Optimized Manufacturing of 1-Mercaptomethylcyclopropyl Acetic Acid for Global Pharmaceutical Supply Chains
Optimized Manufacturing of 1-Mercaptomethylcyclopropyl Acetic Acid for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical active pharmaceutical ingredient (API) intermediates, particularly those serving high-volume therapeutic areas such as respiratory health. Patent CN114835614A introduces a significant technological advancement in the preparation of 1-mercaptomethylcyclopropylacetic acid, a pivotal building block for the blockbuster asthma medication Montelukast Sodium. This novel process addresses long-standing inefficiencies in legacy manufacturing routes by implementing a concise, three-step linear synthesis that bypasses complex cyclization strategies. By leveraging straightforward sulfonylation and thiourea displacement chemistries, the technology offers a compelling value proposition for R&D teams focused on impurity profiling and supply chain leaders demanding consistent, high-yield production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-mercaptomethylcyclopropylacetic acid has been plagued by convoluted reaction sequences that negatively impact overall process economics and environmental footprint. Prior art, such as the route disclosed in Chinese patent CN105541786B, relies on a cumbersome strategy involving the formation of a spiro-cycle followed by a subsequent ring-opening step. This approach not only extends the total number of synthetic operations but also inherently generates significant quantities of structural by-products due to the instability of the cyclic intermediates. Furthermore, alternative methods reported in PCT patent WO9518107 utilize cyclic sulfites which suffer from notoriously low ring-opening yields and present severe challenges in downstream purification, ultimately leading to poor atom economy and increased waste disposal costs for manufacturers.
The Novel Approach
In stark contrast to these legacy methodologies, the process detailed in CN114835614A streamlines the synthesis into a highly efficient three-stage protocol starting directly from 1-hydroxymethylcyclopropylacetonitrile. The innovation lies in the strategic activation of the hydroxyl group via sulfonylation, followed by a clean nucleophilic displacement with thiourea, and a final hydrolytic cleavage. This linear progression eliminates the thermodynamic barriers associated with ring strain management found in older routes. As illustrated in the general reaction scheme below, the pathway maintains the integrity of the cyclopropane ring throughout the sequence, thereby minimizing side reactions and maximizing the recovery of the valuable chiral or achiral scaffold required for Montelukast assembly.
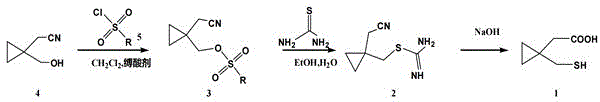
Mechanistic Insights into Sulfonylation-Thiourea Displacement Strategy
The core of this technological breakthrough rests on the precise execution of the sulfonylation step, where the hydroxyl functionality of the starting nitrile is converted into a superior leaving group. By reacting 1-hydroxymethylcyclopropylacetonitrile with substituted sulfonyl chlorides (such as p-toluenesulfonyl chloride) in the presence of an acid scavenger like triethylamine, the process generates a highly reactive sulfonate ester intermediate. This activation is crucial because it primes the molecule for the subsequent nucleophilic attack by thiourea, a reaction that proceeds via an SN2 mechanism to install the sulfur moiety with high regioselectivity. The use of thiourea is particularly advantageous as it acts as a soft nucleophile that effectively displaces the sulfonate group without inducing elimination reactions that could degrade the sensitive cyclopropane ring system.
Following the formation of the isothiourea salt, the final transformation involves alkaline hydrolysis which serves a dual purpose: it cleaves the C-S bond to release the free thiol and simultaneously hydrolyzes the nitrile group to the corresponding carboxylic acid. This tandem transformation is kinetically favorable under the specified basic conditions (using NaOH), allowing for the concurrent generation of both functional groups required in the final API intermediate. The mechanistic elegance of this route ensures that impurity profiles remain clean, as the reaction conditions are mild enough to prevent polymerization of the thiol or racemization of adjacent stereocenters, providing a distinct advantage for producing high-purity pharmaceutical grades suitable for regulatory submission.
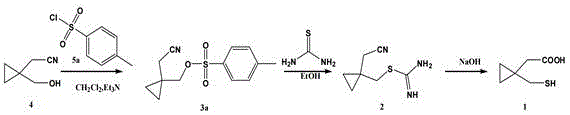
How to Synthesize 1-Mercaptomethylcyclopropylacetic Acid Efficiently
The implementation of this synthesis requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent examples. The process begins with the activation of the alcohol in a non-protic solvent like dichloromethane, followed by a reflux step in ethanol for the sulfur incorporation, and concludes with an aqueous workup. Each stage has been optimized to balance reaction rate with selectivity, ensuring that the final isolation of the mercapto-acid is achieved with minimal loss. For detailed operational parameters, including specific molar ratios and temperature ramps that guarantee reproducibility at scale, please refer to the standardized synthesis guide provided below.
- Sulfonylation of 1-hydroxymethylcyclopropylacetonitrile using substituted sulfonyl chloride in dichloromethane with an acid binding agent.
- Nucleophilic substitution reaction with thiourea in ethanol under reflux conditions to form the isothiourea intermediate.
- Alkaline hydrolysis using sodium hydroxide solution followed by acidification to isolate the final 1-mercaptomethylcyclopropylacetic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into enhanced operational resilience and cost efficiency. By reducing the total number of unit operations and eliminating the need for specialized cyclic precursors, the manufacturing timeline is significantly compressed, allowing for faster turnaround times on purchase orders. The reliance on commodity chemicals such as thiourea and common sulfonyl chlorides mitigates the risk of raw material shortages that often plague supply chains dependent on exotic or custom-synthesized reagents. Furthermore, the simplified purification protocols reduce the consumption of solvents and energy, aligning with modern sustainability goals while driving down the overall cost of goods sold (COGS) for the finished intermediate.
- Cost Reduction in Manufacturing: The elimination of the ring-closing and ring-opening steps found in conventional routes removes entire categories of reagent costs and waste treatment fees. By avoiding the formation of difficult-to-separate by-products, the process reduces the burden on purification infrastructure, leading to substantial savings in chromatography media and distillation energy. The high yield observed in each individual step compounds to deliver a superior overall mass balance, ensuring that more of the expensive starting nitrile is converted into saleable product rather than lost to side reactions.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. Since the reaction conditions do not require extreme temperatures or pressures, the process can be easily transferred between different manufacturing sites without significant re-validation efforts. This flexibility allows for a diversified supply base, reducing the risk of single-source dependency and ensuring that global demand for Montelukast intermediates can be met even during regional disruptions.
- Scalability and Environmental Compliance: The use of standard solvents like dichloromethane and ethanol, combined with aqueous workups, makes this process highly amenable to scale-up from pilot plant to multi-ton commercial production. The waste streams generated are primarily saline and organic solvent mixtures which are well-understood and easily managed within existing environmental compliance frameworks. This ease of waste handling simplifies the permitting process for new production lines and supports the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 1-mercaptomethylcyclopropylacetic acid using this advanced methodology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: How does this new process improve upon traditional Montelukast intermediate synthesis?
A: Unlike conventional methods that require complex ring-closing and ring-opening sequences which generate unnecessary by-products, this patented route utilizes a direct linear three-step strategy. This significantly simplifies the operational workflow, reduces raw material waste, and eliminates the difficult separation steps associated with cyclic sulfite intermediates.
Q: What are the critical quality control parameters for this intermediate?
A: The process emphasizes high conversion rates in the sulfonylation step (over 90% yield in optimized examples) and efficient hydrolysis. Critical parameters include maintaining precise pH levels during the acidification workup (pH 3.5-4.0) and ensuring complete removal of sulfur-containing by-products through rigorous extraction and washing protocols.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the methodology is specifically designed for scalability. It employs common, cost-effective reagents such as thiourea and p-toluenesulfonyl chloride, avoids cryogenic conditions or exotic catalysts, and utilizes standard unit operations like reflux and liquid-liquid extraction, making it ideal for multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Mercaptomethylcyclopropylacetic Acid Supplier
As the global demand for respiratory therapeutics continues to grow, securing a stable source of high-quality intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 1-mercaptomethylcyclopropylacetic acid we deliver adheres to the highest international standards for safety and efficacy.
We invite you to engage with our technical team to discuss how this optimized synthesis route can be integrated into your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are available to provide specific COA data and comprehensive route feasibility assessments, helping you make informed decisions that drive value and efficiency in your Montelukast manufacturing programs.
