Advanced Pyridine-Bridged NHC Ruthenium Catalysts for Scalable Pharmaceutical Manufacturing
Advanced Pyridine-Bridged NHC Ruthenium Catalysts for Scalable Pharmaceutical Manufacturing
The landscape of transition metal catalysis is undergoing a significant transformation with the introduction of robust N-heterocyclic carbene (NHC) ligand systems, as detailed in the pioneering patent CN107442172B. This intellectual property discloses a novel class of pyridine-bridged nitrogen heterocyclic carbene triphenylphosphine ruthenium hydrochloride catalysts, engineered specifically to address the challenges of efficiency and stability in homogeneous hydrogenation. These catalysts represent a critical advancement for the fine chemical industry, particularly in the valorization of cyclic carbonates—a key pathway for converting captured carbon dioxide into high-value commodities like methanol and diols. For research and development directors seeking next-generation catalytic solutions, this technology offers a compelling alternative to traditional phosphine-based systems, combining the electronic benefits of NHCs with the structural rigidity of a pyridine bridge to create a highly active and durable catalytic species suitable for complex organic transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydrogenation of cyclic carbonates has relied heavily on pincer-type PNP-ruthenium complexes or heterogeneous copper-silica nanocomposites, each presenting distinct operational drawbacks that hinder large-scale adoption. Earlier generations of PNP-Ru catalysts, while effective, often require stringent exclusion of air and moisture due to the susceptibility of phosphine ligands to oxidation, which can lead to rapid catalyst deactivation and inconsistent batch-to-batch performance. Furthermore, heterogeneous systems like Cu-SiO2, although easier to separate, frequently demand excessively high temperatures exceeding 160 °C to achieve quantitative conversion, thereby increasing energy consumption and potentially compromising the thermal stability of sensitive functional groups within the substrate. These legacy methods also struggle with the economic implications of ligand synthesis, where complex multistep preparations for specialized pincer ligands drive up the overall cost of goods, making the process less attractive for cost-sensitive applications in the production of bulk pharmaceutical intermediates or fuel additives.
The Novel Approach
In stark contrast, the novel approach utilizing pyridine-bridged NHC ruthenium catalysts leverages the unique electronic properties of N-heterocyclic carbenes to overcome these stability and activity barriers. The incorporation of the NHC ligand provides a stronger sigma-electron donor effect compared to traditional phosphines, which significantly enriches the electron density at the ruthenium center and strengthens the metal-ligand bond against thermal and oxidative degradation. This structural innovation allows the catalyst to maintain high activity under demanding reaction conditions, such as 140 °C and 50 atm of hydrogen pressure, without the rapid decomposition observed in older systems. Additionally, the modular nature of the imidazolium salt precursors allows for facile tuning of steric and electronic parameters through simple substitution on the nitrogen atoms, enabling chemists to optimize the catalyst for specific substrate profiles without resorting to prohibitively expensive synthetic routes. This flexibility ensures that the new catalyst family can be adapted for a wide range of cyclic carbonate derivatives, offering a versatile platform for sustainable chemical manufacturing.
Mechanistic Insights into Pyridine-Bridged NHC Ruthenium Catalysis
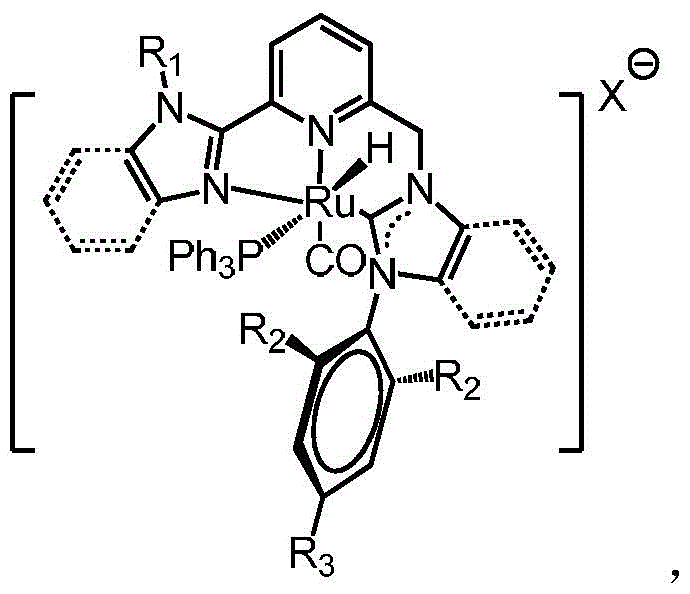
The exceptional performance of these catalysts stems from the synergistic interaction between the rigid pyridine backbone and the strongly donating N-heterocyclic carbene moiety, which creates a highly stabilized coordination environment for the ruthenium atom. Mechanistically, the in situ generation of the active carbene species via deprotonation of the imidazolium salt with a strong base like LiHMDS ensures that the reactive intermediate is formed immediately prior to coordination with the ruthenium precursor, minimizing side reactions and maximizing the yield of the desired cationic complex. Once formed, the ruthenium center is shielded by the bulky triphenylphosphine and the chelating NHC-pyridine framework, which prevents the formation of inactive dimeric species that often plague homogeneous catalysis. This monomeric stability is crucial for maintaining a high turnover number (TON) during the hydrogenation cycle, as it ensures that the active site remains accessible to the substrate while being protected from irreversible deactivation pathways. The presence of the hydride and carbonyl ligands further facilitates the activation of molecular hydrogen and the subsequent insertion into the carbonate bond, driving the reaction towards the formation of methanol and diol products with high selectivity.
From an impurity control perspective, the robustness of the NHC-Ru bond significantly reduces the leaching of ruthenium metal into the final product stream, a critical parameter for pharmaceutical applications where residual metal limits are strictly regulated. The strong sigma-donation of the carbene ligand creates a deep potential well for the metal, making it energetically unfavorable for the ligand to dissociate under the reaction conditions, thereby preventing the release of free ruthenium ions that could catalyze unwanted side reactions or contaminate the product. Furthermore, the use of well-defined precursors and the precipitation of the catalyst as a stable solid allow for rigorous purification steps, such as recrystallization from dichloromethane and pentane, which effectively remove unreacted starting materials and phosphine oxides. This high level of purity in the catalyst itself translates directly to a cleaner reaction profile, simplifying downstream processing and reducing the burden on purification units to remove trace metal contaminants from the final API intermediates or fine chemicals.
How to Synthesize Pyridine-Bridged NHC Ruthenium Catalyst Efficiently
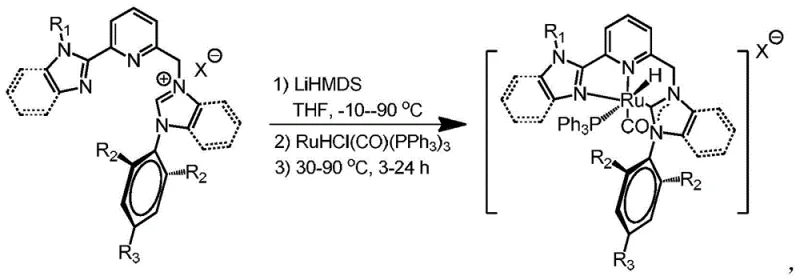
The synthesis of these high-performance catalysts follows a streamlined protocol designed for reproducibility and scalability, beginning with the careful preparation of the pyridine-bridged imidazolium salt ligand precursor. The process involves the deprotonation of this salt under inert atmosphere conditions using lithium bis(trimethylsilyl)amide (LiHMDS) in anhydrous tetrahydrofuran at cryogenic temperatures ranging from -90 °C to -60 °C to generate the free carbene intermediate. This sensitive intermediate is then transferred to a reaction vessel containing tris(triphenylphosphine)carbonylruthenium(II) chloride, where the mixture is heated to promote the displacement of phosphine ligands and the formation of the stable NHC-ruthenium complex. The detailed standardized synthesis steps, including precise stoichiometric ratios, temperature ramping rates, and workup procedures, are outlined in the guide below to ensure consistent production of the catalyst with optimal activity.
- Dissolve pyridine-bridged imidazolium salt and LiHMDS in anhydrous THF under nitrogen at low temperature (-90 to -60 °C) to generate the active carbene intermediate.
- Transfer the carbene solution to a vessel containing RuHCl(CO)(PPh3)3 and heat to 30-90 °C for 3-24 hours to facilitate coordination.
- Cool the reaction, filter the precipitated solid, recrystallize using dichloromethane and n-pentane, and vacuum dry to obtain the final catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel catalyst technology presents a strategic opportunity to optimize manufacturing costs and enhance supply security for critical chemical intermediates. The inherent stability of the N-heterocyclic carbene ligands implies a significantly extended catalyst lifetime compared to traditional phosphine-based systems, which directly correlates to a reduction in the frequency of catalyst replenishment and a decrease in the overall consumption of precious ruthenium metal per ton of product. This durability minimizes the operational risks associated with catalyst failure during long batch runs, ensuring more predictable production schedules and reducing the likelihood of costly downtime or batch rejections due to incomplete conversion. Furthermore, the simplified synthetic route for the ligand precursors, which avoids the complex multi-step syntheses often required for specialized pincer ligands, suggests a more resilient supply chain for the raw materials needed to produce the catalyst itself.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the enhanced stability and activity of the catalyst, which allows for lower catalyst loadings to achieve the same throughput as less active systems. By eliminating the need for excessive catalyst quantities and reducing the frequency of reactor cleaning caused by catalyst decomposition residues, manufacturers can realize substantial savings in both raw material costs and operational expenditures. Additionally, the ability of the catalyst to operate efficiently under standard hydrogenation conditions without requiring exotic additives or extreme pressures contributes to lower energy consumption and reduced wear on high-pressure equipment, further driving down the total cost of ownership for the hydrogenation process.
- Enhanced Supply Chain Reliability: The modular design of the catalyst library, which allows for variations in the R-groups on the imidazolium ring, provides a buffer against supply chain disruptions for specific chemical building blocks. If a particular substituent becomes scarce or expensive, the synthetic flexibility of the system allows for the rapid substitution with alternative alkyl or aryl groups without necessitating a complete redesign of the catalytic process. This adaptability ensures a continuous supply of active catalyst batches, mitigating the risk of production halts due to raw material shortages and providing procurement teams with greater leverage in negotiating contracts with multiple suppliers for the generic precursor materials.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the high selectivity of the catalyst minimizes the formation of by-products, thereby reducing the volume of waste solvent and hazardous chemicals that require treatment and disposal. The quantitative conversion of cyclic carbonates to methanol and diols aligns with green chemistry principles by maximizing atom economy and reducing the carbon footprint of the manufacturing process. As regulatory pressures regarding industrial emissions and waste management intensify, adopting a catalyst system that inherently produces cleaner reaction mixtures offers a proactive strategy for maintaining compliance and avoiding potential fines or remediation costs associated with environmental violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of pyridine-bridged NHC ruthenium catalysts in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, offering a factual basis for evaluating the technology's fit within your existing manufacturing infrastructure. Understanding these nuances is essential for making informed decisions about process integration and long-term technology partnerships.
Q: What are the primary advantages of NHC ligands over traditional phosphine ligands in this catalyst system?
A: N-heterocyclic carbenes (NHCs) offer superior sigma-electron donating capabilities compared to organophosphines, which significantly increases the electron density at the ruthenium metal center. This electronic enhancement stabilizes the metal-carbon bond against air, water, and high temperatures, resulting in a catalyst with prolonged operational lifetime and robustness under rigorous industrial hydrogenation conditions.
Q: Can this catalyst system be applied to the conversion of CO2-derived feedstocks?
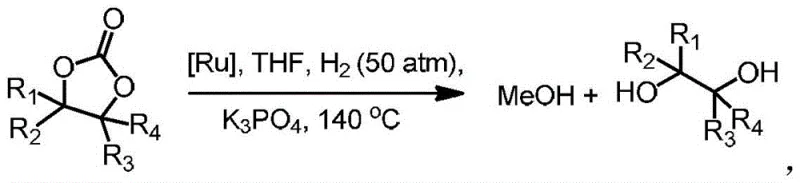
A: Yes, the catalyst is specifically designed for the hydrogenation of cyclic carbonates, which are key intermediates derived from the fixation of carbon dioxide with epoxides or diols. By efficiently converting these carbonates into methanol and corresponding diols, the technology supports green chemistry initiatives aimed at valorizing greenhouse gases into valuable chemical building blocks.
Q: What are the typical reaction conditions required for the catalytic hydrogenation process?
A: The hydrogenation process typically operates under moderate to high pressure conditions, specifically around 50 atm of hydrogen gas, and elevated temperatures near 140 °C in a tetrahydrofuran solvent system. The use of a base such as potassium phosphate facilitates the reaction, ensuring quantitative conversion of the cyclic carbonate substrate into the desired alcohol products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine-Bridged NHC Ruthenium Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced transition metal catalysis in driving the next generation of sustainable chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate we deliver performs consistently in your critical hydrogenation processes. We are committed to supporting your R&D and production teams with the highest quality materials and technical expertise available in the market.
We invite you to engage with our technical procurement team to discuss how this novel catalyst technology can be integrated into your specific workflow to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this robust NHC-ruthenium system for your cyclic carbonate hydrogenation needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your supply chain is optimized for both performance and profitability in the competitive global marketplace.
