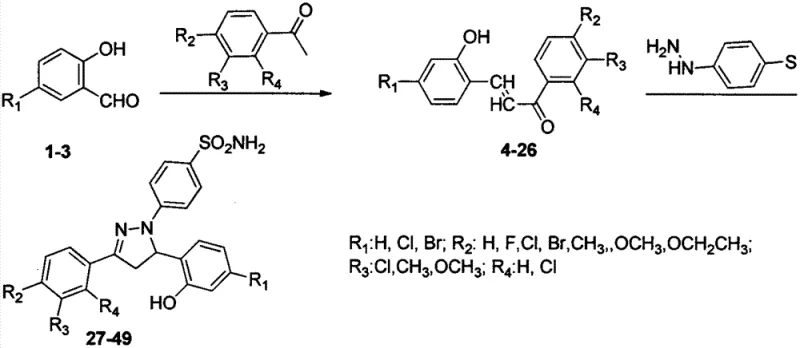
The pharmaceutical industry is constantly seeking robust synthetic pathways for novel heterocyclic scaffolds that demonstrate potent biological activity, particularly in the realm of oncology. Patent CN103664786A discloses a significant advancement in the synthesis of dihydropyrazole sulfonamide derivatives derived from salicylaldehyde. This technology represents a strategic convergence of two pharmacologically privileged structures: the pyrazole ring, known for its anti-inflammatory and analgesic properties, and the sulfonamide moiety, a cornerstone in medicinal chemistry with established antibacterial and diuretic applications. By integrating these motifs, the disclosed method generates a library of compounds (27-49) that exhibit marked inhibitory effects against human breast cancer (MCF-7) and lung cancer (A549) cell lines. For R&D directors and procurement managers alike, understanding the nuances of this synthetic route is critical for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials for preclinical and clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex pyrazole-sulfonamide hybrids often suffer from multi-step inefficiencies and harsh reaction conditions that compromise overall yield and purity. Conventional methods may require expensive transition metal catalysts or extreme temperatures that degrade sensitive functional groups, leading to complex impurity profiles that are difficult to separate. Furthermore, the lack of modularity in older synthetic routes often restricts the ability to rapidly generate structural analogs for structure-activity relationship (SAR) studies. This rigidity can significantly delay drug discovery timelines and increase the cost of goods sold (COGS) due to low atom economy and extensive purification requirements. For supply chain heads, these factors translate into unpredictable lead times and potential bottlenecks in the production of critical API intermediates.
The Novel Approach
The methodology outlined in the patent offers a streamlined, two-step strategy that overcomes these historical hurdles by utilizing a Claisen-Schmidt condensation followed by an acid-catalyzed cyclocondensation. This approach leverages readily available starting materials—substituted salicylaldehydes and acetophenones—to first construct a chalcone intermediate (4-26), which then serves as the electrophilic partner for p-hydrazinobenzenesulfonamide. The reaction conditions are notably mild, typically employing ethanol as a solvent and acetic acid as a catalyst under reflux, which minimizes energy consumption and equipment corrosion. This operational simplicity facilitates cost reduction in pharmaceutical intermediates manufacturing by reducing the need for specialized high-pressure reactors or exotic reagents. The modular nature of the substituents (R1-R4) allows for the rapid diversification of the chemical library, enabling researchers to fine-tune biological activity without overhauling the entire synthetic process.
Mechanistic Insights into Acid-Catalyzed Cyclocondensation
The core of this synthetic innovation lies in the efficient formation of the 4,5-dihydropyrazole ring through a nucleophilic addition-elimination sequence. In the second step of the synthesis, the hydrazine nitrogen of p-hydrazinobenzenesulfonamide acts as a nucleophile, attacking the beta-carbon of the alpha,beta-unsaturated ketone system within the chalcone intermediate. This Michael-type addition is facilitated by the acidic environment provided by acetic acid, which activates the carbonyl group and stabilizes the transition state. Following the initial addition, an intramolecular cyclization occurs where the second nitrogen atom of the hydrazine moiety attacks the carbonyl carbon, resulting in the closure of the five-membered pyrazoline ring. This mechanism is highly regioselective, ensuring that the sulfonamide group remains intact and positioned correctly on the phenyl ring attached to the nitrogen atom, which is crucial for the observed anticancer activity.
Impurity control is inherently managed through the thermodynamic stability of the final crystalline product and the solubility differences between the target molecule and the byproducts. The reaction mixture typically contains unreacted chalcone, excess hydrazine, and oligomeric side products. However, the protocol specifies purification via recrystallization from absolute ethanol or column chromatography, which effectively separates the target dihydropyrazole sulfonamide based on polarity and crystal lattice energy. The presence of the hydroxyl group on the salicylaldehyde-derived portion of the molecule also allows for hydrogen bonding interactions that can be exploited during purification to enhance purity. For quality assurance teams, this predictable crystallization behavior ensures that stringent purity specifications can be met consistently, reducing the risk of batch failures during commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Dihydropyrazole Sulfonamide Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the preparation of the chalcone backbone, followed by the critical cyclization step that installs the pharmacophore. Operators must pay close attention to stoichiometric ratios, particularly the slight excess of hydrazine used to drive the equilibrium towards product formation. Monitoring the reaction progress via Thin Layer Chromatography (TLC) is essential to determine the optimal endpoint, preventing over-reaction or decomposition. Detailed standardized synthesis steps see the guide below.
- Perform Claisen-Schmidt condensation between substituted salicylaldehydes and acetophenones using an alkali solution in an organic solvent to generate chalcone intermediates.
- React the chalcone intermediate with p-hydrazinobenzenesulfonamide in the presence of an acid catalyst (such as acetic acid) and ethanol under reflux conditions.
- Isolate the crude dihydropyrazole product via filtration and purify through recrystallization or column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that align with the goals of modern pharmaceutical supply chains, focusing on reliability, cost-efficiency, and scalability. The elimination of precious metal catalysts and the use of commodity chemicals like ethanol and acetic acid drastically simplify the procurement process and reduce raw material volatility risks. Moreover, the reaction operates at atmospheric pressure and moderate temperatures, which lowers the barrier for technology transfer from R&D to manufacturing facilities. This accessibility means that multiple qualified suppliers can potentially adopt the process, enhancing supply chain resilience and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, bulk-available starting materials and solvents. By avoiding complex catalytic systems and high-energy inputs, the operational expenditure (OPEX) is significantly minimized. The purification strategy relies on standard filtration and recrystallization rather than expensive preparative HPLC, which further drives down the cost per kilogram. Additionally, the high atom economy of the condensation reactions ensures that waste generation is kept to a minimum, reducing disposal costs and environmental compliance burdens.
- Enhanced Supply Chain Reliability: The modular design of the synthesis allows for flexible sourcing of raw materials. Since the R1, R2, R3, and R4 substituents can be varied independently, manufacturers can adapt to supply shortages of specific substituted benzaldehydes or acetophenones by switching to alternative analogs without changing the core process parameters. This flexibility ensures continuous production even when specific precursor availability fluctuates. Furthermore, the robustness of the reaction conditions means that minor variations in utility supply (e.g., steam pressure) are less likely to cause batch deviations, ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: Scaling this synthesis from grams to tons is straightforward due to the absence of exothermic runaways or hazardous reagents. The use of ethanol as a primary solvent is favorable from a green chemistry perspective, as it is less toxic and easier to recover than chlorinated solvents. The solid products are easily isolated by filtration, simplifying downstream processing and drying operations. This ease of scale-up supports the transition from clinical trial material to commercial API production, ensuring that the supply chain can meet increasing demand without requiring major capital investment in new reactor types.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dihydropyrazole sulfonamide derivatives. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary biological application of these dihydropyrazole sulfonamide derivatives?
A: According to patent CN103664786A, these derivatives exhibit significant inhibitory activity against human breast cancer cells (MCF-7) and lung cancer cells (A549), making them valuable candidates for antineoplastic drug development.
Q: What are the critical starting materials required for this synthesis?
A: The synthesis relies on three main components: substituted salicylaldehydes, various substituted acetophenones to form the chalcone backbone, and p-hydrazinobenzenesulfonamide for the final cyclization step.
Q: How does this method address impurity control in large-scale manufacturing?
A: The process utilizes standard purification techniques such as filtration and recrystallization from ethanol. The use of well-defined stoichiometric ratios and TLC monitoring ensures that unreacted hydrazine and chalcone precursors are effectively removed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyrazole Sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the identity and potency of every batch, guaranteeing that the dihydropyrazole sulfonamide derivatives you receive meet the highest industry standards for research and development.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these valuable anticancer intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can lower your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments to ensure that our capabilities align perfectly with your development timeline and quality requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
