Advanced Indenoimidazole Derivatives for High-Efficiency Organic Electroluminescent Devices
The rapid evolution of organic electroluminescent display technologies has created an urgent demand for advanced materials that can overcome the limitations of legacy systems in terms of efficiency, lifespan, and driving voltage. Patent CN107573328B introduces a groundbreaking class of indenoimidazole-based compounds, specifically designed to serve as high-performance electron transport materials in organic light-emitting diodes (OLEDs). These novel derivatives, characterized by the general Formula I, exhibit exceptional carrier transport capabilities and superior film-forming properties, addressing critical bottlenecks in the commercialization of next-generation displays. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a significant leap forward, offering a pathway to devices with markedly reduced startup voltages and enhanced luminous efficiency. The structural versatility of these compounds allows for precise tuning of electronic properties, making them ideal candidates for both small molecule and polymeric OLED architectures.
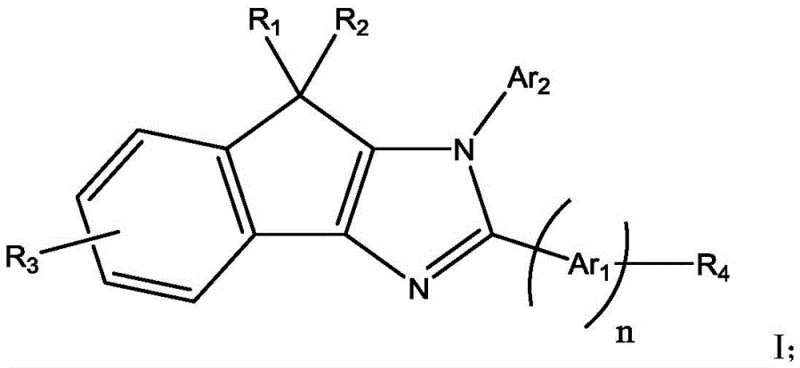
The development of efficient electron transport layers (ETL) has long been a challenge in OLED manufacturing, where conventional materials often suffer from poor electron mobility or unstable morphological characteristics under operational stress. Traditional materials, such as the widely used Alq3 (tris(8-hydroxyquinolinato)aluminum), while effective, frequently require higher driving voltages to achieve desired brightness levels, which inevitably leads to increased power consumption and accelerated device degradation. Furthermore, the synthesis of some high-performance alternatives can be prohibitively complex, involving harsh conditions or expensive catalysts that hinder cost reduction in electronic chemical manufacturing. The indenoimidazole scaffold presented in this patent offers a robust alternative, combining a rigid fused-ring system with flexible substitution patterns that facilitate balanced charge injection and transport. This structural innovation directly translates to practical device improvements, as evidenced by comparative data showing substantial reductions in turn-on voltage and significant extensions in operational half-life compared to standard benchmarks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional electron transport materials often face intrinsic limitations related to their electronic structure and physical stability. Many existing compounds possess relatively low electron mobility, necessitating thicker layers or higher electric fields to drive current, which negatively impacts the overall power efficiency of the display panel. Additionally, the morphological stability of amorphous organic films is a persistent issue; poor film-forming properties can lead to crystallization or phase separation over time, creating dark spots and reducing the uniformity of light emission. From a supply chain perspective, the synthesis of complex heterocyclic compounds often involves multiple steps with low overall yields, generating significant chemical waste and increasing the environmental footprint of production. These factors collectively contribute to higher manufacturing costs and longer lead times for high-purity electronic chemicals, posing challenges for supply chain heads aiming to maintain continuous production schedules.
The Novel Approach
The novel indenoimidazole derivatives disclosed in CN107573328B address these deficiencies through a strategically designed molecular architecture that promotes efficient electron delocalization and stable amorphous states. By incorporating bulky aryl and heteroaryl substituents at specific positions on the indenoimidazole core, the compounds achieve a balance between high carrier mobility and excellent thermal stability. The synthesis route described in the patent utilizes accessible starting materials and standard catalytic processes, such as palladium-catalyzed cross-coupling, which are highly amenable to scale-up. This approach not only simplifies the purification process—often a major cost driver in fine chemical synthesis—but also ensures consistent batch-to-batch quality, a critical requirement for commercial scale-up of complex polymer additives and small molecule dopants. The result is a material that not only performs better in the device but is also more economically viable to produce at an industrial scale.
Mechanistic Insights into Indenoimidazole-Based Electron Transport
The superior performance of these indenoimidazole compounds can be attributed to their unique electronic configuration and intermolecular interactions. The fused ring system provides a rigid planar structure that facilitates pi-pi stacking, thereby enhancing intermolecular electron hopping rates. Substituents such as carbazole, fluorene, or pyridine derivatives further modulate the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels, ensuring optimal alignment with adjacent layers for efficient charge injection. Experimental data from the patent indicates carrier mobilities ranging from 3.2 × 10^-5 cm²/Vs to 8.8 × 10^-5 cm²/Vs, which are competitive with or superior to many state-of-the-art materials. This high mobility allows for thinner electron transport layers, reducing the overall resistance of the device and lowering the operating voltage. Furthermore, the steric bulk introduced by groups like triphenylamine or spirofluorene prevents excessive aggregation, maintaining the amorphous nature of the film and preventing crystallization during prolonged operation.
Impurity control is another critical aspect of the mechanistic success of this synthesis. The multi-step pathway, involving condensation, Grignard addition, and cyclization, is designed to minimize side reactions that could generate deep-level traps for charge carriers. For instance, the use of specific solvents like dichlorobenzene and controlled temperature profiles during the cyclization step ensures high regioselectivity. The final purification via silica gel column chromatography and recrystallization effectively removes residual catalysts and unreacted intermediates, which is essential for achieving the stringent purity specifications required for OLED applications. This rigorous control over the impurity profile directly correlates with the observed improvements in device half-life, as fewer impurities mean fewer sites for non-radiative recombination or chemical degradation within the emissive layer.
How to Synthesize Indenoimidazole Derivatives Efficiently
The synthesis of these high-performance materials follows a logical and scalable four-step sequence that maximizes yield while minimizing waste. The process begins with the condensation of amidine derivatives with chalcones to form the initial imidazole ring, followed by the introduction of functional groups via Grignard reagents. Subsequent acid-catalyzed cyclization creates the rigid indeno-core, and the final step typically involves a Suzuki-Miyaura coupling to attach the desired aryl amines or heterocycles. This modular approach allows for the rapid generation of a diverse library of compounds by simply varying the boronic acid or halide precursors in the final step. Detailed standardized synthesis steps for specific embodiments like CJH-P16 and CJH-P60 are provided in the patent examples, demonstrating yields of up to 84% for the final coupling step.
- Condensation of 4-bromo-N-phenylbenzamidine with chalcone derivatives using ferric chloride and iodine catalysts in dichlorobenzene at 110°C.
- Grignard addition reaction using phenylmagnesium bromide or methylmagnesium iodide in anhydrous tetrahydrofuran to introduce alkyl or aryl groups.
- Acid-catalyzed cyclization in acetic acid and hydrochloric acid to form the rigid indenoimidazole core structure.
- Final palladium-catalyzed Suzuki coupling with boronic acid derivatives to attach functional aryl groups for tuned electronic properties.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this indenoimidazole technology offers compelling strategic advantages beyond mere performance metrics. The synthetic route is designed with manufacturability in mind, utilizing common reagents and avoiding exotic catalysts that might be subject to supply constraints. This inherent simplicity translates into a more resilient supply chain, reducing the risk of production delays caused by raw material shortages. Furthermore, the high yields reported in the patent examples suggest a more efficient use of resources, which is a key driver for cost reduction in manufacturing. By streamlining the purification process and reducing the number of difficult-to-remove impurities, the overall cost of goods sold (COGS) can be significantly optimized without compromising on the quality of the final electronic chemical product.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts in certain steps and utilizes robust reactions like Suzuki coupling which are well-understood in industrial settings. This reduces the complexity of downstream processing, specifically the removal of heavy metal residues, which is often a costly and time-consuming part of pharmaceutical and electronic material production. The high yields achieved in the final coupling steps (e.g., 84% for CJH-P16) indicate a highly efficient atom economy, leading to substantial cost savings in raw material consumption.
- Enhanced Supply Chain Reliability: The starting materials, such as brominated benzamidines and various boronic acids, are commercially available or easily synthesized from commodity chemicals. This accessibility ensures a stable supply base, reducing lead time for high-purity OLED materials. The robustness of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, further enhances the reliability of the manufacturing process, allowing for consistent production schedules even in varying operational environments.
- Scalability and Environmental Compliance: The process is explicitly noted in the patent as being suitable for large-scale production. The use of standard solvents like toluene, ethanol, and ethyl acetate simplifies solvent recovery and recycling systems, aligning with modern environmental compliance standards. The ability to scale from gram-scale laboratory synthesis to multi-kilogram or ton-scale production without significant re-optimization makes this technology an attractive option for companies looking to secure a long-term supply of advanced display materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these indenoimidazole compounds in OLED device fabrication. These insights are derived directly from the experimental data and technical disclosures within patent CN107573328B, providing a clear understanding of the material's capabilities and integration requirements.
Q: What are the key performance advantages of these indenoimidazole compounds in OLED devices?
A: According to patent CN107573328B, these compounds significantly reduce startup voltage (e.g., down to 4.8V compared to 7.8V for Alq3), improve luminous efficiency (up to 5.09 Cd/A), and extend device half-life (over 500 hours in tested configurations).
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent explicitly states that the synthesis and purification methods are simple and suitable for large-scale production. The routes utilize standard reactions like Suzuki coupling and Grignard addition which are well-established in fine chemical manufacturing.
Q: How do these materials compare to conventional electron transport layers?
A: Devices utilizing these indenoimidazole compounds demonstrate superior carrier mobility (ranging from 3.2 × 10^-5 to 8.8 × 10^-5 cm²/Vs) and better film-forming properties compared to traditional materials like Alq3, leading to enhanced brightness and stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indenoimidazole Supplier
As the global demand for high-resolution and energy-efficient displays continues to surge, securing a stable supply of advanced organic electronic materials is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these cutting-edge indenoimidazole derivatives. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch meets the exacting standards required for next-generation OLED panels. We understand the critical nature of material consistency in display manufacturing and have optimized our processes to guarantee batch-to-batch reproducibility.
We invite R&D directors and procurement executives to collaborate with us to explore how these materials can enhance your product portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and device architectures. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate the commercialization of your high-performance organic electroluminescent devices.
