Scalable Production of High-Purity 8-Azachromones Using Novel PyBrop-Mediated Cyclization
Introduction to Advanced 8-Azachromone Manufacturing
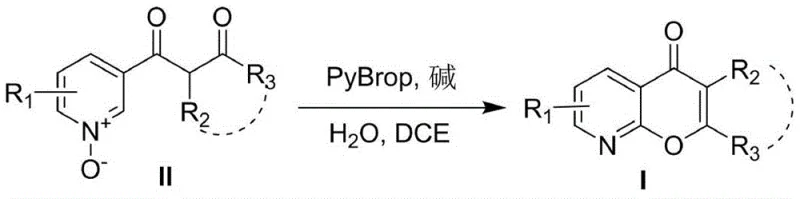
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic scaffolds that offer enhanced bioavailability and structural diversity. Patent CN108329325B introduces a groundbreaking methodology for the concise synthesis of 8-azachromones and 8-azaflavones, addressing critical limitations in existing literature. This technology leverages a PyBrop-mediated cyclization strategy that transforms simple pyridine N-oxides into complex fused ring systems with remarkable efficiency. By utilizing 1,2-dichloroethane as a solvent and operating within a moderate temperature range of 80-85°C, this process eliminates the need for harsh reaction conditions often associated with heterocycle formation. The significance of this innovation lies in its ability to access a broad library of biologically active compounds that were previously difficult to synthesize due to substrate restrictions. For R&D teams focused on oncology and antiviral drug discovery, this platform offers a reliable entry point into high-value chemical space with superior physicochemical properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-azachromone derivatives has been constrained by the reliance on specialized starting materials that are neither commercially abundant nor cost-effective. Traditional routes typically necessitate the use of 2-halo or 2-methoxy substituted nicotinic acid esters as the foundational building blocks. These precursors are not only expensive to procure but also introduce additional synthetic steps to install the requisite leaving groups prior to cyclization. Furthermore, the reactivity of these halogenated species can be unpredictable, often leading to side reactions that compromise the purity of the final API intermediate. The stringent requirement for specific substitution patterns at the 2-position of the pyridine ring severely limits the structural diversity accessible through these classical methods. Consequently, medicinal chemists have faced significant bottlenecks when attempting to explore structure-activity relationships (SAR) around the 8-azachromone core, hindering the rapid development of new therapeutic candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108329325B revolutionizes the landscape by enabling the use of unsubstituted nicotinic acid esters, which are commodity chemicals available at a fraction of the cost. The core innovation involves a tandem activation and cyclization sequence mediated by pyrrolidinylphosphonium bromide hexafluorophosphate (PyBrop) in the presence of a base and water. This approach bypasses the need for pre-functionalized halogenated precursors, streamlining the synthetic route to just three efficient steps from common materials. The reaction demonstrates exceptional substrate tolerance, accommodating various substituents on the pyridine ring and the ketone component without significant loss in yield. By shifting the paradigm from scarce specialty reagents to bulk chemicals, this novel approach drastically reduces the raw material cost basis while simultaneously expanding the chemical space available for exploration.
Mechanistic Insights into PyBrop-Catalyzed Cyclization
The efficacy of this synthesis hinges on the unique activation capability of the PyBrop reagent, which facilitates the formation of the pyrone ring under remarkably mild conditions. Mechanistically, PyBrop acts as a potent coupling agent that activates the carbonyl functionality of the 1,3-dicarbonyl precursor, rendering it susceptible to nucleophilic attack by the oxygen atom of the pyridine N-oxide moiety. The presence of water in the reaction mixture plays a crucial role, likely serving as a proton shuttle or participating in the hydrolysis of intermediate phosphonium species to drive the equilibrium forward. The base, typically sodium acetate or potassium carbonate, neutralizes the acidic byproducts generated during the activation process, maintaining the optimal pH for cyclization. This intricate interplay between the coupling reagent, base, and aqueous additive allows the reaction to proceed smoothly at 80-85°C, avoiding the high-energy barriers that typically require strong acids or Lewis acids. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for maximum throughput and minimal impurity formation.
From an impurity control perspective, the mildness of the PyBrop system offers distinct advantages over traditional acid-catalyzed cyclizations which often promote polymerization or decomposition of sensitive functional groups. The high site selectivity observed in this reaction ensures that the cyclization occurs exclusively at the desired position, minimizing the formation of regioisomers that are notoriously difficult to separate. Additionally, the use of pyridine N-oxides as intermediates provides a handle for electronic tuning; the oxidation state of the nitrogen influences the nucleophilicity of the adjacent oxygen, allowing for fine-tuning of reaction kinetics. This level of control is essential for manufacturing high-purity pharmaceutical intermediates where strict regulatory limits on related substances must be met. The robustness of the catalytic cycle ensures consistent performance across different batches, a critical factor for maintaining supply chain reliability.
How to Synthesize 8-Azachromones Efficiently
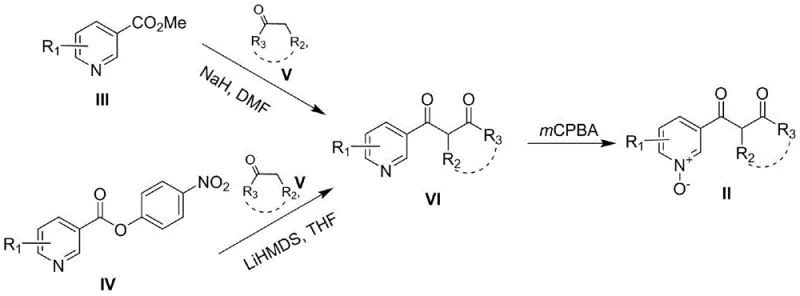
The practical implementation of this technology involves a streamlined three-step sequence that begins with the formation of a 1,3-dicarbonyl intermediate followed by oxidation and final cyclization. The initial step utilizes a Claisen condensation between a nicotinic ester and a ketone, facilitated by strong bases like sodium hydride or LiHMDS, to construct the carbon backbone. Subsequent oxidation with mCPBA converts the pyridine ring into the reactive N-oxide species, setting the stage for the ring-closing event. The final cyclization is the heart of the process, where the PyBrop reagent mediates the intramolecular bond formation to yield the target 8-azachromone scaffold. Detailed standard operating procedures for each stage, including precise stoichiometry, temperature profiles, and workup protocols, are essential for reproducing the high yields reported in the patent literature.
- Perform Claisen ester condensation between methyl nicotinate and a ketone using NaH or LiHMDS to form the 1,3-dicarbonyl intermediate.
- Oxidize the pyridine intermediate using mCPBA in dichloromethane to generate the crucial pyridine N-oxide precursor.
- Execute the key cyclization using PyBrop, sodium acetate, and water in 1,2-dichloroethane at 80-85°C to form the final 8-azachromone scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition driven by raw material accessibility and process simplicity. The shift away from exotic halogenated starting materials to commodity nicotinic esters fundamentally alters the cost structure of the manufacturing process. Since the key reagents are widely produced on a global scale, the risk of supply disruption is minimized, ensuring a steady flow of materials for continuous production campaigns. Moreover, the elimination of stringent anhydrous and oxygen-free requirements for the critical cyclization step reduces the capital expenditure needed for specialized reactor equipment. This operational flexibility allows manufacturers to utilize existing infrastructure more effectively, accelerating the time-to-market for new drug candidates without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of expensive, low-volume precursors with high-volume, low-cost commodity chemicals. By removing the need for 2-halo or 2-methoxy nicotinates, the direct material cost is significantly lowered, creating substantial margin improvements for the final API. Additionally, the high atom economy of the PyBrop-mediated step reduces waste generation, which translates to lower disposal costs and a smaller environmental footprint. The simplified purification protocols, often requiring only standard column chromatography or crystallization, further decrease the operational expenses associated with downstream processing. These cumulative efficiencies result in a highly competitive cost profile that supports long-term commercial viability.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of reagents that are not subject to geopolitical restrictions or complex synthesis chains. Nicotinic acid esters and PyBrop are established industrial chemicals with multiple qualified suppliers worldwide, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production can be maintained even under less-than-ideal logistical scenarios, as the process does not demand ultra-pure solvents or inert atmospheres. This reliability ensures that downstream customers receive their orders on schedule, preventing costly delays in their own drug development timelines. Consistent availability of high-quality intermediates is a key differentiator in the competitive pharmaceutical market.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous reagents and extreme conditions. The moderate temperature range of 80-85°C is easily achievable with standard heating utilities, and the exothermic profile of the reaction is manageable, reducing safety risks during scale-up. Furthermore, the reduced generation of halogenated waste streams aligns with increasingly stringent environmental regulations, facilitating easier permitting and compliance. The ability to produce multi-ton quantities with consistent quality makes this technology an ideal candidate for meeting the growing global demand for 8-azachromone-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this manufacturing route. Understanding these details helps in making informed decisions about process adoption and integration into existing production pipelines.
Q: What are the primary advantages of the PyBrop-mediated synthesis over conventional methods?
A: Unlike conventional methods that rely on scarce and expensive 2-halo or 2-methoxy substituted nicotinic acid esters, this novel approach utilizes readily available unsubstituted nicotinic esters. Furthermore, the key cyclization step operates under mild conditions (80-85°C) without the need for strict anhydrous or oxygen-free environments, significantly simplifying process control and reducing operational costs.
Q: How does the water solubility of 8-azachromones compare to traditional chromones?
A: 8-Azachromones serve as electronic isosteres of chromones but exhibit significantly improved water solubility due to the presence of the nitrogen atom in the pyridine ring. Experimental data indicates a reduction in the LogP value by approximately 0.62 units compared to corresponding chromone derivatives, enhancing their bioavailability potential without requiring the introduction of extraneous solubilizing functional groups.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly amenable to scale-up. The use of common solvents like 1,2-dichloroethane and inexpensive bases such as sodium acetate, combined with the robustness of the PyBrop reagent, allows for straightforward processing. The elimination of sensitive reaction conditions ensures consistent quality and yield during commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Azachromone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the PyBrop-mediated synthesis for 8-azachromones and are fully equipped to bring this technology to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to industrial manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates. Our commitment to technical excellence means we can navigate the complexities of this chemistry to deliver a product that supports your critical drug development milestones.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique needs. Let us be your partner in delivering high-quality 8-azachromone intermediates that drive innovation in the pharmaceutical industry.
