Scalable Total Synthesis of Tenuifoliside B for Neuroprotective Drug Development
Introduction to the Novel Synthetic Pathway
The pharmaceutical industry is constantly seeking reliable sources for bioactive natural products that exhibit potent therapeutic effects, particularly in the realm of neuroprotection. Patent CN103694282A introduces a groundbreaking chemical synthesis method for Tenuifoliside B, a sugar ester compound originally isolated from the traditional Chinese medicine Polygala tenuifolia Willd. This natural product has demonstrated significant efficacy in improving memory deficits induced by scopolamine and sodium cyanide in rat models, showing activity comparable to tacrine. Historically, the supply of Tenuifoliside B has been severely constrained by its low natural abundance, with extraction yields hovering around a mere 0.1%, necessitating the processing of massive quantities of plant material to obtain milligram amounts. This new synthetic approach fundamentally shifts the paradigm from unsustainable extraction to a robust, six-step chemical transformation, utilizing sucrose, syringaldehyde, and p-hydroxybenzoic acid as foundational building blocks. By establishing a fully synthetic route, this technology addresses the critical bottleneck of supply continuity, offering a viable pathway for the commercial scale-up of complex pharmaceutical intermediates required for anti-senile dementia drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the procurement of Tenuifoliside B relied exclusively on isolation from plant sources, a method fraught with inherent inefficiencies and supply chain vulnerabilities. The primary constraint lies in the exceedingly low concentration of the target compound within the host plant, Polygala tenuifolia, which contains approximately 0.1% of the active ingredient. This scarcity translates into an exorbitant cost of goods sold, as tons of raw botanical material must be harvested, processed, and purified to yield negligible amounts of the final product. Furthermore, plant extraction is subject to seasonal variations, geographical inconsistencies, and the presence of numerous structurally similar impurities that complicate downstream purification. Reports indicate that isolating merely 18 mg of Tenuifoliside B required processing 9 kg of plant material, highlighting the impracticality of this method for industrial-scale manufacturing. These factors collectively create a fragile supply chain that cannot support the rigorous demands of clinical trials or commercial drug production, leading to significant lead time delays and unpredictable availability for research and development teams.
The Novel Approach
In stark contrast to the limitations of extraction, the novel synthetic methodology described in the patent offers a streamlined, high-efficiency alternative that leverages abundant commodity chemicals. By initiating the synthesis with sucrose, syringaldehyde, and p-hydroxybenzoic acid, the process bypasses the biological variability of plant sources entirely. The route is designed with operational simplicity in mind, featuring mild reaction conditions that are easily implementable in standard chemical manufacturing facilities. A key advantage of this approach is the high conversion rate observed in the initial stages, particularly the first three steps, which proceed with minimal by-product formation. Crucially, the intermediates generated in these early stages possess sufficient purity to be telescoped directly into subsequent reactions without the need for resource-intensive fine column chromatography. This strategic omission of complex purification steps not only accelerates the overall production timeline but also significantly reduces solvent consumption and waste generation, aligning with modern green chemistry principles while ensuring a consistent and scalable supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into the Multi-Step Synthesis
The synthetic strategy is elegantly divided into the construction of two distinct acyl donors followed by their sequential attachment to the sucrose core. The first phase involves the protection and functionalization of syringaldehyde. Syringaldehyde is initially subjected to acetylation using acetic anhydride in pyridine under reflux conditions for 2 hours, yielding 4-acetoxy-3,5-dimethoxybenzaldehyde. This protected aldehyde then undergoes a Knoevenagel condensation with malonic acid, catalyzed by piperidine in a benzene/pyridine solvent system. This step extends the carbon chain to form the cinnamic acid derivative, a critical structural motif for the final molecule. Subsequently, this acid is activated via dehydration using triphosgene and triethylamine in anhydrous ethyl acetate at 0°C, generating the reactive mixed anhydride intermediate. This anhydride serves as a potent acylating agent, primed for nucleophilic attack by the hydroxyl groups of the sugar moiety. The precision in temperature control during the anhydride formation, specifically maintaining 0°C for 10 minutes followed by room temperature reaction, is vital to prevent decomposition and ensure high selectivity.
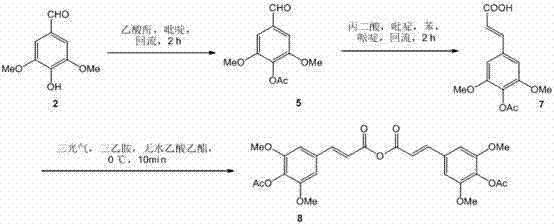
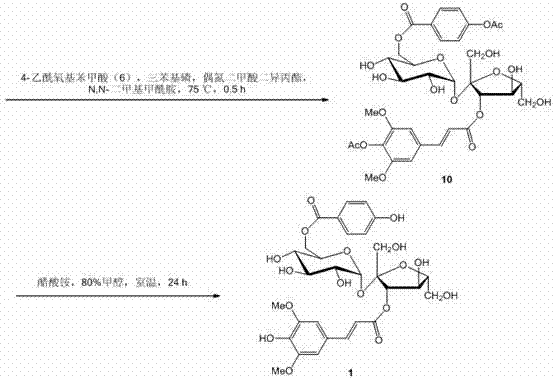
The second phase of the mechanism focuses on the regioselective glycosylation and final assembly. The sucrose molecule, possessing multiple hydroxyl groups, requires careful control to achieve the specific 3',6-di-O-acylation pattern found in Tenuifoliside B. The first esterification occurs at the 3'-position using the previously synthesized cinnamic anhydride, facilitated by cobalt chloride and triethylamine in DMF. This metal-catalyzed step demonstrates remarkable regioselectivity, favoring the primary or secondary hydroxyls as dictated by the steric environment of the sucrose scaffold. Following this, the 6-position is functionalized via a Mitsunobu reaction with 4-acetoxybenzoic acid. This classic transformation utilizes triphenylphosphine and diisopropyl azodicarboxylate (DIAD) to invert the stereochemistry and form the ester bond under thermal conditions at 75°C. The final step involves the global deprotection of the acetyl groups using ammonium acetate in 80% methanol at room temperature. This mild hydrolysis condition is essential to preserve the sensitive glycosidic linkage of the sucrose core while cleanly removing the protecting groups to reveal the bioactive phenolic hydroxyls, resulting in the target molecule with a total yield of approximately 22.3%.
How to Synthesize Tenuifoliside B Efficiently
Executing this synthesis requires strict adherence to the specified molar ratios and reaction parameters to maximize yield and minimize impurity profiles. The process begins with the parallel preparation of the two acid components, ensuring that the 4-acetoxy-3,5-dimethoxycinnamic anhydride and 4-acetoxybenzoic acid are ready for the coupling stages. The esterification of sucrose is particularly sensitive to moisture, necessitating the use of anhydrous solvents and reagents. The cobalt chloride catalyst plays a pivotal role in directing the acylation to the desired position on the sucrose ring, and deviations in the catalyst loading can lead to mixtures of regioisomers that are difficult to separate. Similarly, the Mitsunobu reaction requires precise stoichiometry, with a molar ratio of substrate to acid of 1:2.5 to 1:3.0, to drive the reaction to completion given the steric hindrance at the 6-position. The final deprotection step is remarkably forgiving, proceeding at room temperature over 24 hours, which simplifies the operational complexity of the final stage. For detailed operational procedures and specific workup protocols, please refer to the standardized guide below.
- Protect syringaldehyde and p-hydroxybenzoic acid with acetic anhydride to form acetoxy intermediates.
- Perform Knoevenagel condensation on the protected aldehyde to generate the cinnamic acid derivative.
- Dehydrate the cinnamic acid using triphosgene to form the corresponding anhydride.
- Esterify sucrose with the cinnamic anhydride using cobalt chloride catalysis.
- Conduct a Mitsunobu reaction with 4-acetoxybenzoic acid to attach the second acyl group.
- Remove acetyl protecting groups using ammonium acetate in methanol to yield final Tenuifoliside B.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to this synthetic route represents a substantial opportunity for cost optimization and risk mitigation. The reliance on plant extraction introduced significant volatility due to agricultural dependencies, whereas this chemical synthesis utilizes commodity chemicals that are available in bulk quantities year-round. The elimination of the need for fine column chromatography in the early stages of the synthesis drastically reduces the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical manufacturing. Furthermore, the mild reaction conditions, such as room temperature esterification and ambient deprotection, lower the energy requirements for heating and cooling, contributing to a reduced carbon footprint and lower utility costs. The robustness of the process, evidenced by the ability to telescope intermediates without purification, shortens the overall cycle time, allowing for faster throughput and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive starting materials like sucrose and syringaldehyde, which are produced on a multi-ton scale globally. By avoiding the expensive and labor-intensive isolation processes associated with natural product extraction, manufacturers can achieve significant cost savings per kilogram of the final API intermediate. The high yield of the initial steps and the ability to bypass intermediate purification further compound these savings by reducing material loss and labor hours. Additionally, the avoidance of transition metal catalysts in the final deprotection step, using simple ammonium acetate instead, eliminates the need for costly heavy metal scavenging resins and the associated analytical testing for residual metals, streamlining the quality control budget.
- Enhanced Supply Chain Reliability: Diversifying the source of Tenuifoliside B from a single botanical origin to a synthetic chemical process enhances supply security. Agricultural supply chains are susceptible to weather events, pests, and geopolitical instability, whereas the chemical supply chain for the requisite reagents is well-established and resilient. The synthesis does not rely on any rare or conflict minerals, ensuring ethical sourcing compliance. The scalability of the reaction conditions, which do not require extreme pressures or cryogenic temperatures beyond standard ice baths, means that production can be easily ramped up in existing multipurpose reactors without significant capital expenditure on specialized equipment. This flexibility ensures that supply can be scaled to meet the needs of clinical trials and eventual commercial launch without long lead times for equipment fabrication.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry initiatives, which is increasingly important for regulatory compliance and corporate sustainability goals. The solvents used, such as ethanol, methanol, and ethyl acetate, are generally regarded as safer and more environmentally friendly compared to chlorinated solvents often used in complex natural product synthesis. The high atom economy of the condensation and esterification steps minimizes waste generation. Furthermore, the mild deprotection conditions reduce the generation of hazardous acidic or basic waste streams. The ability to produce the compound with a total yield of roughly 22.3% through a concise six-step sequence demonstrates high efficiency, reducing the overall environmental burden per unit of product produced. This makes the process attractive for companies aiming to reduce their Scope 3 emissions and meet stringent environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Tenuifoliside B synthesized via this patented method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the feasibility and advantages of this route. Understanding these details is crucial for R&D teams evaluating the material for preclinical studies and for procurement teams assessing the long-term viability of the supply source. The answers reflect the specific improvements in yield, purity, and operational simplicity that distinguish this synthetic method from traditional extraction techniques.
Q: Why is chemical synthesis preferred over plant extraction for Tenuifoliside B?
A: Plant extraction yields are extremely low (approximately 0.1% content in Polygala tenuifolia), making it difficult to meet production demands. Chemical synthesis offers a controllable total yield of about 22.3% and uses readily available raw materials.
Q: What are the key starting materials for this synthesis?
A: The process utilizes three inexpensive and commercially available starting materials: sucrose, syringaldehyde, and p-hydroxybenzoic acid, ensuring a stable and cost-effective supply chain.
Q: Does the synthesis require complex purification steps?
A: No, the first three steps of the reaction sequence produce intermediates with high conversion rates and few by-products, allowing them to be used directly in subsequent steps without fine column chromatography purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenuifoliside B Supplier
As a leading CDMO and fine chemical manufacturer, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthetic technology for your neuroprotective drug development programs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from gram-scale research to industrial manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling to support regulatory filings. We understand the critical nature of API intermediates in the drug development timeline and are committed to providing a stable, high-quality supply of Tenuifoliside B that meets the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this synthetic route can be optimized for your specific volume requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this synthetic supply chain. We encourage you to request specific COA data and route feasibility assessments to validate the compatibility of our material with your downstream processes. Let us collaborate to accelerate the development of next-generation therapies for cognitive disorders, backed by a supply chain that is both robust and economically efficient.
