Scalable Four-Step Synthesis of Alpha-Azaspiro Compounds for Advanced Drug Development
Introduction to Advanced Spirocyclic Synthesis Technology
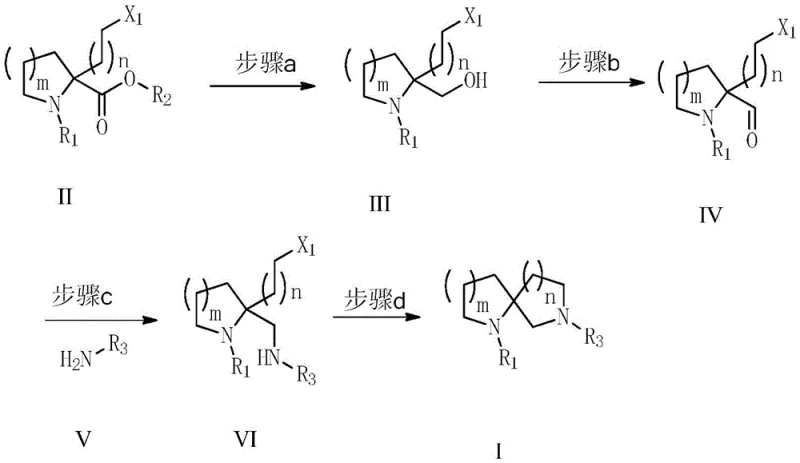
The pharmaceutical industry continuously demands more efficient and safer pathways for constructing complex heterocyclic scaffolds, particularly alpha-azaspiro compounds which serve as critical building blocks for next-generation therapeutics. Patent CN106883238B, published in late 2020, introduces a groundbreaking preparation method that fundamentally reshapes the manufacturing landscape for these high-value intermediates. This technology addresses long-standing inefficiencies in spirocycle construction by condensing the synthetic sequence into a robust four-step protocol involving reduction, oxidation, reductive amination, and cyclization. Unlike traditional approaches that often suffer from cumulative yield losses and safety hazards, this novel methodology achieves a remarkable total yield ranging from 51.5% to 66.7%. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, enabling the rapid generation of diverse spirocyclic libraries essential for modern drug discovery programs targeting GPCRs and ion channels.
From a supply chain perspective, the elimination of exotic or highly unstable reagents ensures a more reliable sourcing strategy for raw materials. The patent explicitly details the use of common reducing agents like sodium borohydride and standard oxidants such as IBX or DMP, which are commercially available in bulk quantities. This shift away from specialized catalysts reduces the risk of supply disruptions and price volatility often associated with niche chemical reagents. Furthermore, the mild reaction conditions described, typically operating between -78°C and 150°C depending on the specific step, allow for the use of standard glass-lined reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure significantly lowers the barrier to entry for contract development and manufacturing organizations (CDMOs) looking to adopt this technology for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for synthesizing diazaspiro compounds have been plagued by inherent inefficiencies that hinder their application in large-scale manufacturing. As illustrated in prior art literature such as J. Med. Chem. (1990) and various patents like WO2004005293, traditional routes frequently require between five to eight distinct reaction steps to achieve the target molecular architecture. These multi-step sequences inevitably lead to substantial material loss at each stage, resulting in overall yields that rarely exceed 30%, with some reported values as low as 7%. Such poor efficiency not only drives up the cost of goods sold (COGS) but also generates excessive chemical waste, creating significant environmental compliance burdens for manufacturing facilities. Additionally, many of these legacy processes rely on hazardous reagents; for instance, certain pathways utilize potassium cyanide for nitrile introduction or explosive nitroethylene derivatives, posing severe safety risks to plant personnel and requiring expensive containment protocols.
The Novel Approach
The methodology disclosed in CN106883238B offers a transformative solution by streamlining the synthesis into a concise four-step sequence that bypasses the need for dangerous reagents and complex purification workflows. By starting with readily available N-protected proline or piperidine carboxylic acid derivatives, the process utilizes a strategic alpha-substitution followed by a cascade of functional group transformations. The key innovation lies in the direct conversion of an ester intermediate to an aldehyde via a stable alcohol, which then undergoes reductive amination and spontaneous cyclization. This approach avoids the isolation of unstable intermediates and minimizes the number of unit operations required. The result is a process that is not only higher yielding but also inherently safer, as it replaces pyrophoric catalysts like Raney-Ni and strong hydrides like lithium aluminum hydride with milder alternatives like sodium borohydride stabilized by calcium chloride. This makes the technology ideal for cost reduction in pharmaceutical intermediate manufacturing where safety and throughput are paramount.
Mechanistic Insights into Reductive Amination and Cyclization
The core chemical transformation driving this synthesis involves a sophisticated interplay of reduction and oxidation states managed within a protected amine framework. The process begins with the selective reduction of an alpha-substituted ester to a primary alcohol using sodium borohydride in the presence of calcium chloride, a combination that enhances reactivity while maintaining selectivity. Following this, the alcohol is oxidized to an aldehyde using reagents such as 2-iodoxybenzoic acid (IBX) or Dess-Martin periodinane (DMP). These oxidants are chosen for their ability to operate under mild conditions without over-oxidizing the sensitive amine functionality or causing racemization of the chiral center. The subsequent reductive amination step is critical, where the generated aldehyde reacts with an organic amine, such as benzylamine, to form an imine intermediate which is immediately reduced in situ. This one-pot strategy prevents the accumulation of reactive aldehydes and drives the equilibrium towards the desired amine product efficiently.
Final ring closure is achieved through an intramolecular nucleophilic substitution where the newly formed secondary amine attacks the pendant halogenated alkyl chain. This cyclization is facilitated by heating the reaction mixture in polar aprotic solvents like DMF or NMP in the presence of a base such as triethylamine or DIPEA. The base serves to scavenge the hydrogen halide byproduct, pushing the reaction to completion. This mechanistic pathway ensures high stereochemical integrity and minimizes the formation of oligomeric byproducts that often plague intermolecular cyclization reactions. The robustness of this mechanism allows for the variation of ring sizes, enabling the production of spiro[4.3], spiro[4.4], and larger systems simply by adjusting the length of the halogenated chain introduced in the initial alkylation step. This flexibility is crucial for medicinal chemists exploring structure-activity relationships (SAR) in drug discovery campaigns.
![Specific example reaction scheme for synthesizing tert-butyl 7-benzyl-1,7-diaza-spiro[4.3]octane-1-carboxylate](/insights/img/alpha-azaspiro-synthesis-pharma-intermediate-supplier-20260305122307-011.webp)
How to Synthesize Alpha-Azaspiro Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control during the reduction and oxidation phases to ensure optimal yield and purity. The patent provides detailed experimental procedures that serve as a blueprint for process chemists aiming to replicate these results in a pilot or production setting. The standardized protocol emphasizes the importance of stoichiometry, recommending specific molar equivalents for reducing agents and oxidants to prevent side reactions. For instance, the use of 2 to 5 equivalents of sodium borohydride ensures complete conversion of the ester without necessitating excessive workup procedures. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in validating this process.
- Reduce the ester group of the protected proline or piperidine derivative to a hydroxymethyl intermediate using sodium borohydride and calcium chloride.
- Oxidize the resulting alcohol to an aldehyde using mild oxidants such as IBX, DMP, or PCC under controlled temperatures.
- Perform reductive amination with an organic amine followed by intramolecular cyclization using a base to form the final spirocyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized and often import-restricted reagents like potassium cyanide or explosive nitroethylene derivatives, companies can source all necessary inputs from standard chemical suppliers with short lead times. This diversification of the supplier base reduces the risk of production stoppages due to material shortages. Furthermore, the replacement of expensive transition metal catalysts with inexpensive inorganic salts like calcium chloride and sodium borohydride leads to substantial cost savings in raw material expenditure. The avoidance of heavy metals also simplifies the downstream purification process, removing the need for costly scavenger resins or extensive chromatography steps typically required to meet strict residual metal specifications for API intermediates.
- Cost Reduction in Manufacturing: The streamlined four-step process significantly lowers operational expenditures by reducing solvent consumption, energy usage, and labor hours associated with multiple isolation and purification stages. The high overall yield of over 50% means that less starting material is required to produce the same amount of final product, directly improving the margin profile. Additionally, the use of non-hazardous reagents reduces the costs associated with waste disposal and environmental compliance, as the effluent streams are easier to treat compared to those containing cyanides or heavy metals. This economic efficiency makes the process highly competitive for generic drug manufacturers looking to optimize their cost structures.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals ensures a stable and continuous supply of inputs, mitigating the volatility often seen with specialty reagents. Since the reaction conditions are mild and do not require extreme pressures or cryogenic temperatures for extended periods, the process is less susceptible to equipment failures or utility fluctuations. This reliability translates into more predictable production schedules and shorter lead times for delivering high-purity pharmaceutical intermediates to customers. The robustness of the chemistry also allows for flexible batch sizing, enabling manufacturers to respond quickly to changes in market demand without compromising product quality or consistency.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale reactor systems. The absence of explosive intermediates allows for safer operation at larger volumes, reducing the need for specialized blast-proof facilities. From an environmental standpoint, the atom economy of the reaction is superior to prior art methods, generating less chemical waste per kilogram of product. This aligns with green chemistry principles and helps manufacturers meet increasingly stringent regulatory requirements regarding carbon footprint and waste generation. The ease of scaling this technology facilitates the rapid transition from laboratory benchtop to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-azaspiro synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this process for potential adoption. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The patented method reduces the synthesis from 5-8 steps down to just 4 steps, increasing total yield from typically 5-30% to 51.5%-66.7% while eliminating hazardous reagents like potassium cyanide and explosive nitroethylene.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction conditions and readily available reagents such as sodium borohydride and common organic bases, making it highly suitable for industrial scale-up without requiring specialized high-pressure equipment.
Q: What types of spirocyclic structures can be generated using this methodology?
A: This versatile route supports the formation of various ring sizes including spiro[4.3], spiro[4.4], spiro[4.5], and spiro[5.5] systems by varying the halogenated alkyl chain length and the starting amino acid derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Azaspiro Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in accelerating drug development timelines and reducing time-to-market for new therapies. Our team of expert process chemists has extensively evaluated the technology disclosed in CN106883238B and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab scale to industrial manufacturing is seamless and robust. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies, guaranteeing that every batch of alpha-azaspiro compound delivered meets the highest quality standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their specific projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements and purity needs. We encourage you to contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and achieve your development goals with our reliable alpha-azaspiro compound supply solutions.
