Scalable Production of High-Purity cis,cis-3,5-dimethyl-1-cyclohexanol for Global Pharma
Scalable Production of High-Purity cis,cis-3,5-dimethyl-1-cyclohexanol for Global Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for chiral cyclohexanol derivatives, which serve as critical building blocks for a wide array of bioactive molecules. A significant technological advancement in this domain is documented in patent CN111484393A, which discloses a novel preparation method for cis,cis-3,5-dimethyl-1-cyclohexanol. This specific stereoisomer is highly valued for its structural rigidity and specific spatial arrangement, making it an indispensable pharmaceutical intermediate for the synthesis of complex active pharmaceutical ingredients. The innovation lies not merely in the creation of the molecule but in the strategic orchestration of reaction steps that prioritize stereocontrol without relying on prohibitively expensive chiral catalysts. By leveraging a combination of Knoevenagel condensation, selective hydrogenation, and a sophisticated resolution strategy via esterification, this method addresses the longstanding challenges of impurity control and scalability. For research and development directors evaluating new supply chains, understanding the underlying chemical architecture of this process is essential to ensure the consistency and quality of the final drug substance. The ability to access this compound with high optical purity directly impacts the efficacy and safety profile of downstream medications, underscoring the strategic importance of adopting such advanced synthetic methodologies in modern manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dimethyl-1-cyclohexanol has been fraught with significant technical hurdles that compromise both economic efficiency and product quality. Traditional routes often involve the direct high-temperature and high-pressure hydrogenation of 3,5-dimethyl phenol, a process that demands specialized equipment capable of withstanding extreme conditions which are not universally available in standard fine chemical facilities. Furthermore, these conventional methods frequently suffer from poor chiral selectivity, resulting in a complex mixture of stereoisomers that are notoriously difficult to separate. The presence of these unwanted isomers not only complicates the purification process but also poses a risk to the safety profile of the final pharmaceutical product, necessitating rigorous and costly analytical testing. Another prevalent route involves the reduction of 3,5-dimethyl-2-cyclohexen-1-one, which often yields a chiral mixture requiring stepwise reduction strategies that still fail to guarantee high stereochemical purity. These limitations create substantial bottlenecks for procurement managers seeking reliable sources, as the variability in batch quality can lead to supply chain disruptions and increased costs associated with waste disposal and reprocessing. The reliance on harsh conditions also raises environmental and safety concerns, making these legacy processes less attractive in the context of modern green chemistry initiatives.
The Novel Approach
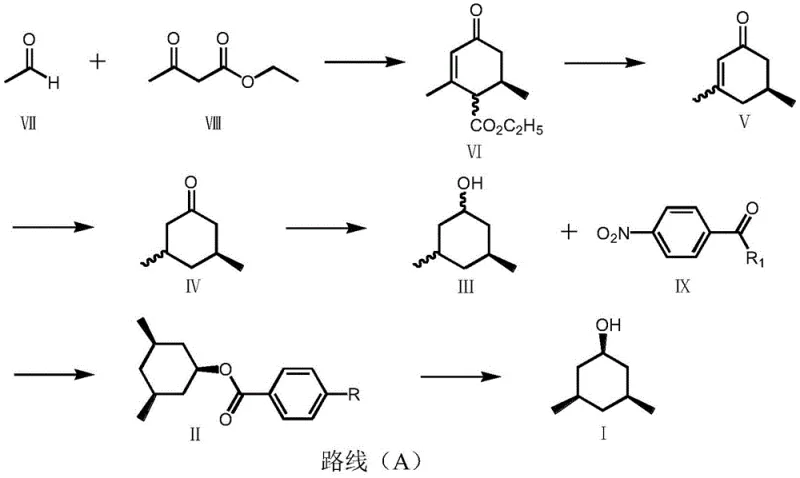
In stark contrast to these legacy methods, the novel approach outlined in the patent data utilizes a stepwise construction of the cyclohexane ring starting from readily available raw materials such as acetaldehyde and ethyl acetoacetate. This strategy allows for precise control over the introduction of methyl groups and the establishment of the ketone functionality before the critical reduction steps occur. The process employs a Knoevenagel condensation to form a cyclohexenone intermediate, followed by hydrolysis and decarboxylation to refine the ring structure. Crucially, the method incorporates a catalytic hydrogenation step under mild conditions to saturate the carbon-carbon double bond, avoiding the extreme pressures associated with phenol reduction. The true breakthrough, however, lies in the subsequent resolution step, where the alcohol intermediate is converted into a substituted benzoate ester. This derivatization enables the separation of stereoisomers based on solubility differences during crystallization, a technique that is both scalable and highly effective. By decoupling the formation of the carbon skeleton from the establishment of chirality, this route offers a level of flexibility and control that is unmatched by direct reduction methods. For supply chain heads, this translates to a more predictable manufacturing timeline and a reduced risk of batch failure, ensuring a steady flow of high-quality intermediates for downstream production.
Mechanistic Insights into Chiral Resolution via Esterification
The core of this synthesis technology rests on a sophisticated chiral resolution mechanism that bypasses the need for asymmetric catalysis. After the initial reduction of the ketone to the alcohol, the resulting mixture contains various stereoisomers that must be separated to isolate the desired cis,cis configuration. The patent describes a process where the crude alcohol mixture is reacted with a substituted benzoic acid, such as p-nitrobenzoic acid, to form a corresponding ester. This chemical modification is pivotal because the resulting esters of different stereoisomers exhibit distinct physical properties, particularly regarding their solubility in specific organic solvents. By carefully selecting the solvent system, such as methanol or ethanol, and controlling the crystallization temperature, the desired isomer can be selectively precipitated out of the solution while the unwanted isomers remain in the mother liquor. This phenomenon relies on the subtle differences in crystal lattice energy and molecular packing efficiency between the diastereomeric esters. The use of p-nitrobenzoic acid is particularly advantageous due to its ability to form stable crystalline structures that facilitate easy filtration and washing. Once the high-purity ester is isolated, a final hydrolysis step cleaves the ester bond to regenerate the free alcohol, now with significantly enhanced optical purity. This mechanism ensures that the final product meets the stringent high-purity pharmaceutical intermediate standards required by regulatory bodies, without the complexity of enzymatic resolution or chiral chromatography.
Furthermore, the impurity control mechanism inherent in this design provides an additional layer of security for the manufacturing process. The intermediate esterification step acts as a purification gate, effectively removing not only stereoisomers but also other organic impurities that may have carried over from the reduction steps. The crystallization process is highly selective, meaning that even if the preceding reaction steps generate minor byproducts, they are unlikely to co-crystallize with the target ester. This robustness is critical for maintaining batch-to-batch consistency, a key metric for research and development directors when qualifying a new supplier. The ability to achieve a product-to-isomer ratio of 30:1 to 100:1 through simple crystallization is a testament to the efficacy of this chemical design. It eliminates the need for complex and expensive purification technologies, thereby streamlining the production workflow. The hydrolysis step that follows is equally well-controlled, utilizing mild alkaline conditions to ensure that the delicate stereochemistry established during crystallization is not compromised. This comprehensive approach to impurity management ensures that the final cis,cis-3,5-dimethyl-1-cyclohexanol is delivered with the highest possible quality, ready for immediate use in sensitive synthetic applications.
How to Synthesize cis,cis-3,5-dimethyl-1-cyclohexanol Efficiently
The implementation of this synthesis route requires a systematic approach to reaction engineering, focusing on the optimization of each step to maximize yield and purity. The process begins with the condensation of acetaldehyde and ethyl acetoacetate, where precise control of temperature and catalyst loading is essential to drive the reaction to completion while minimizing side reactions. Following the formation of the cyclohexenone intermediate, the hydrolysis and decarboxylation steps must be carefully managed to ensure the complete removal of the ester group without degrading the ring structure. The subsequent hydrogenation requires the selection of an appropriate metal catalyst, such as palladium on carbon, to achieve full saturation of the double bond under mild pressure. The critical resolution step involves the formation of the p-nitrobenzoate ester, where solvent choice and cooling rates determine the efficiency of the isomer separation. Finally, the hydrolysis of the purified ester releases the target alcohol, completing the synthesis. For detailed operational parameters and safety guidelines, the standardized synthesis steps are provided below.
- Perform Knoevenagel condensation between acetaldehyde and ethyl acetoacetate to form the cyclohexenone intermediate.
- Execute hydrolysis and decarboxylation followed by catalytic hydrogenation to saturate the ring structure.
- Conduct chiral resolution via p-nitrobenzoate ester formation and crystallization, followed by final hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit is the significant reduction in manufacturing costs driven by the use of commodity raw materials. Acetaldehyde and ethyl acetoacetate are bulk chemicals with stable pricing and widespread availability, insulating the production process from the volatility associated with specialized chiral reagents. Moreover, the elimination of expensive chiral catalysts removes a major cost center from the bill of materials, allowing for more competitive pricing structures without sacrificing quality. The mild reaction conditions further contribute to cost efficiency by reducing energy consumption and extending the lifespan of production equipment. This economic model supports cost reduction in fine chemical manufacturing, enabling pharmaceutical companies to optimize their overall production budgets. The simplicity of the operation also means that the process can be transferred to manufacturing sites with standard equipment capabilities, reducing the need for capital investment in specialized high-pressure reactors.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing a classical chemical resolution strategy instead of relying on costly asymmetric hydrogenation catalysts or enzymatic processes. By avoiding the use of precious metals like rhodium or ruthenium often required for chiral synthesis, the raw material costs are drastically lowered. Additionally, the recovery and recycling of solvents used in the crystallization steps further enhance the economic viability of the route. The high yield of the desired isomer through crystallization minimizes waste generation, reducing the costs associated with waste treatment and disposal. This efficient use of resources ensures that the overall cost of goods sold remains low, providing a strong value proposition for long-term supply contracts.
- Enhanced Supply Chain Reliability: The reliance on easily obtainable raw materials ensures a robust and resilient supply chain that is less susceptible to disruptions. Unlike processes that depend on single-source specialty reagents, this method can be sustained by multiple suppliers of bulk chemicals, mitigating the risk of shortages. The mild operating conditions also reduce the likelihood of equipment failure or safety incidents, which can often lead to unplanned production downtime. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it allows for consistent production scheduling and reliable delivery timelines. The scalability of the process means that production volumes can be ramped up quickly to meet surges in demand without compromising product quality, ensuring continuity of supply for critical drug manufacturing.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for commercial scale-up of complex chiral intermediates, with reaction steps that translate seamlessly from laboratory to pilot and full-scale production. The absence of extreme high-pressure and high-temperature conditions simplifies the engineering requirements for large reactors, making it easier to implement in existing facilities. Furthermore, the process aligns with environmental compliance standards by minimizing the use of hazardous reagents and generating less toxic waste compared to traditional phenol reduction methods. The solvents used are common organic liquids that can be effectively managed through standard recovery systems. This environmental friendliness not only reduces regulatory burdens but also enhances the sustainability profile of the supply chain, a growing priority for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cis,cis-3,5-dimethyl-1-cyclohexanol. These answers are derived from the specific technical disclosures and beneficial effects outlined in the patent data, providing clarity on the capabilities and limitations of this synthesis method. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their manufacturing workflows. The information covers aspects of purity, scalability, and process safety, ensuring a comprehensive understanding of the technology.
Q: What is the chiral purity achievable with this synthesis method?
A: The patented process achieves a product-to-isomer ratio ranging from 30:1 to 100:1, ensuring high stereochemical integrity suitable for sensitive pharmaceutical applications.
Q: Does this method require expensive chiral catalysts?
A: No, the method relies on classical chemical resolution using substituted benzoic acid derivatives and crystallization, avoiding the need for costly transition metal chiral catalysts.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes mild temperatures and pressures, avoiding extreme high-pressure hydrogenation of phenols, which makes it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis,cis-3,5-dimethyl-1-cyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development and commercialization. Our team of expert chemists has extensively analyzed the synthetic pathway described in CN111484393A and possesses the technical capability to execute this route with precision and efficiency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale manufacturing. Our facilities are equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of cis,cis-3,5-dimethyl-1-cyclohexanol meets the highest industry standards. We are committed to being a strategic partner who not only supplies chemicals but also provides technical solutions to optimize your production processes.
We invite you to engage with our technical team to discuss how this advanced synthesis route can benefit your specific project requirements. By leveraging our manufacturing expertise, you can achieve significant process improvements and cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your needs. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you secure a reliable supply of this critical intermediate and accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
