Revolutionizing 2-Bromofluorenone Production with Green Aqueous Catalytic Oxidation Technology
Revolutionizing 2-Bromofluorenone Production with Green Aqueous Catalytic Oxidation Technology
The landscape of fine chemical synthesis for advanced electronic materials is undergoing a significant transformation, driven by the urgent need for sustainable manufacturing processes that do not compromise on product quality. A pivotal advancement in this domain is detailed in patent CN108276261B, which discloses a novel method for preparing 2-bromofluorenone through molecular oxygen oxidation catalyzed by transition metal Salen complexes in an aqueous phase. This technology represents a paradigm shift away from the reliance on hazardous organic solvents, offering a pathway to produce high-purity intermediates essential for the electroluminescent industry. By utilizing water as the reaction medium and air as the oxidant, this process addresses critical environmental and safety concerns while maintaining exceptional catalytic efficiency. For global supply chain leaders and R&D directors, understanding the mechanistic depth and commercial viability of this aqueous oxidation route is paramount for securing a competitive edge in the production of next-generation display materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
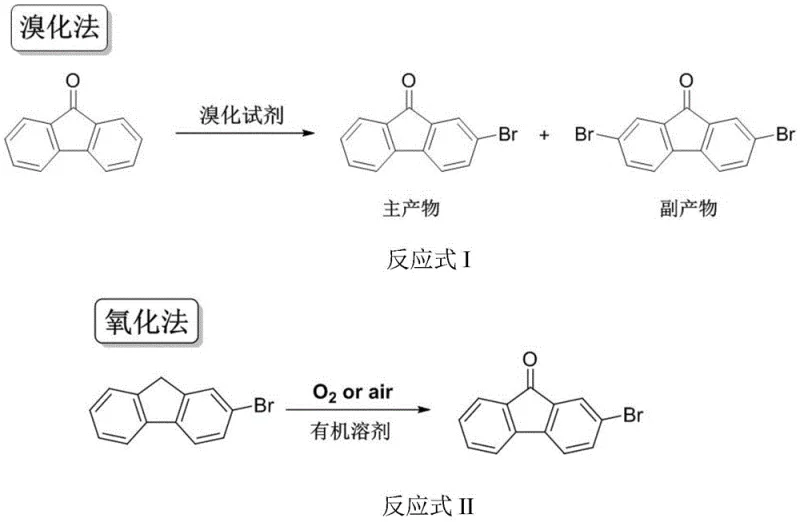
Historically, the synthesis of 2-bromofluorenone has been plagued by significant technical and environmental hurdles inherent to traditional bromination and oxidation protocols. The conventional bromination method, which utilizes fluorenone and brominating agents, generates harmful hydrogen bromide gas as a byproduct, necessitating complex scrubbing systems and posing severe safety risks to personnel. Furthermore, this route invariably produces 2,7-dibromofluorenone as a difficult-to-remove impurity due to its poor solubility, which drastically complicates downstream purification and limits the final product's suitability for high-performance optical applications. Alternatively, existing oxidation methods relying on organic solvents such as pyridine, tetrahydrofuran, or N,N-dimethylformamide introduce their own set of challenges, including high toxicity, flammability, and the generation of substantial volumes of hazardous waste that require expensive treatment. These legacy processes are increasingly untenable in a regulatory environment that demands stricter adherence to green chemistry principles and energy conservation policies.
The Novel Approach
In stark contrast to these legacy methodologies, the innovative aqueous phase oxidation technique described in the patent offers a streamlined and environmentally benign alternative that fundamentally redefines the production workflow. By employing a transition metal Salen complex catalyst within a water-based medium, the reaction proceeds efficiently under air bubbling conditions at moderate temperatures ranging from 60 to 100°C. This approach completely eliminates the need for volatile organic compounds (VOCs), thereby removing the risks associated with solvent storage, recovery, and disposal. The simplicity of the workup procedure, which involves merely filtering the solid product from the aqueous mixture, stands in sharp relief against the multi-step extractions and distillations required by organic solvent-based routes. Moreover, the ability to recycle the aqueous mother liquor multiple times without significant loss of catalytic activity underscores the economic and ecological superiority of this new method.
Mechanistic Insights into Transition Metal Salen-Catalyzed Aqueous Oxidation
The core of this technological breakthrough lies in the unique coordination chemistry of the transition metal Salen complexes, which serve as highly active catalysts capable of activating molecular oxygen in a polar aqueous environment. The Salen ligand, formed by the condensation of salicylaldehyde and ethylenediamine, creates a robust tetradentate pocket that stabilizes various transition metal centers such as Manganese, Cobalt, Iron, Nickel, or Rhodium. In the aqueous phase, these metal centers facilitate the transfer of oxygen atoms to the 2-bromofluorene substrate through a radical or concerted mechanism that is surprisingly efficient despite the hydrophobic nature of the organic substrate. The presence of water does not inhibit the catalytic cycle; rather, it appears to enhance the selectivity of the oxidation, preventing over-oxidation or side reactions that typically plague organic phase oxidations. This mechanistic resilience allows the reaction to proceed with high turnover numbers, ensuring that even low loadings of the catalyst can drive the conversion to near completion.
From an impurity control perspective, the aqueous medium plays a critical role in suppressing the formation of poly-brominated byproducts that are common in electrophilic aromatic substitution pathways. Since the reaction relies on oxidation rather than halogenation, the risk of generating 2,7-dibromofluorenone is inherently minimized, leading to a much cleaner crude product profile. The solubility characteristics of the reaction components in water further aid in purification; the product precipitates out of the solution upon cooling or simply remains as a solid that can be easily filtered, leaving soluble inorganic salts and catalyst residues in the mother liquor. This intrinsic separation capability reduces the reliance on chromatographic purification, which is often a bottleneck in scaling up the production of electronic grade intermediates. Consequently, the final product achieves purity levels exceeding 99.8%, meeting the rigorous specifications required for OLED and electroluminescent device fabrication.
How to Synthesize 2-Bromofluorenone Efficiently
The operational simplicity of this synthesis route makes it highly attractive for immediate adoption in pilot and commercial plants, requiring only standard glass-lined or stainless steel reactors equipped with reflux condensers and gas spargers. The process begins by charging the reactor with the substrate 2-bromofluorene and the selected transition metal Salen catalyst, followed by the addition of water as the sole reaction medium. The mixture is then heated to the optimal temperature range while air is bubbled through the solution to maintain a constant supply of the oxidant.
- Charge the reactor with 2-bromofluorene, transition metal Salen catalyst (0.01-1 wt%), and water as the sole medium.
- Heat the mixture to 60-100°C while mechanically stirring and bubbling air or oxygen through the solution for 8-24 hours.
- Upon completion, cool the reaction, filter the solid product directly, and retain the aqueous mother liquor for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aqueous oxidation technology translates into tangible strategic benefits that extend far beyond simple regulatory compliance. The elimination of expensive and hazardous organic solvents like DMF and THF removes a significant variable cost component from the manufacturing budget, while simultaneously simplifying the logistics of raw material sourcing and storage. Without the need for complex solvent recovery distillation columns or hazardous waste incineration services, the overall operational expenditure is drastically reduced, allowing for more competitive pricing structures in the global market. Furthermore, the robustness of the catalyst system and the reusability of the water phase create a more resilient supply chain that is less susceptible to fluctuations in solvent availability or price spikes in the petrochemical sector.
- Cost Reduction in Manufacturing: The economic impact of switching to a water-based system is profound, primarily driven by the complete removal of organic solvent procurement and recovery costs. Traditional methods require vast quantities of solvents that must be purchased, stored under strict safety conditions, and subsequently distilled for reuse or disposed of as hazardous waste, all of which incur substantial financial penalties. By replacing these with tap water or deionized water, the direct material costs are slashed, and the energy consumption associated with heating and distilling volatile organics is virtually eliminated. Additionally, the high catalytic efficiency means that precious metal loading can be kept minimal, further optimizing the cost of goods sold without sacrificing reaction throughput or yield.
- Enhanced Supply Chain Reliability: Relying on water as a reaction medium significantly de-risks the supply chain by removing dependence on petrochemical-derived solvents that are subject to market volatility and geopolitical supply disruptions. The raw materials for this process, specifically 2-bromofluorene and the Salen catalyst precursors, are readily available and stable, ensuring a consistent flow of production inputs. The ability to recycle the aqueous mother liquor for 20 to 30 cycles means that the facility operates with a closed-loop water system, minimizing freshwater intake and wastewater discharge permits, which are often major bottlenecks for chemical manufacturing expansion in regulated regions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is straightforward because it avoids the engineering complexities associated with handling large volumes of flammable or toxic solvents. The reaction conditions are mild, operating at atmospheric pressure with air, which reduces the need for high-pressure rated vessels and specialized safety infrastructure. From an environmental standpoint, the process aligns perfectly with green chemistry mandates, producing negligible hazardous waste and no VOC emissions, thereby facilitating faster regulatory approvals and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous oxidation technology, derived directly from the experimental data and claims within the patent literature. These insights are intended to clarify the operational parameters and quality outcomes expected when adopting this synthesis route for commercial production.
Q: What are the environmental advantages of this aqueous oxidation method?
A: Unlike traditional methods using toxic organic solvents like DMF or pyridine, this process uses water, eliminating VOC emissions and hazardous waste disposal costs.
Q: Can the catalyst and solvent system be recycled?
A: Yes, the patent demonstrates that the aqueous mother liquor can be reused 20-30 times after replenishing fresh catalyst, significantly reducing raw material consumption.
Q: What purity levels can be achieved with this synthesis route?
A: The method yields 2-bromofluorenone with exceptional purity, reaching up to 99.8% as analyzed by liquid chromatography, suitable for high-end electroluminescent applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromofluorenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the shift towards greener, more efficient synthesis routes is not just a regulatory requirement but a strategic imperative for maintaining leadership in the electronic materials sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and robust. We are committed to delivering 2-bromofluorenone with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the exacting standards required for high-performance electroluminescent applications. Our infrastructure is designed to support the continuous improvement of catalytic processes, allowing us to offer clients a supply partner that is both technically agile and commercially reliable.
We invite forward-thinking partners to engage with our technical procurement team to discuss how this innovative aqueous oxidation method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and supply security.
