Advanced Synthesis of 1-Aryl-2-Indolinone Derivatives for Commercial Scale-Up
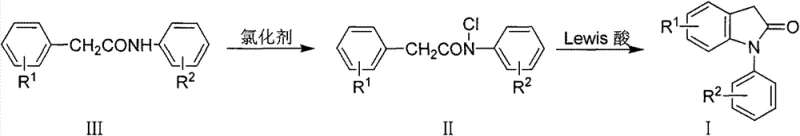
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical building blocks for active pharmaceutical ingredients. Patent CN103288708A discloses a highly efficient preparation method for 1-aryl-2-indolinone derivatives, a structural motif prevalent in numerous bioactive compounds including nonsteroidal anti-inflammatory drugs. This innovation addresses long-standing challenges in heterocycle synthesis by replacing harsh traditional conditions with a mild, two-step sequence involving N-chlorination followed by Lewis acid-catalyzed cyclization. The technical significance of this disclosure lies in its ability to utilize inexpensive, commercially available reagents while maintaining high yields and operational simplicity. For global supply chains, this represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolinone ring system has heavily relied on classical Friedel-Crafts acylation or related cyclization strategies that impose severe constraints on process engineering. These conventional pathways frequently necessitate elevated reaction temperatures and the use of stoichiometric amounts of aggressive reagents, which can lead to significant safety hazards and increased energy consumption. Furthermore, the substrate scope in traditional methods is often limited by the sensitivity of functional groups to harsh acidic or thermal conditions, resulting in lower overall yields and complex purification requirements. The reliance on expensive starting materials and the generation of difficult-to-treat waste streams further exacerbate the economic and environmental burden of these legacy processes. Consequently, there is a pressing industrial need for alternative methodologies that can circumvent these thermodynamic and kinetic barriers without compromising product quality.
The Novel Approach
The methodology outlined in the patent introduces a transformative strategy by utilizing an N-chloro-N-aryl-substituted phenylacetamide intermediate to drive the ring closure under much gentler conditions. By pre-functionalizing the amide nitrogen with a chlorine atom, the subsequent intramolecular electrophilic substitution is activated sufficiently to proceed at temperatures ranging from 0°C to reflux, depending on the specific solvent system employed. This approach effectively bypasses the need for extreme thermal activation, thereby preserving sensitive substituents on the aromatic rings and minimizing side reactions such as polymerization or decomposition. The versatility of the method is underscored by its compatibility with a wide array of chlorination reagents, including sodium hypochlorite and trichloroisocyanuric acid, allowing manufacturers to optimize reagent selection based on local availability and cost. This strategic modification of the reaction pathway not only enhances safety but also streamlines the downstream processing steps required to isolate the final high-purity product.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this synthetic innovation relies on the precise activation of the N-chloro amide species by a Lewis acid catalyst to generate a highly reactive electrophilic center. Upon coordination with the Lewis acid, such as anhydrous aluminum trichloride or iron trichloride, the nitrogen-chlorine bond becomes polarized, facilitating the formation of a nitrenium-like ion or a strongly electrophilic iminium species. This activated intermediate then undergoes an intramolecular electrophilic aromatic substitution with the pendant phenyl ring, effectively closing the five-membered lactam ring to form the indolinone core. The choice of Lewis acid is critical, as it must be strong enough to promote this cyclization yet compatible with the solvent system to prevent excessive degradation of the product. The mechanism ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 1-aryl-2-indolinone structure over potential isomeric byproducts. Understanding this catalytic cycle is essential for R&D teams aiming to fine-tune reaction parameters for specific substituted derivatives.
Impurity control in this process is inherently managed through the mildness of the reaction conditions and the specificity of the cyclization mechanism. Unlike high-temperature processes that often generate tars and polymeric residues, this low-temperature protocol limits the energy available for non-specific radical reactions or over-chlorination of the aromatic rings. The use of specific chlorinating agents allows for precise control over the degree of halogenation, ensuring that the N-chloro intermediate is formed cleanly before the cyclization step begins. Furthermore, the workup procedures described, which often involve simple aqueous washes and recrystallization, are highly effective at removing residual Lewis acids and inorganic salts. This results in a final product with a clean impurity profile, reducing the burden on analytical quality control laboratories and ensuring that the material meets the stringent specifications required for pharmaceutical applications. The ability to consistently produce high-purity material is a direct consequence of the mechanistic elegance of this two-step sequence.
How to Synthesize 1-Aryl-2-Indolinone Efficiently
The synthesis of 1-aryl-2-indolinone derivatives via this patented route involves a straightforward sequence that begins with the dissolution of the N-aryl-substituted phenylacetamide precursor in a suitable organic solvent. Following the addition of a selected chlorination reagent under controlled conditions, the reaction mixture is processed to yield the N-chloro intermediate, which serves as the key substrate for the subsequent cyclization. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature controls, and workup procedures necessary to achieve optimal yields. This protocol is designed to be adaptable, allowing chemists to substitute various Lewis acids and solvents to accommodate different scale requirements or substrate sensitivities. Adherence to these guidelines ensures reproducible results and maximizes the efficiency of the transformation from simple amides to complex heterocyclic structures.
- Dissolve N-aryl-substituted phenylacetamide in an organic solvent and react with a chlorination reagent to form the N-chloro intermediate.
- Add a Lewis acid catalyst such as aluminum trichloride or iron trichloride to the reaction mixture at controlled temperatures.
- Maintain the reaction under mild conditions to facilitate intramolecular cyclization, yielding the target 1-aryl-2-indolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis route offers substantial advantages by eliminating the dependency on scarce or prohibitively expensive catalysts often found in transition-metal-mediated couplings. The reagents required, such as aluminum trichloride, sodium hypochlorite, and common organic solvents like dichloromethane or acetonitrile, are commodity chemicals available from multiple global suppliers. This diversification of the supply base significantly mitigates the risk of raw material shortages and price volatility, ensuring a stable and continuous production flow for downstream API manufacturing. The simplicity of the operation also translates to reduced labor costs and shorter training periods for plant operators, as the process does not require specialized high-pressure equipment or inert atmosphere techniques beyond standard industry practices. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive chlorinating agents drastically lower the direct material costs associated with production. By avoiding the need for expensive ligands or specialized reagents, the overall cost of goods sold is significantly reduced, allowing for more competitive pricing in the global market. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further enhancing the economic viability of the process on a large scale. The high yields reported for both the chlorination and cyclization steps minimize waste generation, leading to better atom economy and lower disposal costs for chemical byproducts.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals ensures that production schedules are not disrupted by the lead times associated with custom-synthesized reagents. Since the process tolerates a variety of solvents and Lewis acids, procurement teams have the flexibility to switch suppliers or substitute materials based on real-time market availability without compromising product quality. This adaptability is crucial for maintaining business continuity in the face of global logistical challenges or regional supply constraints. The robust nature of the chemistry also means that batch-to-batch variability is minimized, providing downstream customers with consistent material quality and reliable delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram to multi-kilogram scales without significant loss in efficiency. The use of standard organic solvents facilitates easy recovery and recycling, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. Furthermore, the absence of heavy metal residues simplifies the wastewater treatment process, ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up allows manufacturers to rapidly respond to market demand surges, making it an ideal candidate for the commercial production of high-volume pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to assist technical decision-makers in evaluating the feasibility of adopting this route for their specific production needs. The answers reflect the practical advantages and operational parameters defined in the intellectual property, providing a clear roadmap for successful technology transfer.
Q: What are the primary advantages of this synthesis method over traditional Friedel-Crafts reactions?
A: This method operates under significantly milder conditions compared to traditional Friedel-Crafts reactions, which often require high temperatures and expensive materials. The use of readily available chlorination reagents and common Lewis acids reduces overall production costs and simplifies operational safety.
Q: Which Lewis acids are suitable for the cyclization step in this process?
A: The patent specifies a range of effective Lewis acids including anhydrous aluminum trichloride, iron trichloride, zinc chloride, and various silver salts. This flexibility allows manufacturers to select catalysts based on availability and specific substrate compatibility.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability with simple operation steps and easily removable byproducts. The use of common organic solvents and commodity chemicals supports large-scale manufacturing without requiring specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aryl-2-Indolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into viable industrial processes. We are committed to delivering high-purity 1-aryl-2-indolinone derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies and optimized process engineering, we provide our partners with a secure and reliable source of complex heterocyclic intermediates essential for drug discovery and development.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your project's economic and technical outcomes. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain strategy and accelerate your time to market.
