Scalable Synthesis of S-Fluorobutyramid via (R)-2-Sulfonylbutyric Acid Alkyl Ester Intermediates
The global demand for high-efficacy herbicides continues to drive innovation in agrochemical intermediate synthesis, particularly for chiral molecules where specific enantiomers exhibit vastly superior biological activity. Patent CN116217426A introduces a robust and economically viable method for synthesizing S-fluorobutyramid (Beflubutamid-M), the biologically active S-enantiomer of the herbicide Beflubutamid. Unlike traditional racemic mixtures, this S-configured isomer demonstrates herbicidal activity at least 1000 times greater than its R-counterpart, making the development of efficient asymmetric synthesis routes a critical priority for the industry. This patent discloses a novel pathway starting from fermentation-derived (R)-2-hydroxybutyric acid, converting it into a key (R)-2-sulfonylbutyric acid alkyl ester, and subsequently utilizing a stereospecific SN2 substitution to achieve the desired chiral inversion. This approach effectively bypasses the limitations of resolution methods and expensive catalytic hydrogenation, offering a streamlined solution for the production of high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure S-fluorobutyramid has been hindered by significant technical and economic barriers inherent in conventional synthetic strategies. The resolution method, while conceptually simple, suffers from a theoretical maximum yield of 50% for the desired enantiomer and requires costly chiral resolving agents or specialized biological enzymes that are difficult to recover and recycle. Alternatively, the chiral source method relying on R-2-aminobutyric acid involves hazardous diazotization reactions using alkali metal nitrites and hydrobromic acid, generating substantial amounts of toxic wastewater and posing serious safety risks in a plant environment. Furthermore, asymmetric hydrogenation techniques, although elegant, depend heavily on precious metal catalysts such as rhodium or ruthenium complexes, which are not only expensive to procure but also present challenges regarding metal residue removal and catalyst recovery, thereby inflating the overall cost of goods sold (COGS) for the final active ingredient.
The Novel Approach
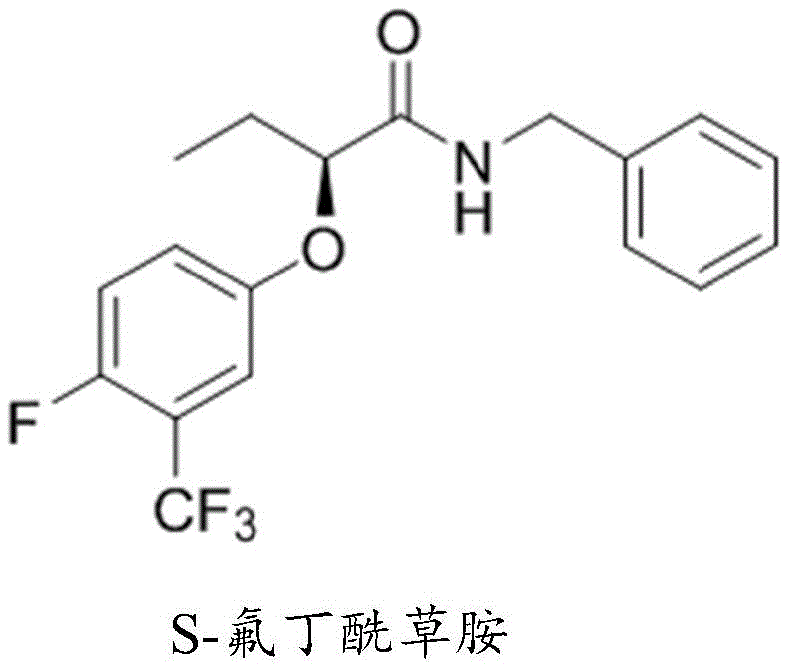
In stark contrast, the methodology outlined in CN116217426A leverages the abundance and low cost of bio-fermented (R)-2-hydroxybutyric acid to construct the chiral backbone. By transforming this readily available starting material into a reactive sulfonyl ester intermediate, the process sets the stage for a highly efficient nucleophilic displacement. The core innovation lies in the use of a sulfonate leaving group, which facilitates a clean SN2 reaction with 4-fluoro-3-(trifluoromethyl)phenol. This strategy not only eliminates the need for expensive transition metal catalysts but also ensures a predictable and complete inversion of stereochemistry, converting the inexpensive (R)-starting material directly into the valuable (S)-product. The result is a synthesis route characterized by fewer steps, safer operating conditions, and significantly reduced environmental impact due to the avoidance of heavy metals and hazardous diazonium chemistry.
Mechanistic Insights into SN2 Chiral Inversion
The cornerstone of this synthetic strategy is the stereospecific nucleophilic substitution that transforms the (R)-configured sulfonyl ester into the (S)-configured phenoxy ether. In this critical step, the phenoxide anion, generated in situ from 4-fluoro-3-(trifluoromethyl)phenol and a base such as potassium carbonate, acts as a strong nucleophile. It attacks the chiral carbon center of the (R)-2-sulfonylbutyric acid alkyl ester from the side opposite to the sulfonate leaving group. This backside attack forces the Walden inversion, a fundamental stereochemical phenomenon where the spatial arrangement of substituents around the chiral center is flipped, much like an umbrella turning inside out in a strong wind. The sulfonate group, being an excellent leaving group due to the resonance stabilization of the resulting sulfonate anion, departs readily, allowing the formation of the new carbon-oxygen bond with high fidelity.
From an impurity control perspective, this mechanism offers distinct advantages over radical or non-stereospecific pathways. Because the SN2 reaction is concerted and highly dependent on steric and electronic factors, side reactions such as elimination (E2) can be minimized by carefully selecting the reaction temperature and solvent polarity. The patent specifies the use of polar aprotic solvents like acetone or DMF, which enhance the nucleophilicity of the phenoxide ion without stabilizing carbocation intermediates that could lead to racemization. Furthermore, the high reactivity of the sulfonyl ester allows the reaction to proceed to completion under relatively mild reflux conditions, reducing the thermal stress on the chiral center and preserving the optical integrity of the molecule. This precise control over the reaction trajectory ensures that the final product maintains an enantiomeric excess (ee) exceeding 95%, meeting the stringent quality standards required for modern agrochemical registrations.
How to Synthesize S-Fluorobutyramid Efficiently
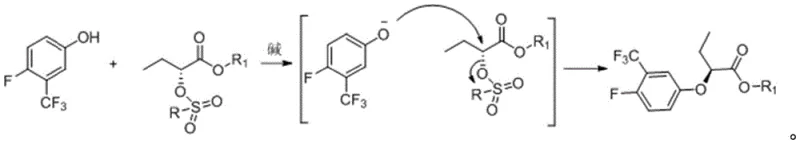
The practical execution of this synthesis involves a logical sequence of unit operations designed for scalability and ease of purification. The process begins with the protection of the carboxylic acid functionality via esterification, followed by the activation of the hydroxyl group through sulfonylation. Once the key chiral building block is prepared, the stereochemical inversion is effected, and the final amide bond is formed either directly from the ester or via an acid chloride intermediate. This flexibility allows manufacturers to optimize the final step based on their specific equipment and reagent availability. For a detailed breakdown of the standardized synthetic steps, including specific molar ratios, temperatures, and workup procedures, please refer to the technical guide below.
- Esterify (R)-2-hydroxybutyric acid with ethanol using an acidic catalyst to form ethyl (R)-2-hydroxybutyrate.
- React the hydroxy ester with a sulfonyl chloride (e.g., benzenesulfonyl chloride) and a base to generate the key (R)-2-sulfonylbutyric acid alkyl ester intermediate.
- Perform an SN2 nucleophilic substitution using 4-fluoro-3-(trifluoromethyl)phenol and a base to invert the chirality and form the S-configured ether intermediate.
- Convert the ester intermediate directly to the amide using benzylamine and a base, or via hydrolysis to the acid followed by acyl chloride formation and amidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfonyl ester route represents a strategic opportunity to optimize the cost structure and reliability of the agrochemical intermediate supply chain. By shifting away from processes reliant on scarce noble metals or complex enzymatic resolutions, manufacturers can secure a more stable raw material base that is less susceptible to market volatility. The use of fermentation-derived starting materials aligns with broader sustainability goals, potentially qualifying the production process for green chemistry incentives and reducing the regulatory burden associated with hazardous waste disposal. Moreover, the high yields reported in the patent examples suggest a significant improvement in material throughput, meaning less raw material is wasted per kilogram of final product, directly contributing to margin expansion.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and precious metal catalysts drastically reduces the direct material costs associated with production. Additionally, the high atom economy of the SN2 substitution step minimizes the generation of by-products, lowering the costs related to waste treatment and solvent recovery. The ability to use common industrial solvents like toluene and acetone further simplifies the supply chain logistics, avoiding the need for specialized or controlled reagents that often carry premium pricing and long lead times.
- Enhanced Supply Chain Reliability: Relying on (R)-2-hydroxybutyric acid, which can be produced on a massive scale via microbial fermentation, decouples the production of S-fluorobutyramid from the fluctuations of the petrochemical market. This bio-based origin ensures a consistent and renewable supply of the chiral precursor, mitigating the risk of shortages that can plague synthetic chiral pools. The robustness of the chemical steps, which do not require sensitive inert atmosphere conditions or ultra-low temperatures, also enhances operational reliability, reducing the likelihood of batch failures due to equipment malfunction or operator error.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, utilizing standard reactor configurations and separation techniques such as distillation and crystallization that are well-understood in the fine chemical industry. The avoidance of heavy metal catalysts simplifies the purification process, removing the need for expensive scavenging resins or complex filtration steps to meet residual metal specifications. This cleaner profile not only reduces the environmental footprint of the manufacturing site but also streamlines the regulatory approval process for new manufacturing sites, facilitating faster technology transfer and global supply network expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of S-fluorobutyramid via the sulfonyl ester pathway. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this technology for your specific application. Understanding these nuances is essential for R&D teams planning pilot runs and procurement teams assessing vendor capabilities.
Q: What is the primary advantage of using the sulfonyl ester route for S-fluorobutyramid?
A: The primary advantage is the utilization of fermentation-derived (R)-2-hydroxybutyric acid as a low-cost chiral pool starting material, combined with a highly efficient SN2 inversion step that ensures excellent optical purity without requiring expensive resolution agents or noble metal catalysts.
Q: How is the stereochemistry controlled in this synthesis?
A: Stereochemistry is controlled through a classic SN2 nucleophilic substitution mechanism. The reaction of the (R)-configured sulfonyl ester with the phenol nucleophile results in a complete inversion of configuration at the chiral center, yielding the desired (S)-configured ether intermediate with high enantiomeric excess.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly suitable for scale-up. It relies on standard unit operations such as esterification, low-temperature sulfonylation, and reflux substitution, utilizing common industrial solvents like toluene, acetone, and dichloroethane, which facilitates easy technology transfer and commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Fluorobutyramid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the development of next-generation agrochemicals. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN116217426A can be successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee the optical purity and chemical integrity of every batch of S-fluorobutyramid we supply. Our commitment to quality assurance means that our clients receive intermediates that consistently meet the demanding requirements of global herbicide formulations.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can drive value and efficiency in your agrochemical projects.
