Advanced Catalytic Hydrogenation Route for High-Purity Tofacitinib Citrate Manufacturing
Advanced Catalytic Hydrogenation Route for High-Purity Tofacitinib Citrate Manufacturing
The pharmaceutical industry continuously seeks robust and scalable synthetic pathways for critical small molecule inhibitors, particularly for Janus kinase (JAK) inhibitors like Tofacitinib. Patent CN110724146B discloses a highly efficient preparation method for Tofacitinib Citrate that addresses key bottlenecks in prior art, specifically focusing on reaction yield, operational simplicity, and safety. This novel methodology employs a strategic four-step sequence involving nucleophilic substitution, catalytic hydrogenation, active anhydride acylation, and final salt formation. By leveraging advanced catalytic techniques and optimized reagent selection, this process achieves superior purity profiles while minimizing the use of toxic intermediates. For R&D teams and procurement specialists, understanding this pathway is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials for rheumatoid arthritis treatments.
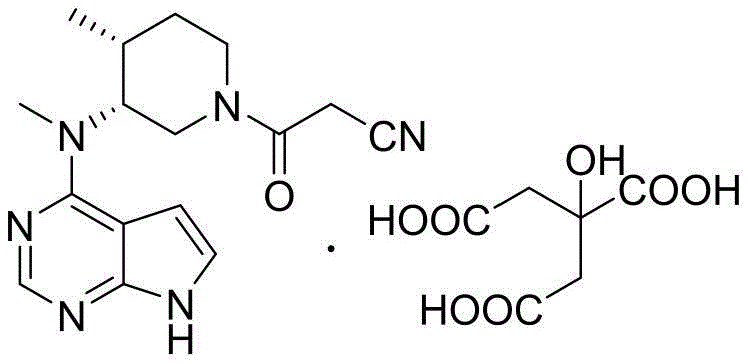
The core innovation lies in the specific manipulation of the side-chain installation and the deprotection strategy. Traditional methods often struggle with the stability of acylating agents or the removal of complex protecting groups. In contrast, the disclosed method utilizes an active acid anhydride generated in situ from cyanoacetic acid and an acyl chloride. This modification drastically improves the electrophilicity of the acylating species, facilitating a smoother reaction with the piperidine amine intermediate. Furthermore, the final salt formation step utilizes an ethanol-water system, which is not only environmentally friendlier but also effective in controlling crystal morphology and purity. This comprehensive approach ensures that the final product meets stringent regulatory standards for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tofacitinib has faced significant challenges related to reagent stability and impurity control. The pioneering route reported by Pfizer, as illustrated in patent WO2001042246A1, relies on cyanoacetic acid-N-hydroxysuccinimide ester for the amidation step. While chemically feasible, this activated ester is inherently unstable and commercially expensive, leading to supply chain vulnerabilities and increased production costs. Additionally, the storage and handling of such sensitive reagents require strict environmental controls, complicating the manufacturing logistics.
Another prevalent method involves the use of p-toluenesulfonyl (tosyl) protecting groups, as seen in patent WO2007012953A2. Although this route attempts to streamline the synthesis, it introduces a critical safety hazard: the potential presence of genotoxic impurities. The hydrolysis of the tosyl group using sodium hydroxide carries a risk of incomplete removal of p-toluenesulfonate derivatives, which are known mutagens. For a commercial API, the burden of proof to demonstrate the absence of these genotoxins below threshold limits adds substantial analytical and processing overhead, making this route less attractive for large-scale cost reduction in API manufacturing.
The Novel Approach
The methodology presented in CN110724146B offers a transformative solution by replacing problematic reagents with more robust alternatives. The key breakthrough is the substitution of the unstable NHS ester with an active acid anhydride. This reagent is prepared simply by reacting cyanoacetic acid with an acyl chloride, both of which are commodity chemicals. This switch not only stabilizes the supply chain but also enhances the reaction kinetics, allowing the acylation to proceed efficiently at temperatures ranging from -20°C to 50°C. The result is a significant improvement in yield and a reduction in reaction time.
Furthermore, the novel approach optimizes the catalytic hydrogenation step. By adjusting the temperature and pressure parameters, the process ensures complete debenzylation without compromising the integrity of the pyrrolo-pyrimidine core. The final crystallization from an ethanol-water mixture eliminates the need for hazardous organic solvents in the finishing stage, aligning with green chemistry principles. This streamlined workflow reduces the total number of unit operations and simplifies the isolation of the final citrate salt, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Active Anhydride Acylation and Catalytic Hydrogenation
The mechanistic superiority of this route centers on the enhanced electrophilicity provided by the active acid anhydride intermediate. In standard amidation reactions using free carboxylic acids, the poor leaving group ability of the hydroxyl moiety often necessitates harsh coupling agents or high temperatures, which can degrade sensitive heterocyclic systems. By converting cyanoacetic acid into a mixed anhydride using an acyl chloride, the leaving group becomes a carboxylate anion, which is far more stable and easier to displace. This electronic activation allows the nucleophilic attack by the secondary amine of the piperidine ring to occur rapidly and selectively, minimizing side reactions such as over-acylation or hydrolysis.
Simultaneously, the catalytic hydrogenation step is engineered for maximum efficiency. The use of palladium-based catalysts (either Pd/C or Pd(OH)2) under controlled hydrogen pressure facilitates the cleavage of the benzyl C-N bond. The patent specifies that elevating the reaction temperature promotes the kinetics of this heterogeneous catalysis, thereby shortening the reaction time significantly. This thermal promotion is critical for preventing the accumulation of partially deprotected intermediates, which could act as difficult-to-remove impurities in downstream processing. The combination of these mechanistic refinements ensures a clean reaction profile with high HPLC purity, often exceeding 99% before the final salt formation.
How to Synthesize Tofacitinib Citrate Efficiently
The synthesis of Tofacitinib Citrate via this patented route is designed to be operationally straightforward, requiring standard equipment found in most multipurpose chemical plants. The process begins with the coupling of the chiral piperidine amine with the dichloro-pyrrolo-pyrimidine scaffold, followed by a critical hydrogenation step to reveal the reactive amine. The subsequent acylation with the active anhydride is the rate-determining step for quality, demanding precise temperature control to maximize yield. Finally, the salt formation is a simple mixing and cooling operation. For detailed laboratory protocols and specific stoichiometric ratios, the standardized synthesis steps are outlined below.
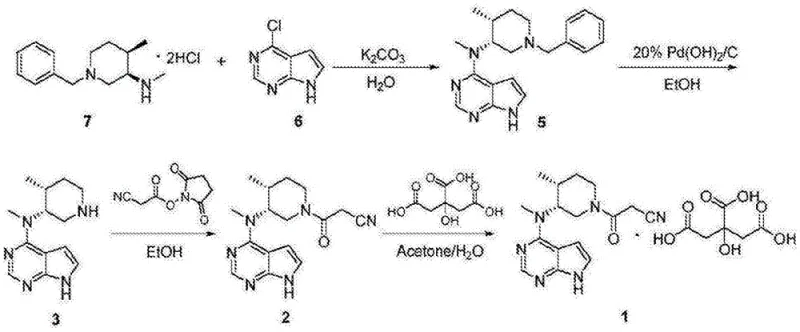
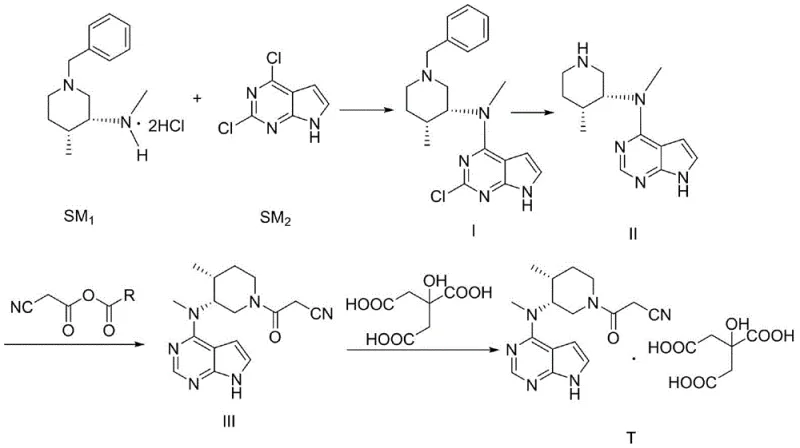
- Perform nucleophilic substitution between (3R,4R)-1-benzyl-N,4-dimethylpiperidine-3-amine dihydrochloride and 2,4-dichloro-7H-pyrrolo[2,3-d]pyrimidine using a base in solvent A to obtain Compound I.
- Execute catalytic hydrogenation on Compound I using a palladium catalyst in solvent B under elevated temperature and pressure to remove the benzyl protecting group, yielding Compound II.
- React Compound II with an active acid anhydride (prepared from cyanoacetic acid and acyl chloride) in solvent C to perform efficient acylation, resulting in Compound III.
- Mix Compound III with citric acid in a solvent mixture of ethanol and water to induce crystallization and form the final Tofacitinib Citrate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible business value. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for specialized, unstable activated esters like NHS-cyanoacetate, the manufacturing process becomes less dependent on niche suppliers who may have long lead times or volatile pricing. Instead, the process relies on bulk commodity chemicals such as cyanoacetic acid, acyl chlorides, and common solvents, which are widely available in the global market. This shift significantly mitigates supply risk and enhances the overall resilience of the production schedule.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the replacement of expensive coupling reagents with cost-effective active anhydrides. The in situ generation of the acylating agent removes the need for purchasing pre-activated, high-cost intermediates, leading to substantial raw material savings. Additionally, the improved reaction yields and reduced reaction times lower the consumption of utilities and solvent volumes per kilogram of product. The elimination of complex purification steps required to remove genotoxic tosyl impurities further reduces waste disposal costs and analytical testing burdens, contributing to a leaner cost structure.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted supply to downstream API manufacturers. The use of stable intermediates means that materials can be stockpiled without significant degradation, providing a buffer against market fluctuations. Moreover, the simplified workflow reduces the likelihood of batch failures due to reagent instability, thereby improving the predictability of delivery timelines. This reliability is essential for partners seeking a reliable pharmaceutical intermediate supplier for long-term contracts.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route offers distinct advantages for large-scale operations. The avoidance of genotoxic reagents simplifies the containment requirements and reduces the hazard classification of the manufacturing site. The final salt formation using ethanol and water minimizes the discharge of halogenated solvents, aligning with increasingly strict environmental regulations. The process is inherently scalable, as the exothermic nature of the reactions can be managed effectively in larger reactors, ensuring that production capacity can be expanded from pilot scale to multi-ton annual output without fundamental process changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Tofacitinib Citrate using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of the process for potential partners. Understanding these details is key to evaluating the technology for integration into your existing supply chain.
Q: How does the active anhydride method improve yield compared to traditional NHS ester routes?
A: The use of active acid anhydrides significantly enhances the electrophilicity of the reactant compared to cyanoacetic acid or less stable NHS esters. This allows for rapid and efficient aminolysis at mild temperatures (-20°C to 50°C), leading to higher conversion rates and simplified purification processes.
Q: What are the safety advantages of this synthesis route regarding genotoxic impurities?
A: Unlike routes utilizing p-toluenesulfonyl protecting groups which carry a risk of residual genotoxic p-toluenesulfonate impurities, this method avoids such hazardous reagents entirely. The process relies on benign benzyl protection which is cleanly removed via hydrogenation, ensuring a safer impurity profile for the final API.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes readily available starting materials, common solvents like ethanol and water for the final salt formation, and robust catalytic hydrogenation steps that can be easily optimized for reactor volume, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tofacitinib Citrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for high-value pharmaceutical intermediates. Our technical team has extensively analyzed the route disclosed in CN110724146B and confirmed its viability for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. Our facilities are equipped with state-of-the-art hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of Tofacitinib intermediate meets the highest international standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and secure a competitive edge in the market for JAK inhibitor therapies.
