Scalable Synthesis of High-Efficiency Chiral Foldamer Fluorescent Compounds for Advanced Optoelectronics
The landscape of chiral luminescent materials is undergoing a significant transformation driven by the demand for advanced optoelectronic components. Patent CN110357880B introduces a groundbreaking class of chiral fluorescent compounds based on a foldamer architecture, specifically designed to exhibit robust circularly polarized luminescence (CPL) properties. This technology addresses critical limitations in existing small-molecule emitters, offering a controllable emission wavelength and a remarkably high fluorescence quantum yield that can exceed 65%. For R&D directors and procurement specialists in the electronic chemical sector, this represents a pivotal shift towards more efficient and stable OLED materials. The asymmetry factor of the circularly polarized emission reaches the order of 10^-3, a significant benchmark for practical application in optical storage devices and chiral sensors. By leveraging a rigid quinoline-based backbone functionalized with chiral oxazole units, the invention ensures that the chirality is effectively transferred to the excited state, resulting in strong CPL signals without the need for expensive lanthanide complexes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral fluorescent materials has been hindered by reliance on supramolecular self-assembly or coordination with rare-earth metals. These conventional approaches often suffer from instability under operational conditions, as the non-covalent interactions driving the chirality can be easily disrupted by solvent changes or thermal stress. Furthermore, lanthanide-based systems, while effective, introduce significant cost burdens and supply chain vulnerabilities due to the scarcity of raw materials. Organic small molecules have been explored as alternatives, but they typically exhibit low quantum yields and weak circular polarization asymmetry factors, limiting their utility in high-performance displays. The lack of structural rigidity in many traditional organic emitters leads to non-radiative decay pathways, drastically reducing efficiency. Consequently, there has been a persistent need for a covalent strategy that locks the chiral conformation while maintaining high photoluminescence efficiency.
The Novel Approach
The methodology disclosed in CN110357880B overcomes these hurdles through a sophisticated foldamer design that integrates chirality directly into the molecular skeleton. By constructing a helical or folded structure via covalent bonds, the compound maintains its chiral integrity regardless of the environment. The introduction of alkynyl groups at specific positions on the quinoline core enhances the conjugation length, which is crucial for red-shifting the emission and improving the quantum yield. This structural modification allows for precise tuning of the CPL signal intensity and color simply by varying the R3 substituent, ranging from hydrogen to complex aromatic systems like pyrene. 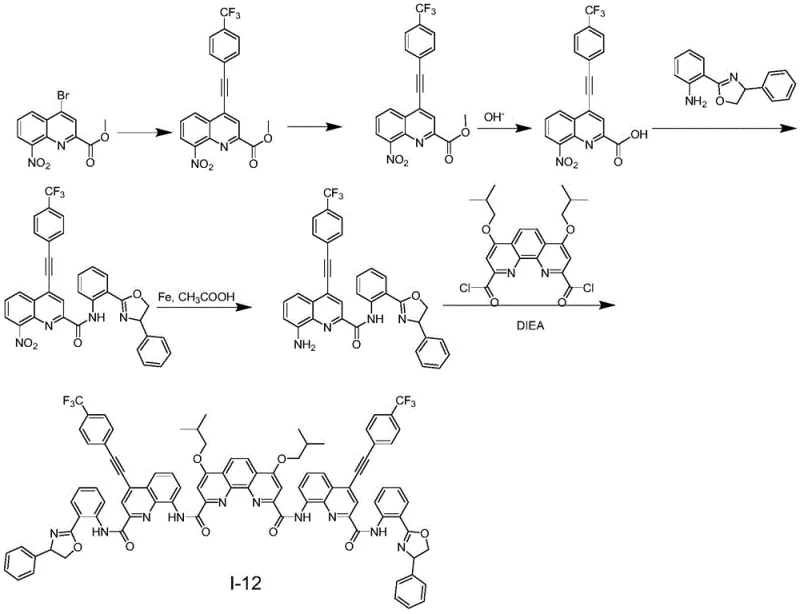 This versatility makes the material exceptionally suitable for cost reduction in electronic chemical manufacturing, as a single synthetic platform can generate a library of emitters for different color gamuts without redesigning the entire process flow.
This versatility makes the material exceptionally suitable for cost reduction in electronic chemical manufacturing, as a single synthetic platform can generate a library of emitters for different color gamuts without redesigning the entire process flow.
Mechanistic Insights into Sonogashira Coupling and Foldamer Assembly
The core of this synthesis relies on a palladium-catalyzed Sonogashira cross-coupling reaction, which serves as the foundation for building the conjugated pi-system. In the initial step, methyl 8-nitro-4-bromoquinoline-2-carboxylate reacts with terminal alkynes, such as trimethylsilylacetylene or substituted phenylacetylenes, in the presence of a palladium catalyst and copper iodide. This reaction is performed under inert atmosphere conditions to prevent oxidation of the sensitive intermediates. The choice of solvent, typically a mixture of anhydrous tetrahydrofuran and triethylamine, is critical for solubilizing the reactants and facilitating the catalytic cycle. Following the coupling, a deprotection step using tetrabutylammonium fluoride removes the silyl protecting group if necessary, revealing the terminal alkyne for further functionalization. This sequence establishes the rigid rod-like segment of the foldamer, which is essential for the subsequent folding behavior.
Subsequent steps involve the strategic formation of amide bonds to link the quinoline units with chiral amine auxiliaries. The carboxylic acid derivative, obtained via hydrolysis of the ester, is activated using coupling reagents like HATU and reacted with chiral aminophenyl oxazoles. This step is pivotal as it introduces the stereogenic center that dictates the handedness of the final foldamer. The reduction of the nitro group to an amine using iron powder in acetic acid is another key transformation, converting the electron-withdrawing nitro group into an electron-donating amine, which significantly alters the electronic properties of the quinoline ring. Finally, the diamine intermediate undergoes condensation with 1,10-diisobutoxyphenanthroline-2,9-dicarbonyl chloride to close the foldamer structure. 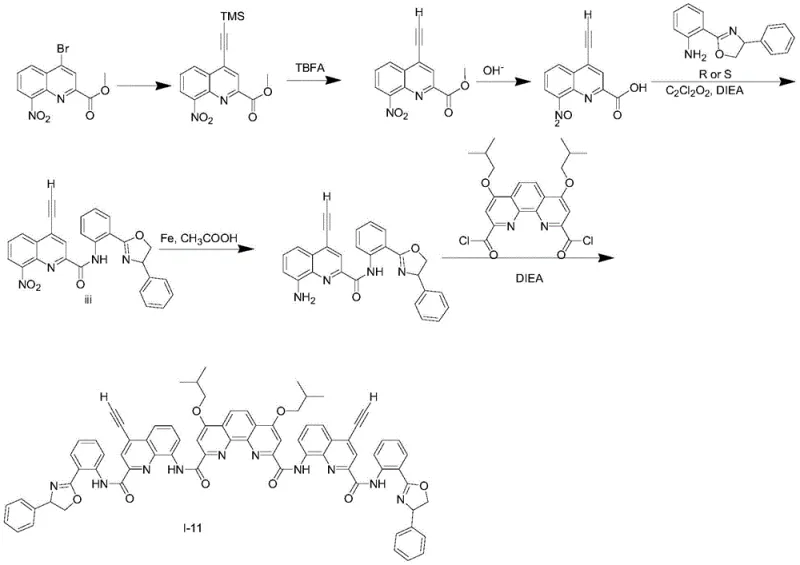 This final cyclization locks the molecule into its folded conformation, ensuring that the chiral information is effectively communicated to the fluorophore, resulting in the observed high g_lum values.
This final cyclization locks the molecule into its folded conformation, ensuring that the chiral information is effectively communicated to the fluorophore, resulting in the observed high g_lum values.
How to Synthesize Chiral Foldamer Fluorescent Compounds Efficiently
The synthesis of these high-performance materials follows a modular approach that allows for the systematic variation of substituents to optimize optical properties. The process begins with the preparation of the quinoline monomer, followed by the attachment of the chiral auxiliary and final assembly of the foldamer backbone. Each step has been optimized for yield and purity, utilizing standard purification techniques such as column chromatography with silica gel. The detailed standardized synthesis steps are provided below to guide process development teams in replicating these results.
- Perform Sonogashira coupling between methyl 8-nitro-4-bromoquinoline-2-carboxylate and substituted alkynes using Pd/Pd catalysts.
- Hydrolyze the ester intermediate to the corresponding carboxylic acid using alkaline conditions followed by acidification.
- Couple the acid with chiral aminophenyl oxazole derivatives using HATU or similar coupling reagents, then reduce the nitro group to an amine.
- Finalize the foldamer structure by condensing the diamine intermediate with 1,10-diisobutoxyphenanthroline-2,9-dicarbonyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this foldamer technology offers substantial strategic benefits beyond mere performance metrics. The synthetic route utilizes widely available starting materials, such as bromoquinolines and common terminal alkynes, which mitigates the risk of raw material shortages often associated with exotic metal complexes. The elimination of expensive lanthanide salts and the reliance on earth-abundant palladium catalysts, which can potentially be recovered, leads to significant cost optimization in the overall manufacturing budget. Furthermore, the robustness of the covalent foldamer structure implies a longer shelf-life and greater stability during device fabrication, reducing waste and improving yield in downstream applications. This reliability is crucial for maintaining consistent production schedules in the fast-paced display industry.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of precious metal emitters and complex supramolecular assembly conditions, relying instead on standard organic transformations that are well-understood and easily scalable. By utilizing commercially available reagents and avoiding cryogenic conditions, the energy consumption and operational costs are drastically simplified. The high quantum yields reported mean that less material is required to achieve the same brightness, effectively lowering the cost-per-unit of the final electronic device. Additionally, the ability to tune properties via simple substituent changes reduces the R&D expenditure needed to develop new product variants.
- Enhanced Supply Chain Reliability: The precursors for this synthesis, including quinoline derivatives and chiral oxazoles, are accessible through established chemical supply chains, ensuring continuity of supply. Unlike rare-earth dependent technologies, this organic approach is not subject to the geopolitical volatility often seen in the mining sector. The synthetic steps are robust and tolerant to minor variations in conditions, which enhances the reproducibility of the process across different manufacturing sites. This consistency is vital for qualifying materials in stringent automotive or aerospace display applications where supply security is paramount.
- Scalability and Environmental Compliance: The reaction conditions described, such as room temperature couplings and ambient pressure reductions, are inherently safer and easier to scale from gram to kilogram quantities. The use of iron powder for reduction generates less hazardous waste compared to catalytic hydrogenation requiring high-pressure equipment. Moreover, the purification methods rely on standard chromatography, which can be adapted to preparative HPLC or crystallization processes for large-scale production. This alignment with green chemistry principles facilitates regulatory compliance and simplifies the environmental impact assessment for new facility approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral foldamer technology. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy for engineering and procurement teams evaluating this material for integration into next-generation optoelectronic devices.
Q: What is the primary advantage of this foldamer structure over traditional chiral luminescent materials?
A: Unlike traditional materials that rely on weak supramolecular interactions, this covalent foldamer structure offers superior stability, higher quantum yields exceeding 65%, and tunable emission wavelengths via the R3 substituent.
Q: Can the circularly polarized luminescence (CPL) signal be adjusted for specific applications?
A: Yes, the CPL signal intensity, luminescence intensity, and emission color are highly tunable by modifying the R3 group on the quinoline backbone, allowing customization for different optoelectronic requirements.
Q: Is the synthesis process suitable for large-scale commercial production?
A: The process utilizes standard organic synthesis techniques such as Sonogashira coupling and amide condensation with commercially available reagents, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Fluorescent Compounds Supplier
As the demand for advanced display technologies grows, securing a partner with deep expertise in complex organic synthesis is essential. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of chiral fluorescent compounds meets the exacting standards required for high-end OLED and sensor applications. We understand the critical nature of supply chain stability and are equipped to handle the nuanced chemistry required for foldamer assembly.
We invite you to collaborate with us to accelerate your product development cycles. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact our technical procurement team to request specific COA data and route feasibility assessments for integrating these high-efficiency chiral materials into your manufacturing pipeline. Together, we can drive the next generation of circularly polarized light technology forward.
