Revolutionizing Amine Synthesis: A Solvent-Free, Aldehyde-Catalyzed Approach For Industrial Scale-Up
Introduction To Breakthrough Amine Synthesis Technology
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, more cost-effective synthetic routes. Patent CN102617259A introduces a transformative methodology for synthesizing amine compounds through the direct dehydration coupling of alcohols and amines. This innovation stands out by replacing costly and environmentally burdensome transition metal catalysts with inexpensive, non-metallic aldehyde catalysts. For R&D directors and procurement specialists alike, this represents a significant shift towards sustainable chemistry that does not compromise on efficiency or yield. The technology leverages readily available alcohols as alkylating agents, bypassing the need for hazardous halides or complex activation steps often seen in classical alkylation protocols.
Furthermore, the process is designed to operate under solvent-free conditions, which drastically reduces the volume of organic waste generated during production. This aligns perfectly with modern regulatory pressures regarding volatile organic compound (VOC) emissions and waste disposal costs. By utilizing simple inorganic bases such as potassium carbonate or sodium hydroxide, the reaction system remains robust and easy to handle. The ability to conduct these reactions under air, rather than requiring strict inert atmospheres, further lowers the barrier to entry for industrial scale-up, making it an attractive option for the reliable pharmaceutical intermediates supplier seeking to optimize their production lines.

The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
Historically, the synthesis of secondary and tertiary amines has relied heavily on transition metal-catalyzed cross-coupling reactions or reductive amination processes. Traditional Hofmann alkylation often suffers from poor selectivity, leading to over-alkylation and difficult separation of poly-alkylated byproducts. Moreover, methods employing palladium or copper catalysts necessitate the use of excessive amounts of strong bases and expensive ligands to maintain catalytic activity. These transition metals are not only costly but also pose significant challenges in terms of residual metal contamination in the final active pharmaceutical ingredients (APIs), requiring additional purification steps that drive up manufacturing costs.
Additionally, many conventional catalytic systems are highly sensitive to oxygen and moisture, demanding sophisticated reactor setups capable of maintaining rigorous inert gas environments. This sensitivity increases operational complexity and capital expenditure for manufacturing facilities. Reductive amination, another common pathway, requires the use of aldehydes or ketones alongside stoichiometric reducing agents like sodium borohydride or hydrogen gas, introducing safety hazards and atom economy issues. The cumulative effect of these limitations is a synthesis route that is economically inefficient and environmentally taxing, prompting the industry to seek alternatives that offer better sustainability profiles without sacrificing yield.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues by employing a catalytic amount of an aldehyde, often corresponding to the alcohol substrate, to facilitate the dehydration coupling. For instance, when benzyl alcohol is used as the substrate, benzaldehyde serves as the catalyst. This organocatalytic strategy eliminates the need for precious metals entirely, thereby removing the risk of heavy metal contamination in the product stream. The reaction proceeds efficiently at moderate temperatures, typically between 100°C and 150°C, which is energy-efficient compared to high-temperature pyrolysis or extreme pressure conditions required by other methods.
Moreover, the solvent-free nature of this reaction is a game-changer for cost reduction in pharmaceutical intermediates manufacturing. By removing the solvent, the reaction concentration is maximized, leading to higher space-time yields and smaller reactor footprints for the same output. The workup procedure is simplified significantly; since there is no solvent to evaporate and no metal catalyst to filter, the crude product can often be purified directly via crystallization or simple chromatography. This streamlined workflow enhances the overall throughput of the manufacturing process, allowing for faster turnaround times and improved supply chain reliability for high-purity amine derivatives needed in drug discovery and development.
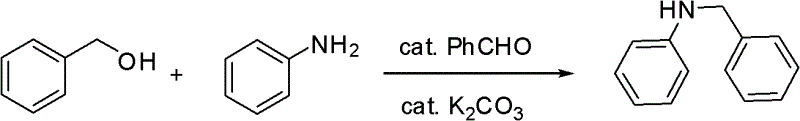
Mechanistic Insights Into Aldehyde-Catalyzed Dehydrative Coupling
The mechanistic pathway of this transformation is believed to involve a redox-neutral cycle where the aldehyde catalyst plays a pivotal role in activating the alcohol substrate. Initially, the alcohol may undergo a transient oxidation to form an aldehyde intermediate, potentially facilitated by the added aldehyde catalyst acting as a hydride acceptor or through a hemiacetal exchange mechanism. This in situ generated aldehyde then condenses with the amine nucleophile to form an imine or iminium ion intermediate. The subsequent reduction of this imine species back to the amine product completes the cycle, regenerating the catalytic aldehyde or utilizing the hydrogen equivalents derived from the initial alcohol oxidation.
This "borrowing hydrogen" or hydrogen-autotransfer-like mechanism ensures that no external reducing agents are required, enhancing the atom economy of the process. The presence of the inorganic base is crucial for deprotonating the amine to increase its nucleophilicity and facilitating the elimination of water during the imine formation step. The choice of base, ranging from mild carbonates to stronger hydroxides like CsOH, allows for fine-tuning the reaction kinetics to suit different substrate electronic properties. For electron-deficient amines or sterically hindered alcohols, stronger bases and higher temperatures might be employed to drive the equilibrium towards the desired amine product, ensuring high conversion rates even with challenging substrates.
Impurity control is inherently superior in this system due to the absence of metal-mediated side reactions such as homocoupling or beta-hydride elimination often seen with palladium catalysts. The primary byproducts are typically water and unreacted starting materials, which are easily separated. The high selectivity observed in the patent examples, with isolated yields frequently exceeding 90% for model substrates like N-benzylbenzenesulfonamide, underscores the cleanliness of the reaction profile. This purity is critical for pharmaceutical applications where strict impurity thresholds must be met, reducing the burden on quality control laboratories and minimizing batch rejection rates during commercial production.
How To Synthesize N-Benzylbenzenesulfonamide Efficiently
To implement this synthesis route effectively, precise control over reaction parameters is essential. The protocol involves charging a reactor with the alcohol and amine in a molar ratio ranging from 3:1 to 1:3, depending on the relative cost and volatility of the reagents. A catalytic amount of the corresponding aldehyde, typically between 1 mol% and 30 mol%, is added along with 5 mol% to 40 mol% of an inorganic base. The mixture is then heated to the optimal temperature window, usually around 135°C for benzyl alcohol derivatives, and stirred for a period of 6 to 24 hours.
- Charge a reactor with the target alcohol, amine substrate, catalytic amount of corresponding aldehyde (1-30 mol%), and inorganic base (1-80 mol%).
- Heat the reaction mixture to a temperature range of 100-150°C under air or inert atmosphere with stirring for 6-24 hours.
- Monitor reaction progress via TLC or GC-MS, then purify the crude product using flash column chromatography to isolate the high-purity amine derivative.
Commercial Advantages For Procurement And Supply Chain Teams
From a procurement perspective, the shift to this aldehyde-catalyzed technology offers substantial strategic benefits. The elimination of expensive transition metal catalysts directly translates to significant cost reduction in manufacturing. Precious metals like palladium are subject to volatile market pricing and supply constraints; replacing them with commodity chemicals like benzaldehyde stabilizes the raw material cost structure. Furthermore, the solvent-free operation reduces the procurement volume of organic solvents, which are increasingly regulated and taxed due to their environmental impact. This reduction in material input lowers the overall variable cost per kilogram of the produced amine intermediate.
Enhanced supply chain reliability is another critical advantage. Since the reaction can be performed under air without the need for specialized inert gas infrastructure, manufacturing can be decentralized or scaled up in facilities with standard equipment capabilities. This flexibility reduces lead time for high-purity pharmaceutical intermediates by removing bottlenecks associated with specialized reactor availability. The simplicity of the workup process also means that production cycles are shorter, allowing for more batches to be run within the same timeframe. This increased throughput capability ensures a steady supply of key building blocks, mitigating the risk of stockouts that can delay downstream drug development programs.
Scalability and environmental compliance are seamlessly integrated into this process design. The absence of heavy metals simplifies waste treatment protocols, as effluent streams do not require complex metal scavenging or hazardous waste disposal procedures. This aligns with global trends towards greener chemistry and helps manufacturers meet stringent environmental, social, and governance (ESG) targets. The commercial scale-up of complex amine derivatives becomes more feasible as the process safety profile is improved by avoiding pyrophoric reagents or high-pressure hydrogenation steps. Consequently, this technology supports sustainable growth strategies for chemical enterprises aiming to expand their portfolio of eco-friendly fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding this synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on implementation.
Q: What are the primary advantages of this aldehyde-catalyzed method over traditional transition metal catalysis?
A: Unlike traditional methods requiring expensive palladium or copper catalysts that are air-sensitive and difficult to remove, this technology utilizes cheap, non-transition metal aldehyde catalysts. It operates under solvent-free conditions, significantly reducing environmental pollution and downstream purification costs associated with heavy metal removal.
Q: Can this synthesis method be performed under ambient atmospheric conditions?
A: Yes, one of the key operational benefits described in patent CN102617259A is that the reaction can proceed effectively under air conditions, eliminating the need for rigorous inert gas protection systems often required for sensitive organometallic catalysts, thereby simplifying equipment requirements.
Q: What is the substrate scope for the alcohol and amine components in this reaction?
A: The method demonstrates broad applicability, accommodating various substituted benzyl alcohols, heteroaryl alcohols, and aliphatic alcohols. It is compatible with diverse amine nucleophiles including sulfonamides, anilines, and heterocyclic amines, yielding products with high isolation yields ranging typically from 65% to 97%.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable N-Benzylbenzenesulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this aldehyde-catalyzed synthesis technology for the production of high-value amine intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing reaction conditions to maximize yield and purity, ensuring that every batch meets stringent purity specifications required by global regulatory bodies. We utilize rigorous QC labs equipped with advanced analytical instrumentation to verify the absence of residual catalysts and impurities, guaranteeing the quality of our pharmaceutical intermediates.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific project needs. Whether you require custom synthesis of novel sulfonamide derivatives or scale-up of established aniline couplings, our experts can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timeline while achieving your sustainability and cost-efficiency goals through innovative chemical manufacturing solutions.
