Revolutionizing Bromfenac Intermediate Production via Advanced Nickel-Catalyzed Carbonylation
Revolutionizing Bromfenac Intermediate Production via Advanced Nickel-Catalyzed Carbonylation
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical non-steroidal anti-inflammatory drug (NSAID) precursors. Patent CN109608380B introduces a groundbreaking synthetic methodology for 7-(p-bromobenzoyl)indol-2-one, a pivotal intermediate in the manufacture of Bromfenac Sodium. This innovation addresses long-standing inefficiencies in heterocyclic chemistry by replacing multi-step, low-yield processes with a direct, Nickel-catalyzed addition of arylboronic acids to nitriles. For R&D directors and procurement specialists, this technology represents a paradigm shift towards greener, more cost-effective manufacturing of ophthalmic therapeutic agents.
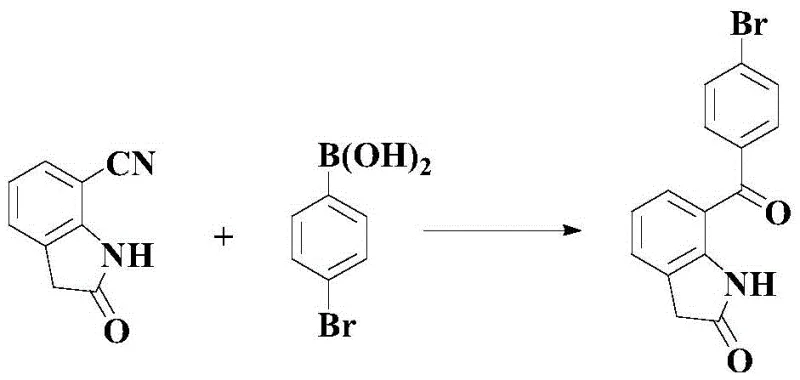
The Limitations of Conventional Methods vs. The Novel Approach
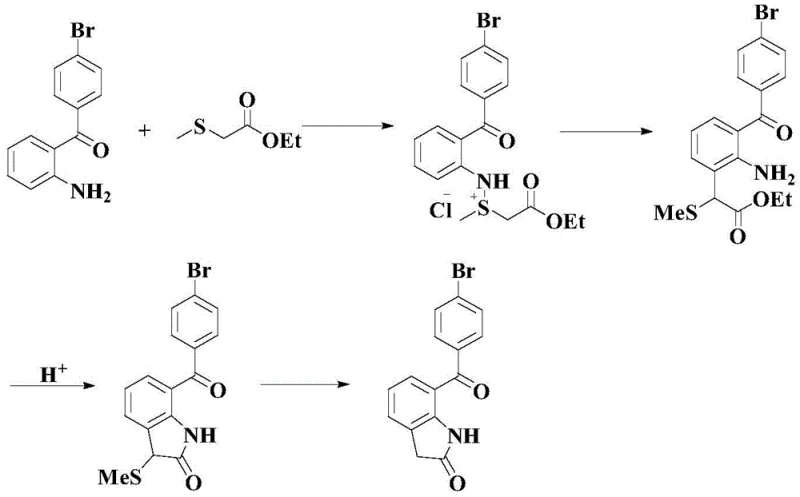
The Limitations of Conventional Methods
Historically, the synthesis of 7-(p-bromobenzoyl)indol-2-one has been plagued by operational complexity and environmental hazards. Prior art, such as US Patents 4126635 and 4182774, relies on a tedious four-step sequence starting from 2-amino-4'-bromo-benzophenone. This legacy approach necessitates cryogenic conditions at -70°C for thio-salt formation, followed by rearrangement and cyclization steps that utilize hazardous reagents like Raney Nickel. Furthermore, alternative routes described in EP0221753 employ massive quantities of aluminum trichloride, a corrosive Lewis acid that generates significant acidic waste streams. These conventional methodologies typically suffer from overall yields below 30%, creating substantial material loss and inflating the cost of goods sold (COGS) for downstream API production.
The Novel Approach
In stark contrast, the methodology disclosed in CN109608380B streamlines the entire synthesis into a single catalytic step. By leveraging the reactivity of 7-cyanoindole-2-one with p-bromophenylboronic acid, this novel route bypasses the need for pre-functionalized benzophenones or harsh Friedel-Crafts conditions. The process operates under relatively mild thermal conditions (70-100°C) and achieves isolated yields exceeding 82%, effectively tripling the efficiency of previous best-in-class methods. This drastic reduction in step count not only minimizes solvent consumption and waste generation but also significantly shortens the production cycle time, offering a compelling value proposition for high-volume manufacturing.
Mechanistic Insights into Ni(dppe)Cl2-Catalyzed Nitrile Addition
The success of this transformation hinges on the precise tuning of the catalytic system to overcome inherent electronic and steric challenges. Standard Nickel-catalyzed additions of boronic acids to nitriles often fail when applied to substrates containing coordinating groups, such as the imino moiety present in 7-cyanoindole-2-one. The inventors identified that the imino nitrogen can competitively coordinate with the Nickel center, leading to catalyst deactivation and preventing the crucial insertion of the nitrile carbon into the Nickel-aryl bond. To counteract this, the protocol employs Ni(dppe)Cl2, a catalyst featuring a bidentate phosphine ligand that stabilizes the active metal species against poisoning while maintaining sufficient electrophilicity for catalytic turnover.
Furthermore, the inclusion of a strong acid promoter, specifically trifluoromethanesulfonic acid, plays a dual role in activating the nitrile group and facilitating the hydrolysis of the intermediate imine to the final ketone. Without this specific acid additive, or when using weaker alternatives like methanesulfonic acid, the reaction stalls or proceeds with negligible conversion. The synergy between the specific Nickel-ligand complex and the superacidic promoter ensures that the electron-rich aromatic ring does not inhibit the nucleophilic attack, allowing for the efficient construction of the sterically hindered ketone linkage essential for the Bromfenac scaffold.
How to Synthesize 7-(p-Bromobenzoyl)indol-2-one Efficiently
The implementation of this synthesis requires careful attention to stoichiometry and reaction atmosphere to ensure reproducibility at scale. The process begins with the suspension of the nitrile substrate and the boronic acid coupling partner in an anhydrous etheral solvent, followed by the sequential addition of the catalyst and acid promoter. Maintaining an inert atmosphere is critical to prevent oxidation of the low-valent Nickel species. Detailed standardized operating procedures regarding mixing rates, temperature ramping profiles, and quenching protocols are essential for safety and quality control.
- Combine 7-cyanoindole-2-one and p-bromophenylboronic acid in 2-MeTHF solvent with Ni(dppe)Cl2 catalyst.
- Add trifluoromethanesulfonic acid as a promoter and heat the mixture to 70-100°C for 12-24 hours.
- Perform standard aqueous workup and purification via flash column chromatography to isolate the high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Nickel-catalyzed route offers transformative economic benefits beyond simple yield improvements. The shift from a four-step linear synthesis to a convergent one-step process fundamentally alters the cost structure of the intermediate. By eliminating three distinct unit operations, manufacturers can drastically reduce labor costs, energy consumption for heating and cooling, and the capital expenditure associated with reactor occupancy time. This efficiency gain translates directly into a more competitive pricing model for the final API, enhancing margin potential in the crowded NSAID market.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as aluminum trichloride and Raney Nickel removes the need for specialized waste treatment facilities and costly neutralization steps. Additionally, the high atom economy of the boronic acid coupling means that raw material spend is optimized, with less feedstock lost to side reactions or purification losses. The use of 2-MeTHF, a bio-based and recyclable solvent, further aligns the process with modern sustainability goals, potentially reducing regulatory compliance costs associated with volatile organic compound (VOC) emissions.
- Enhanced Supply Chain Reliability: Traditional routes depend on 2-amino-4'-bromo-benzophenone, a specialized building block that may have limited suppliers and long lead times. In contrast, the new method utilizes p-bromophenylboronic acid and 7-cyanoindole-2-one, which are commodity chemicals available from multiple global sources. This diversification of the raw material base mitigates the risk of supply disruptions and provides procurement teams with greater leverage in vendor negotiations, ensuring continuous production flow even during market volatility.
- Scalability and Environmental Compliance: The simplified workflow inherently reduces the physical footprint required for production, making it easier to scale from pilot plant to commercial tonnage without proportional increases in facility size. The absence of heavy metal contaminants like aluminum residues simplifies the purification train, reducing the burden on QC labs for impurity profiling. Moreover, the reduced generation of acidic and heavy metal waste streams facilitates easier disposal and lowers the environmental impact score of the manufacturing site, supporting corporate ESG initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these nuances is vital for technology transfer teams evaluating the feasibility of integrating this route into existing manufacturing lines. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation.
Q: Why is the Nickel-catalyzed method superior to traditional Friedel-Crafts acylation?
A: Traditional methods require harsh Lewis acids like AlCl3 and multiple steps, resulting in yields below 30%. The new Nickel-catalyzed route is a one-step process with yields exceeding 82%, eliminating toxic waste and simplifying purification.
Q: What are the critical reaction parameters for maximizing yield?
A: The selection of the ligand and acid promoter is crucial. Ni(dppe)Cl2 paired with trifluoromethanesulfonic acid in 2-MeTHF solvent provides the optimal environment to overcome catalyst deactivation by the imino group.
Q: Is this process scalable for commercial API production?
A: Yes, the use of commercially available boronic acids and robust nickel catalysts, combined with mild reaction temperatures (70-100°C), makes this pathway highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-(p-Bromobenzoyl)indol-2-one Supplier
As the demand for high-quality ophthalmic therapeutics grows, securing a stable supply of key intermediates like 7-(p-bromobenzoyl)indol-2-one is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle sensitive Nickel-catalyzed reactions under strict inert conditions, ensuring that every batch meets stringent purity specifications required for GMP API synthesis. Our rigorous QC labs utilize advanced analytical techniques to verify identity and assay, guaranteeing consistency that generic manufacturers demand.
We invite global partners to collaborate with us to leverage this superior synthetic route for their Bromfenac projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most robust and economically viable chemical foundation available today.
