Advanced One-Pot Synthesis Of Benzodipyrrolone Fluoroboron Dyes For Commercial Scale-Up
Advanced One-Pot Synthesis Of Benzodipyrrolone Fluoroboron Dyes For Commercial Scale-Up
The rapid advancement of bioimaging and optoelectronic technologies has created an urgent demand for fluorophores that combine structural rigidity with exceptional photophysical properties. Patent CN110872312B addresses this critical market need by disclosing a novel class of benzodipyrrolone fluoroboron fluorescent dyes. These compounds are characterized by their unique ability to lock free-rotating conjugated ligands through four-coordinate electron-deficient boron atoms, thereby rigidifying the molecular structure and significantly enhancing fluorescence quantum yield. Unlike traditional synthetic routes that often suffer from complex multi-step procedures and harsh purification requirements, this invention introduces a streamlined "three-step one-pot" methodology. This approach not only simplifies the operational workflow but also leverages inexpensive, commercially available raw materials such as isoindolinone and pyrrole derivatives. For R&D directors and procurement specialists alike, this patent represents a significant breakthrough in the efficient manufacturing of high-performance optical materials, offering a robust pathway to scalable production without compromising on spectral tunability or brightness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-efficiency fluoroboron dyes, such as BODIPY analogues, has been plagued by several inherent inefficiencies that hinder large-scale commercial adoption. Traditional methods frequently rely on the condensation of aldehydes with pyrroles followed by oxidation and complexation, a sequence that often requires stringent anhydrous conditions and the use of expensive oxidizing agents like DDQ or chloranil. Furthermore, the intermediate dipyrromethenes generated in these processes are often unstable and prone to decomposition, necessitating immediate subsequent reactions or difficult isolation steps that lower overall throughput. The reliance on transition metal catalysts in some variations introduces another layer of complexity, as residual metal impurities must be rigorously removed to meet the purity standards required for biological applications. These factors collectively contribute to elevated production costs, extended lead times, and significant challenges in waste management, making conventional routes less attractive for cost-sensitive industrial applications where margin compression is a constant pressure.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN110872312B utilizes a direct condensation strategy that fundamentally restructures the synthetic logic. By employing isoindolinone as a key building block, the process bypasses the need for external oxidants, as the isoindolinone scaffold inherently provides the necessary oxidation state for the final fluorophore. The reaction proceeds through a sequential addition of reagents in a single vessel: first, the condensation of isoindolinone with pyrrole under Lewis acid catalysis; second, the acylation with acid chlorides to introduce diversity; and finally, the complexation with boron trifluoride etherate. This telescoped approach minimizes solvent usage and eliminates the need for intermediate workups, drastically reducing the environmental footprint and operational labor. The result is a highly efficient protocol that delivers structurally diverse dyes with excellent molar absorptivity and tunable emission profiles, effectively solving the scalability issues associated with previous generations of fluorescent probes.
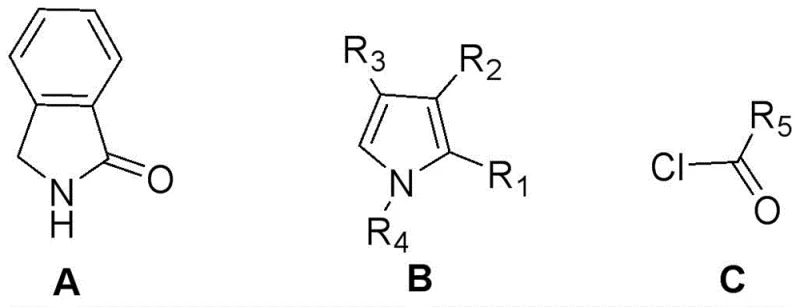
Mechanistic Insights into Lewis Acid-Catalyzed Condensation and Boron Coordination
The core of this synthetic innovation lies in the precise orchestration of Lewis acid-mediated activation and nucleophilic attack. In the initial step, phosphorus oxychloride (POCl3) acts as both a solvent and a potent Lewis acid, activating the carbonyl group of the isoindolinone towards nucleophilic attack by the electron-rich pyrrole ring. This interaction facilitates the formation of a carbon-carbon bond between the heterocycles, generating an asymmetric dipyrrole intermediate in situ. The choice of POCl3 is critical, as it promotes the dehydration necessary for aromatization without over-oxidizing the sensitive pyrrole moiety. Following this condensation, the introduction of an acyl chloride serves to functionalize the nitrogen atom or the adjacent carbon, depending on the specific substitution pattern desired, effectively capping the reactive intermediate and setting the stage for the final rigidification. This step is performed at controlled low temperatures (0-30°C) to prevent side reactions and ensure regioselectivity, demonstrating a sophisticated control over the reaction kinetics that is essential for high-purity outcomes.
The final and perhaps most crucial mechanistic event is the coordination of the difluoroboron moiety. Upon the addition of triethylamine to neutralize the acidic byproducts, boron trifluoride etherate is introduced to coordinate with the nitrogen and oxygen atoms of the ligand system. This coordination creates a rigid, planar four-membered ring structure that locks the conformational freedom of the molecule. From a photophysical perspective, this rigidification is paramount; it suppresses non-radiative decay pathways caused by molecular vibration and rotation, thereby channeling the excited state energy almost exclusively into radiative fluorescence. The resulting dyes exhibit large Stokes shifts and high quantum yields, attributes that are directly attributable to this specific boron chelation mechanism. Understanding this interplay between structural rigidity and electronic delocalization allows chemists to rationally design derivatives with specific optical properties by simply varying the R-groups on the starting materials, providing a powerful tool for custom material development.
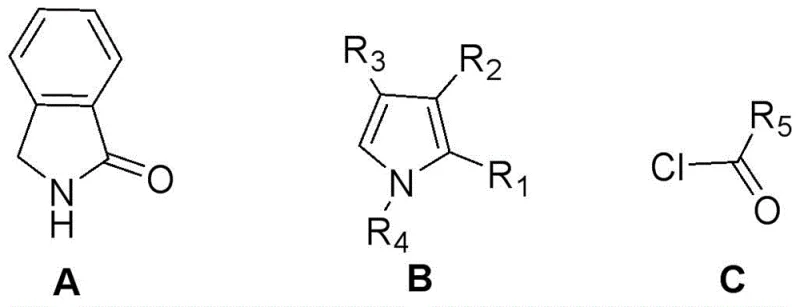
How to Synthesize Benzodipyrrolone Fluoroboron Dye Efficiently
The practical implementation of this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process begins by dissolving isoindolinone and the chosen pyrrole derivative in an anhydrous chlorinated solvent, such as chlorobenzene or dichloroethane, under an inert nitrogen atmosphere to prevent moisture interference. Phosphorus oxychloride is then added, and the mixture is heated to temperatures ranging between 70°C and 150°C for several hours to drive the condensation to completion. Once the initial coupling is verified, typically via TLC monitoring, the reaction mixture is cooled, and the acyl chloride is added dropwise to manage the exotherm. The final complexation step involves the sequential addition of a base like triethylamine followed by the boron source, with the temperature carefully maintained to optimize the coordination equilibrium. For a detailed breakdown of the specific stoichiometric ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Mix isoindolinone, pyrrole derivative, solvent, and Lewis acid (e.g., POCl3) and heat to 70-150°C for the first contact reaction.
- Add acyl chloride to the mixture at 0-30°C to perform the second contact reaction for acylation.
- Sequentially add triethylamine and boron trifluoride etherate at 0-120°C to complete the fluoroboron coordination.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere technical performance. The reliance on commodity chemicals such as isoindolinone and simple pyrroles means that the raw material supply chain is robust and less susceptible to the volatility often seen with specialized organometallic reagents. This stability in sourcing translates directly into more predictable pricing models and reduced risk of production stoppages due to material shortages. Furthermore, the "one-pot" nature of the synthesis significantly reduces the consumption of solvents and the generation of aqueous waste streams, which are major cost drivers in chemical manufacturing. By consolidating three distinct chemical transformations into a single reactor run, manufacturers can achieve substantial reductions in utility costs, labor hours, and equipment occupancy time, thereby enhancing the overall economic efficiency of the production line without the need for capital-intensive new infrastructure.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the avoidance of expensive oxidizing agents or transition metal catalysts lead to a drastic simplification of the downstream processing requirements. This streamlined workflow removes the need for costly chromatography columns or extensive recrystallization cycles that are typical in multi-step syntheses, resulting in significant operational expenditure savings. Additionally, the high atom economy of the condensation reaction ensures that a larger proportion of the input mass is converted into the final valuable product, minimizing waste disposal fees and maximizing the return on raw material investment.
- Enhanced Supply Chain Reliability: Because the starting materials are widely available bulk chemicals rather than niche custom synthons, the lead time for procuring inputs is significantly shortened. This accessibility allows for more agile inventory management and the ability to scale production volumes rapidly in response to market demand fluctuations. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, further insulates the manufacturing process from minor variations in raw material quality, ensuring consistent output and reliable delivery schedules for downstream customers in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as the exothermic nature of the reactions can be easily managed in larger reactors through controlled dosing and cooling systems. The reduced solvent volume per unit of product lowers the burden on solvent recovery systems and decreases the facility's overall volatile organic compound (VOC) emissions. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the corporate sustainability profile, a factor that is increasingly critical for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fluorophores. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating these materials into existing product lines or research pipelines. We encourage stakeholders to review these points carefully to appreciate the full scope of the technology's capabilities and limitations.
Q: What are the key advantages of the one-pot synthesis method described in CN110872312B?
A: The method eliminates the need for isolating unstable intermediates, significantly reducing processing time and solvent consumption while maintaining high fluorescence quantum yields up to 0.91.
Q: Can the emission spectrum of these benzodipyrrolone dyes be tuned?
A: Yes, by modifying the R1-R5 substituents on the pyrrole and benzene rings, the maximum emission wavelength can be adjusted between 490 nm and 640 nm to suit specific optical applications.
Q: What raw materials are required for this synthesis?
A: The process utilizes commercially available and cost-effective starting materials such as isoindolinone, pyrrole derivatives, acyl chlorides, and standard Lewis acids like phosphorus oxychloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodipyrrolone Fluoroboron Dye Supplier
As the global demand for high-performance fluorescent markers continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex heterocyclic synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging the innovative methodologies described in patents like CN110872312B to deliver superior optical materials. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without sacrificing quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzodipyrrolone fluoroboron dye meets the exacting standards required for bioimaging and sensor applications.
We invite you to collaborate with us to explore how these advanced dyes can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how our optimized synthesis routes can improve your bottom line. Please contact us today to request specific COA data for our current inventory or to discuss route feasibility assessments for custom derivatives. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and driving innovation in your field.
