Advanced Synthesis of Triarylamine-Substituted Terpyridine Ruthenium Complexes for Molecular Electronics
Advanced Synthesis of Triarylamine-Substituted Terpyridine Ruthenium Complexes for Molecular Electronics
The rapid evolution of molecular electronics demands precise engineering of organic-inorganic hybrid structures capable of efficient long-range charge transport. Patent CN113150036B discloses a groundbreaking methodology for synthesizing triarylamine-substituted terpyridine ruthenium complexes, which serve as robust models for molecular wires. This technology addresses the critical need for materials that exhibit strong electronic interactions between terminal groups, a prerequisite for next-generation optoelectronic devices. The core innovation lies in the strategic modification of the terpyridine ligand with triarylamine moieties prior to metal coordination, creating a unique architecture that merges the stability of ruthenium complexes with the redox versatility of organic amines. 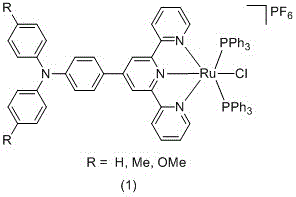 As detailed in the patent, these complexes are not merely static structures but dynamic systems where the chlorine atom on the ruthenium center offers a handle for further functionalization. This flexibility allows chemists to build increasingly sophisticated multi-redox active centers, paving the way for complex molecular circuitry. For R&D directors seeking reliable molecular wire material suppliers, this patent represents a significant leap forward in designing materials with tunable electrochemical properties. The ability to systematically vary the substituents on the triarylamine group (R = H, Me, OMe) provides a powerful toolkit for optimizing energy levels and charge transfer rates without altering the core metal-ligand framework.
As detailed in the patent, these complexes are not merely static structures but dynamic systems where the chlorine atom on the ruthenium center offers a handle for further functionalization. This flexibility allows chemists to build increasingly sophisticated multi-redox active centers, paving the way for complex molecular circuitry. For R&D directors seeking reliable molecular wire material suppliers, this patent represents a significant leap forward in designing materials with tunable electrochemical properties. The ability to systematically vary the substituents on the triarylamine group (R = H, Me, OMe) provides a powerful toolkit for optimizing energy levels and charge transfer rates without altering the core metal-ligand framework.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing functionalized terpyridine metal complexes often suffer from inefficiencies related to late-stage functionalization or harsh reaction conditions that compromise yield and purity. In many conventional routes, attempting to introduce electron-rich amine groups after the formation of the metal complex can lead to competitive coordination or decomposition of the sensitive metal center. Furthermore, standard purification protocols for these intermediates frequently rely on extensive column chromatography, which is not only time-consuming but also difficult to scale for industrial production. The lack of modularity in older methods means that creating a library of derivatives with different electronic properties requires entirely new synthetic pathways for each variant, drastically increasing development time and cost. Additionally, achieving high purity in the final metal complex is often hindered by the presence of unreacted ligands or metal salts that are difficult to separate due to similar solubility profiles. These limitations pose significant barriers for procurement managers looking for cost reduction in electronic chemical manufacturing, as low yields and complex purification drive up the unit price of the final active material.
The Novel Approach
The methodology outlined in CN113150036B overcomes these hurdles through a rational, two-step synthetic strategy that prioritizes modularity and scalability. The first step involves a palladium-catalyzed Buchwald-Hartwig amination to attach the triarylamine group to the terpyridine backbone before metal coordination occurs. This sequence ensures that the sensitive ruthenium center is only introduced once the organic ligand is fully constructed and purified, minimizing the risk of side reactions. 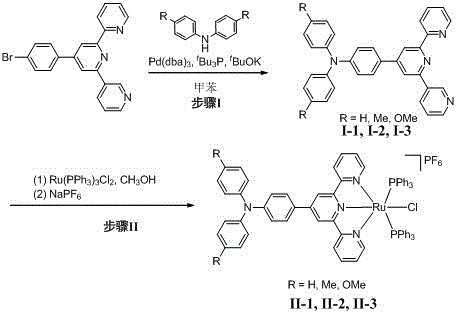 As illustrated in the reaction scheme, the process utilizes robust catalysts like Pd(dba)3 and ligands such as tBu3P to achieve high conversion rates under relatively mild reflux conditions in toluene. The second step involves a straightforward coordination reaction in methanol, followed by an anion exchange with sodium hexafluorophosphate to precipitate the final product. This approach eliminates the need for complex chromatographic separations in the final stage, as the product can be isolated via simple recrystallization. For supply chain heads concerned with the commercial scale-up of complex electronic materials, this streamlined workflow offers a clear path from gram-scale laboratory synthesis to kilogram-level production without sacrificing quality or consistency.
As illustrated in the reaction scheme, the process utilizes robust catalysts like Pd(dba)3 and ligands such as tBu3P to achieve high conversion rates under relatively mild reflux conditions in toluene. The second step involves a straightforward coordination reaction in methanol, followed by an anion exchange with sodium hexafluorophosphate to precipitate the final product. This approach eliminates the need for complex chromatographic separations in the final stage, as the product can be isolated via simple recrystallization. For supply chain heads concerned with the commercial scale-up of complex electronic materials, this streamlined workflow offers a clear path from gram-scale laboratory synthesis to kilogram-level production without sacrificing quality or consistency.
Mechanistic Insights into Pd-Catalyzed Amination and Ru-Coordination
The success of this synthesis hinges on the efficiency of the initial C-N bond formation via palladium catalysis. The mechanism involves the oxidative addition of the aryl bromide (4'-(4-bromophenyl)-terpyridine) to the Pd(0) species, followed by coordination and deprotonation of the diphenylamine substrate. The use of bulky phosphine ligands like tri-tert-butylphosphine is critical here, as they stabilize the active Pd(0) species and facilitate the reductive elimination step to form the C-N bond. This catalytic cycle is highly tolerant of the pyridine nitrogen atoms present in the terpyridine scaffold, which could otherwise poison the catalyst in less optimized systems. By completing this functionalization step first, the process ensures that the resulting triarylamine-substituted terpyridine ligand is structurally defined and free from metal contaminants before it encounters the ruthenium precursor. This sequential logic is essential for maintaining the high purity specifications required for molecular electronics applications, where trace metal impurities can act as charge traps and degrade device performance.
Following ligand synthesis, the coordination step leverages the lability of the triphenylphosphine ligands on the ruthenium precursor. Under reflux in methanol, the tridentate terpyridine ligand displaces the monodentate phosphines and chloride ions to form a stable octahedral geometry around the ruthenium center. The subsequent addition of sodium hexafluorophosphate drives the equilibrium towards the final cationic complex by precipitating the less soluble hexafluorophosphate salt. This anion exchange is a classic technique in coordination chemistry that serves a dual purpose: it replaces the chloride counterion, which might be labile or reactive, with the non-coordinating PF6- anion, thereby enhancing the stability and solubility profile of the complex in organic solvents. From an impurity control perspective, the crystallization of the PF6 salt acts as a final polishing step, excluding unreacted starting materials and byproducts that remain in the mother liquor. This mechanistic understanding allows process chemists to fine-tune reaction parameters such as temperature and stoichiometry to maximize yield while ensuring the structural integrity of the redox-active centers.
How to Synthesize Triarylamine-Substituted Terpyridine Ruthenium Complex Efficiently
The synthesis protocol described in the patent provides a reproducible framework for generating high-purity molecular wire precursors suitable for both research and development purposes. The process is designed to be operationally simple, utilizing standard laboratory glassware and common solvents like toluene and methanol, which facilitates easy technology transfer to pilot plants. The key to success lies in the strict control of stoichiometry during the palladium-catalyzed step and the careful management of the recrystallization process to ensure maximum recovery of the intermediate ligand. Detailed standard operating procedures regarding temperature profiles, inert atmosphere requirements, and workup techniques are essential for maintaining batch-to-batch consistency. For technical teams preparing to implement this route, it is crucial to note that the detailed standardized synthesis steps are provided in the guide below to ensure safe and effective execution.
- Perform Pd-catalyzed Buchwald-Hartwig amination between 4'-(4-bromophenyl)-terpyridine and substituted diphenylamine in toluene under reflux.
- Purify the resulting triarylamine-substituted terpyridine intermediate via recrystallization using dichloromethane and n-hexane.
- Coordinate the purified ligand with tris(triphenylphosphine)ruthenium dichloride in methanol under reflux, followed by anion exchange with sodium hexafluorophosphate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers substantial strategic benefits for organizations aiming to optimize their supply chain for advanced electronic materials. The elimination of column chromatography in favor of recrystallization represents a major operational improvement, significantly reducing solvent consumption and waste generation while shortening the overall production cycle time. This simplification directly translates to lower operational expenditures, making the final complex more cost-competitive compared to analogous materials produced via labor-intensive purification methods. Furthermore, the use of commercially available starting materials such as substituted diphenylamines and bromoterpyridines ensures a stable and reliable supply base, mitigating the risks associated with sourcing exotic or custom-synthesized reagents. For procurement managers, this means reduced lead times and greater predictability in raw material availability, which is critical for maintaining continuous production schedules in fast-moving technology sectors.
- Cost Reduction in Manufacturing: The streamlined two-step process minimizes the number of unit operations required to reach the final product, thereby lowering labor and utility costs associated with extended reaction times and complex separations. By avoiding the use of expensive silica gel and large volumes of eluents typically required for chromatography, the process achieves significant savings in consumable materials. Additionally, the high yields reported for both the ligand synthesis and the final coordination step mean that less raw material is wasted, improving the overall atom economy of the process. These efficiencies collectively contribute to a lower cost of goods sold, allowing manufacturers to offer high-purity electronic chemicals at more attractive price points without compromising on quality or performance metrics.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations such as Buchwald-Hartwig amination and ligand exchange ensures that the process is less susceptible to variability and failure modes often seen in novel or unproven methodologies. The use of standard solvents like toluene and methanol, which are widely available in bulk quantities globally, further insulates the supply chain from regional shortages or logistics bottlenecks. This stability is paramount for supply chain heads who must guarantee the uninterrupted delivery of critical materials to downstream device manufacturers. Moreover, the scalability of the reflux conditions means that production can be ramped up from laboratory to industrial scale using existing reactor infrastructure, eliminating the need for capital-intensive equipment upgrades.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by reducing solvent usage and avoiding hazardous reagents where possible. The ability to isolate products via crystallization rather than chromatography significantly reduces the volume of organic waste generated, simplifying waste treatment and disposal compliance. As regulatory pressures on chemical manufacturing continue to intensify, having a synthesis route that minimizes environmental impact provides a distinct competitive advantage. The robustness of the reaction conditions also suggests that the process can be safely operated in large-scale reactors with appropriate engineering controls, ensuring that safety and environmental standards are met even at high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized ruthenium complexes. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this material into their product pipelines. The answers are derived directly from the technical specifications and experimental data provided in the source patent, ensuring accuracy and relevance for decision-makers. For further clarification on specific batch data or customization options, direct consultation with our technical team is recommended.
Q: What are the key advantages of the triarylamine substitution in this ruthenium complex?
A: The triarylamine substitution introduces strong electron-donating capabilities and additional redox-active centers, significantly enhancing electronic interaction between terminal groups compared to unsubstituted analogues.
Q: Is column chromatography required for purifying the intermediate ligand?
A: No, the patented process demonstrates that the intermediate triarylamine-substituted terpyridine can be effectively purified via simple recrystallization using dichloromethane and n-hexane, eliminating the need for costly column chromatography.
Q: Can the chlorine atom on the ruthenium center be further modified?
A: Yes, the chlorine atom on the ruthenium center remains reactive and can be further substituted, providing a versatile pathway for constructing more complex molecular wires with multiple redox-active centers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylamine-Substituted Terpyridine Ruthenium Complex Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise requirements of synthesizing complex organometallic compounds like the triarylamine-substituted terpyridine ruthenium complex described in CN113150036B. We understand that for applications in molecular electronics and optoelectronics, stringent purity specifications are non-negotiable. Our rigorous QC labs employ advanced analytical techniques to verify the structural integrity and electrochemical properties of every batch, ensuring that the material performs consistently in your final devices. Whether you require custom substitution patterns on the triarylamine moiety or specific counterions to tune solubility, our R&D team is ready to adapt the patented route to meet your exact needs.
We invite potential partners to engage with our technical procurement team to discuss how we can support your supply chain goals. By leveraging our manufacturing expertise, you can secure a stable source of high-quality molecular wire materials while benefiting from our commitment to continuous process improvement. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our production capabilities align with your project timelines and quality standards. Let us help you accelerate your development cycle with reliable, scalable, and cost-effective chemical solutions.
